
A chemical bond is a lasting attraction between atoms, ions or molecules that enables the formation of chemical compounds. The bond may result from the electrostatic force of attraction between oppositely charged ions as in ionic bonds or through the sharing of electrons as in covalent bonds. The strength of chemical bonds varies considerably; there are "strong bonds" or "primary bonds" such as covalent, ionic and metallic bonds, and "weak bonds" or "secondary bonds" such as dipole–dipole interactions, the London dispersion force and hydrogen bonding.

A hydrogen bond is a partially electrostatic force of attraction between a hydrogen (H) atom which is bound to a more electronegative atom or group, such as nitrogen (N), oxygen (O), or fluorine (F)—the hydrogen bond donor—and another adjacent atom bearing a lone pair of electrons—the hydrogen bond acceptor. Weak hydrogen bonds occur even with C-H groups as donor
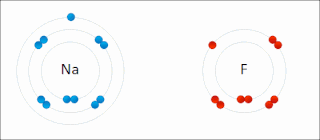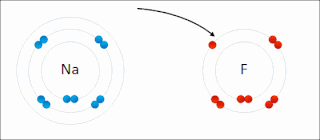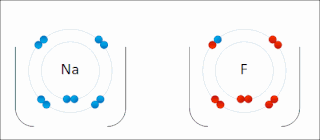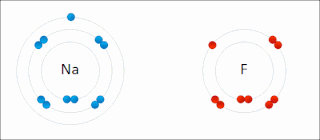
Ionic bonding is a type of chemical bonding that involves the electrostatic attraction between oppositely charged ions, and is the primary interaction occurring in ionic compounds. It is one of the main bonds along with Covalent bond and Metallic bonding. Ions are atoms that have gained one or more electrons and atoms that have lost one or more electrons. This transfer of electrons is known as electrovalence in contrast to covalence. In the simplest case, the cation is a metal atom and the anion is a nonmetal atom, but these ions can be of a more complex nature, e.g. molecular ions like NH+
4 or SO2−
4. In simpler words, an ionic bond is the transfer of electrons from a metal to a non-metal in order to obtain a full valence shell for both atoms.
A coordinate covalent bond, also known as a dative bond or coordinate bond is a kind of 2-center, 2-electron covalent bond in which the two electrons derive from the same atom. The bonding of metal ions to ligands involves this kind of interaction. This type of interaction is central to Lewis acid-base theory.

In chemistry, polarity is a separation of electric charge leading to a molecule or its chemical groups having an electric dipole moment, with a negatively charged end and a positively charged end.

A triple bond in chemistry is a chemical bond between two atoms involving six bonding electrons instead of the usual two in a covalent single bond. The most common triple bond, that between two carbon atoms, can be found in alkynes.
Other functional groups containing a triple bond are cyanides and isocyanides. Some diatomic molecules, such as dinitrogen and carbon monoxide, are also triple bonded. In skeletal formula the triple bond is drawn as three parallel lines (≡) between the two connected atoms.
In chemistry, delocalized electrons are electrons in a molecule, ion or solid metal that are not associated with a single atom or a covalent bond.
The term is general and can have slightly different meanings in different fields. In organic chemistry, this refers to resonance in conjugated systems and aromatic compounds. In solid-state physics, this refers to free electrons that facilitate electrical conduction. In quantum chemistry, this refers to molecular orbital electrons that extend over several adjacent atoms.
In chemistry, valence bond (VB) theory is one of two basic theories, along with molecular orbital (MO) theory, that were developed to use the methods of quantum mechanics to explain chemical bonding. It focuses on how the atomic orbitals of the dissociated atoms combine to give individual chemical bonds when a molecule is formed. In contrast, molecular orbital theory has orbitals that cover the whole molecule.

