In organic chemistry, an electrophile is a reagent attracted to electrons. Electrophiles are positively charged or neutral species having vacant orbitals that are attracted to an electron rich centre. It participates in a chemical reaction by accepting an electron pair in order to bond to a nucleophile. Because electrophiles accept electrons, they are Lewis acids. Most electrophiles are positively charged, have an atom that carries a partial positive charge, or have an atom that does not have an octet of electrons. They appear to attract electrons as well and seem to behave as though they are partially empty. These partially empty substances thus require an electron rich center, and thus they are filled. Electrophiles can be observed as electron-sensitive or photo-sensitive.

George Andrew Olah was a Hungarian and American chemist. His research involved the generation and reactivity of carbocations via superacids. For this research, Olah was awarded a Nobel Prize in Chemistry in 1994 "for his contribution to carbocation chemistry." He was also awarded the Priestley Medal, the highest honor granted by the American Chemical Society and F.A. Cotton Medal for Excellence in Chemical Research of the American Chemical Society in 1996. According to György Marx he was one of The Martians.

Tetrahedrane is a platonic hydrocarbon with chemical formula C4H4 and a tetrahedral structure. The molecule is subject to considerable angle strain. A number of derivatives have been prepared. Tetrahedranes describe a class of molecules and ions with related structures, e.g. white phosphorus.
In organic chemistry, Hückel's rule estimates whether a planar ring molecule will have aromatic properties. The quantum mechanical basis for its formulation was first worked out by physical chemist Erich Hückel in 1931. The succinct expression as the 4n + 2 rule has been attributed to W. v. E. Doering (1951), although several authors were using this form at around the same time.
Dodecahedrane is a chemical compound (C20H20) first synthesised by Leo Paquette of Ohio State University in 1982, primarily for the "aesthetically pleasing symmetry of the dodecahedral framework". It is the simplest hydrocarbon with full icosahedral symmetry.
Barbituric acid or malonylurea or 6-hydroxyuracil is an organic compound based on a pyrimidine heterocyclic skeleton. It is an odorless powder soluble in water. Barbituric acid is the parent compound of barbiturate drugs, although barbituric acid itself is not pharmacologically active. The compound was first synthesised by Adolf von Baeyer.

The Baeyer–Villiger oxidation is an organic reaction that forms an ester from a ketone or a lactone from a cyclic ketone, using peroxyacids or peroxides as the oxidant. The reaction is named after Adolf von Baeyer and Victor Villiger who first reported the reaction in 1899.
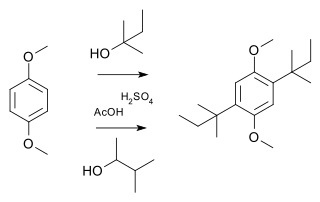
A Wagner–Meerwein rearrangement is a class of carbocation 1,2-rearrangement reactions in which a hydrogen, alkyl or aryl group migrates from one carbon to a neighboring carbon. They can be described as cationic [1,2]-sigmatropic rearrangements, proceeding suprafacially and with stereochemical retention. As such, a Wagner–Meerwein shift is a thermally allowed pericyclic process with the Woodward-Hoffmann symbol [ω0s + σ2s]. They are usually facile, and in many cases, they can take place at temperatures as low as –120 °C. The reaction is named after the Russian chemist Yegor Yegorovich Vagner; he had German origin and published in German journals as Georg Wagner; and Hans Meerwein.

The Prato reaction is a particular example of the well-known 1,3-dipolar cycloaddition of azomethine ylides to olefins. In fullerene chemistry this reaction refers to the functionalization of fullerenes and nanotubes. The amino acid sarcosine reacts with paraformaldehyde when heated at reflux in toluene to an ylide which reacts with a double bond in a 6,6 ring position in a fullerene via a 1,3-dipolar cycloaddition to yield a N-methylpyrrolidine derivative or pyrrolidinofullerene or pyrrolidino[[3,4:1,2]] [60]fullerene in 82% yield based on C60 conversion.
The term "gauche" refers to conformational isomers (conformers) where two vicinal groups are separated by a torsion angle of 60°. IUPAC defines groups as gauche if they have a "synclinal alignment of groups attached to adjacent atoms".

Homoaromaticity, in organic chemistry, refers to a special case of aromaticity in which conjugation is interrupted by a single sp3 hybridized carbon atom. Although this sp3 center disrupts the continuous overlap of p-orbitals, traditionally thought to be a requirement for aromaticity, considerable thermodynamic stability and many of the spectroscopic, magnetic, and chemical properties associated with aromatic compounds are still observed for such compounds. This formal discontinuity is apparently bridged by p-orbital overlap, maintaining a contiguous cycle of π electrons that is responsible for this preserved chemical stability.

In organometallic chemistry, a sandwich compound is a chemical compound featuring a metal bound by haptic covalent bonds to two arene ligands. The arenes have the formula CnHn, substituted derivatives (for example Cn(CH3)n) and heterocyclic derivatives (for example BCnHn+1). Because the metal is usually situated between the two rings, it is said to be "sandwiched". A special class of sandwich complexes are the metallocenes.

Fullerene chemistry is a field of organic chemistry devoted to the chemical properties of fullerenes. Research in this field is driven by the need to functionalize fullerenes and tune their properties. For example, fullerene is notoriously insoluble and adding a suitable group can enhance solubility. By adding a polymerizable group, a fullerene polymer can be obtained. Functionalized fullerenes are divided into two classes: exohedral fullerenes with substituents outside the cage and endohedral fullerenes with trapped molecules inside the cage.
William Henry Perkin Jr., FRS HFRSE was an English organic chemist who was primarily known for his groundbreaking research work on the degradation of naturally occurring organic compounds.

Anthony Joseph Arduengo III is the Saxon Professor Emeritus of Chemistry at the University of Alabama, an adjunct professor at the Institute for Inorganic Chemistry of Braunschweig University of Technology in Germany, and co-founder of the StanCE coalition for sustainable chemistry based on woody biomass. He is notable for his work on chemical compounds with unusual valency, especially in the field of stable carbene research.

The Society of German Chemists is a learned society and professional association founded in 1949 to represent the interests of German chemists in local, national and international contexts. GDCh "unites those people associated with the chemical and molecular sciences and supports them in their responsible and sustainable endeavors for the good of the public and of our environment."
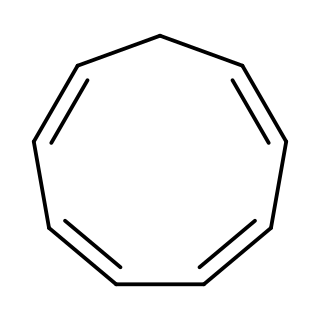
Cyclononatetraene is an organic compound with the formula C9H10. It was first prepared in 1969 by protonation of the corresponding aromatic anion (described below). It is unstable and isomerizes with a half-life of 50 min at room temperature to 3a,7a-dihydro-1H-indene via a thermal 6π disrotatory electrocyclic ring closing. Upon exposure to ultraviolet light, it undergoes a photochemical 8π electrocyclic ring closing to give bicyclo[6.1.0]nona-2,4,6-triene.

Maurizio Prato, is an Italian Organic Chemist, who is best known for his work on the functionalization of carbon nanostructures, including fullerenes, carbon nanotubes and graphene. He developed a series of organic reactions that make these materials more biocompatible, less or even non toxic, amenable to further functionalization, and easier to manipulate. He is Professor of Organic Chemistry at the University of Trieste and Research Professor at CIC BiomaGUNE in San Sebastián, Spain.













