According to the classical definition, a superacid is an acid with an acidity greater than that of 100% pure sulfuric acid, which has a Hammett acidity function (H0) of −12. According to the modern definition, a superacid is a medium in which the chemical potential of the proton is higher than in pure sulfuric acid. Commercially available superacids include trifluoromethanesulfonic acid (CF3SO3H), also known as triflic acid, and fluorosulfuric acid (HSO3F), both of which are about a thousand times stronger (i.e. have more negative H0 values) than sulfuric acid. Most strong superacids are prepared by the combination of a strong Lewis acid and a strong Brønsted acid. A strong superacid of this kind is fluoroantimonic acid. Another group of superacids, the carborane acid group, contains some of the strongest known acids.

Antimony trisulfide (Sb2S3) is found in nature as the crystalline mineral stibnite and the amorphous red mineral metastibnite. It is manufactured for use in safety matches, military ammunition, explosives and fireworks. It also is used in the production of ruby-colored glass and in plastics as a flame retardant. Historically the stibnite form was used as a grey pigment in paintings produced in the 16th century. It is a semiconductor with a direct band gap of 1.8-2.5 eV. With suitable doping, p and n type materials can be produced.

Stibine (IUPAC name: stibane) is a chemical compound with the formula SbH3. A pnictogen hydride, this colourless gas is the principal covalent hydride of antimony, and a heavy analogue of ammonia. The molecule is pyramidal with H–Sb–H angles of 91.7° and Sb–H distances of 170.7 pm (1.707 Å). This gas has an offensive smell like hydrogen sulfide (rotten eggs).

Antimony(III) oxide is the inorganic compound with the formula Sb2O3. It is the most important commercial compound of antimony. It is found in nature as the minerals valentinite and senarmontite. Like most polymeric oxides, Sb2O3 dissolves in aqueous solutions with hydrolysis.

Sodium thioantimoniate is an inorganic compound with the formula Na3SbS4. The nonahydrate of this material is known as Schlippe's salt, named after K. F. Schlippe (1799–1867), These compounds are examples of sulfosalts. They were once of interest as species generated in qualitative inorganic analysis.
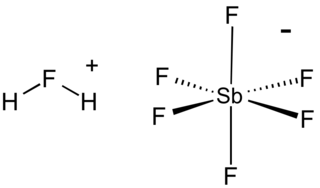
Fluoroantimonic acid is an inorganic compound with the chemical formula H
2FSbF
6 (also written H
2F[SbF
6], 2HF·SbF5, or simply HF-SbF5). It is an extremely strong acid, easily qualifying as a superacid. The Hammett acidity function, H0, has been measured for different ratios of HF:SbF5. While the H0 of pure HF is −15, addition of just 1 mol % of SbF5 lowers it to around −20. However, further addition of SbF5 results in rapidly diminishing returns, with the H0 reaching −21 at 10 mol %. The use of an extremely weak base as indicator shows that the lowest attainable H0, even with > 50 mol % SbF5, is somewhere between −21 and −23.

Antimony oxychloride, known since the 15th century, has been known by a plethora of alchemical names. Since the compound functions as both an emetic and a laxative, it was originally used as a purgative.

Antimony pentachloride is a chemical compound with the formula SbCl5. It is a colourless oil, but typical samples are yellowish due to impurities. Owing to its tendency to hydrolyse to hydrochloric acid, SbCl5 is a highly corrosive substance and carbonizes non-fluorinated plastics.

Antimony pentoxide (molecular formula: Sb2O5) is a chemical compound of antimony and oxygen. It always occurs in hydrated form, Sb2O5·nH2O. It contains antimony in the +5 oxidation state.

Antimony trichloride is the chemical compound with the formula SbCl3. The soft colorless solid with a pungent odor was known to the alchemists as butter of antimony.
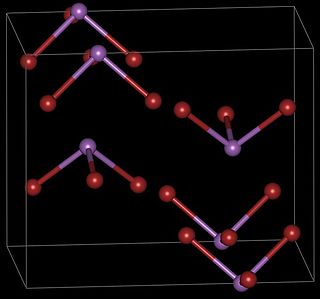
Antimony tribromide (SbBr3) is a chemical compound containing antimony in its +3 oxidation state.

Antimony triiodide is the chemical compound with the formula SbI3. This ruby-red solid is the only characterized "binary" iodide of antimony, i.e. the sole compound isolated with the formula SbxIy. It contains antimony in its +3 oxidation state. Like many iodides of the heavier main group elements, its structure depends on the phase. Gaseous SbI3 is a molecular, pyramidal species as anticipated by VSEPR theory. In the solid state, however, the Sb center is surrounded by an octahedron of six iodide ligands, three of which are closer and three more distant. For the related compound BiI3, all six Bi—I distances are equal.

Antimony(III) acetate is the compound of antimony with the chemical formula of Sb(CH3CO2)3. It is a white powder, is moderately water-soluble, and is used as a catalyst in the production of polyesters.
Antimony pentasulfide is an inorganic compound of antimony and sulfur, also known as antimony red. It is a nonstoichiometric compound with a variable composition. Its exact structure is unknown. Commercial samples are usually contaminated with sulfur, which may be removed by washing with carbon disulfide in a Soxhlet extractor.

Antimony tetroxide is an inorganic compound with the formula Sb2O4. This material, which exists as the mineral cervantite, is white but reversibly yellows upon heating. The material, with empirical formula SbO2, is called antimony tetroxide to signify the presence of two kinds of Sb centers.
Organoantimony chemistry is the chemistry of compounds containing a carbon to antimony (Sb) chemical bond. Relevant oxidation states are Sb(V) and Sb(III). The toxicity of antimony limits practical application in organic chemistry.
Antimony sulfate, Sb2(SO4)3, is a hygroscopic material is formed by reacting antimony or its compounds with hot sulfuric acid. It is used in doping of semiconductors and in the production of explosives and fireworks.