In chemistry, a single bond is a chemical bond between two atoms involving two valence electrons. That is, the atoms share one pair of electrons where the bond forms. Therefore, a single bond is a type of covalent bond. When shared, each of the two electrons involved is no longer in the sole possession of the orbital in which it originated. Rather, both of the two electrons spend time in either of the orbitals which overlap in the bonding process. As a Lewis structure, a single bond is denoted as AːA or A-A, for which A represents an element. In the first rendition, each dot represents a shared electron, and in the second rendition, the bar represents both of the electrons shared in the single bond.
The atomic radius of a chemical element is the distance from the centre of the nucleus to the outermost shell of the electron. Since the boundary is not a well-defined physical entity, there are various non-equivalent definitions of atomic radius. Depending on the definition, the term may apply only to isolated atoms, or also to atoms in condensed matter, covalently bound in molecules, or in ionized and excited states; and its value may be obtained through experimental measurements, or computed from theoretical models. Under some definitions, the value of the radius may depend on the atom's state and context.
In chemistry, bond energy (E) or bond enthalpy (H) is the measure of bond strength in a chemical bond. IUPAC defines bond energy as the average value of the gas-phase bond dissociation energies (usually at a temperature of 298 K) for all bonds of the same type within the same chemical species. For example, the carbon–hydrogen bond energy in methane H(C–H) is the enthalpy change involved with breaking up one molecule of methane into a carbon atom and four hydrogen radicals, divided by 4. Tabulated bond energies are generally values of bond energies averaged over a number of selected typical chemical species containing that type of bond. Bond energy (E) or bond enthalpy (H) should not be confused with bond-dissociation energy. Bond energy is the average of all the bond-dissociation energies in a molecule, and will show a different value for a given bond than the bond-dissociation energy would. This is because the energy required to break a single bond in a specific molecule differs for each bond in that molecule. For example, methane has four C–H bonds and the bond-dissociation energies are 435 kJ/mol for D(CH3–H), 444 kJ/mol for D(CH2–H), 444 kJ/mol for D(CH–H) and 339 kJ/mol for D(C–H). Their average, and hence the bond energy, is 414 kJ/mol, even though not a single bond required specifically 414 kJ/mol to be broken.
In chemistry, heterolysis or heterolytic fission is the process of cleaving a covalent bond where one previously bonded species takes both original bonding electrons from the other species. During heterolytic bond cleavage of a neutral molecule, a cation and an anion will be generated. Most commonly the more electronegative atom keeps the pair of electrons becoming anionic while the more electropositive atom becomes cationic.
A non-covalent interaction differs from a covalent bond in that it does not involve the sharing of electrons, but rather involves more dispersed variations of electromagnetic interactions between molecules or within a molecule. The chemical energy released in the formation of non-covalent interactions is typically on the order of 1-5 kcal/mol. Non-covalent interactions can be classified into different categories, such as electrostatic, π-effects, van der Waals forces, and hydrophobic effects.
An intramolecular force is any force that binds together the atoms making up a molecule or compound, not to be confused with intermolecular forces, which are the forces present between molecules. The subtle difference in the name comes from the Latin roots of English with inter meaning between or among and intra meaning inside. Chemical bonds are considered to be intramolecular forces, for example. These forces are often stronger than intermolecular forces, which are present between atoms or molecules that are not bonded.
Molecular binding is an attractive interaction between two molecules that results in a stable association in which the molecules are in close proximity to each other. It often but not always involves some chemical bonding.

A chemical compound is a chemical substance composed of many identical molecules composed of atoms from more than one element held together by chemical bonds. A chemical element bonded to an identical chemical element is not a chemical compound since only one element, not two different elements, is involved.
Solids can be classified according to the nature of the bonding between their atomic or molecular components. The traditional classification distinguishes four kinds of bonding:
The charge-shift bond has been proposed as a new class of chemical bond that sits alongside the three familiar families of covalent, ionic bonds, and metallic bonds where electrons are shared or transferred respectively. The charge shift bond derives its stability from the resonance of ionic forms rather than the covalent sharing of electrons which are often depicted as having electron density between the bonded atoms. A feature of the charge shift bond is that the predicted electron density between the bonded atoms is low. It has long been known from experiment that the accumulation of electronic charge between the bonded atoms is not necessarily a feature of covalent bonds. An example where charge shift bonding has been used to explain the low electron density found experimentally is in the central bond between the inverted tetrahedral carbons in [1.1.1]propellanes. Theoretical calculations on a range of molecules have indicated that a charge shift bond is present, a striking example being fluorine, F2, which is normally described as having a typical covalent bond.








