
A monoclonal antibody is an antibody produced from a cell lineage made by cloning a unique white blood cell. All subsequent antibodies derived this way trace back to a unique parent cell.

Niels Kaj Jerne, FRS was a Danish immunologist. He shared the Nobel Prize in Physiology or Medicine in 1984 with Georges J. F. Köhler and César Milstein "for theories concerning the specificity in development and control of the immune system and the discovery of the principle for production of monoclonal antibodies".
In immunology, antiserum is a blood serum containing antibodies that is used to spread passive immunity to many diseases via blood donation (plasmapheresis). For example, convalescent serum, passive antibody transfusion from a previous human survivor, used to be the only known effective treatment for ebola infection with a high success rate of 7 out of 8 patients surviving.

Hybridoma technology is a method for producing large numbers of identical antibodies. This process starts by injecting a mouse with an antigen that provokes an immune response. A type of white blood cell, the B cell, produces antibodies that bind to the injected antigen. These antibody producing B-cells are then harvested from the mouse and, in turn, fused with immortal myeloma cancer cells, to produce a hybrid cell line called a hybridoma, which has both the antibody-producing ability of the B-cell and the longevity and reproductivity of the myeloma. The hybridomas can be grown in culture, each culture starting with one viable hybridoma cell, producing cultures each of which consists of genetically identical hybridomas which produce one antibody per culture (monoclonal) rather than mixtures of different antibodies (polyclonal). The myeloma cell line that is used in this process is selected for its ability to grow in tissue culture and for an absence of antibody synthesis. In contrast to polyclonal antibodies, which are mixtures of many different antibody molecules, the monoclonal antibodies produced by each hybridoma line are all chemically identical.

Georges Jean Franz Köhler was a German biologist.

Sir Gregory Paul Winter is a Nobel Prize-winning English molecular biologist best known for his work on the therapeutic use of monoclonal antibodies. His research career has been based almost entirely at the MRC Laboratory of Molecular Biology and the MRC Centre for Protein Engineering, in Cambridge, England.
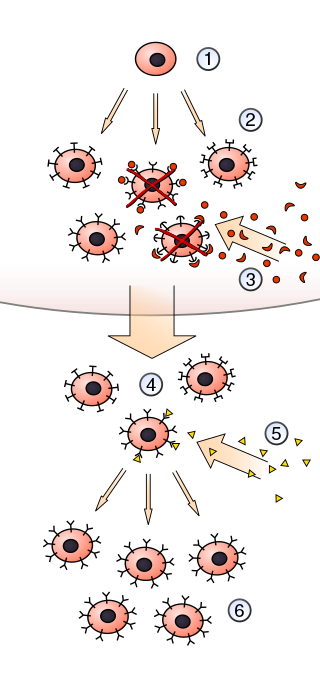
In immunology, clonal selection theory explains the functions of cells of the immune system (lymphocytes) in response to specific antigens invading the body. The concept was introduced by Australian doctor Frank Macfarlane Burnet in 1957, in an attempt to explain the great diversity of antibodies formed during initiation of the immune response. The theory has become the widely accepted model for how the human immune system responds to infection and how certain types of B and T lymphocytes are selected for destruction of specific antigens.

HAT Medium is a selection medium for mammalian cell culture, which relies on the combination of aminopterin, a drug that acts as a powerful folate metabolism inhibitor by inhibiting dihydrofolate reductase, with hypoxanthine and thymidine which are intermediates in DNA synthesis. The trick is that aminopterin blocks DNA de novo synthesis, which is absolutely required for cell division to proceed, but hypoxanthine and thymidine provide cells with the raw material to evade the blockage, provided that they have the right enzymes, which means having functioning copies of the genes that encode them.

The Medical Research Council (MRC) Laboratory of Molecular Biology (LMB) is a research institute in Cambridge, England, involved in the revolution in molecular biology which occurred in the 1950–60s. Since then it has remained a major medical research laboratory at the forefront of scientific discovery, dedicated to improving the understanding of key biological processes at atomic, molecular and cellular levels using multidisciplinary methods, with a focus on using this knowledge to address key issues in human health.
The following are notable events in the Timeline of immunology:
The nomenclature of monoclonal antibodies is a naming scheme for assigning generic, or nonproprietary, names to monoclonal antibodies. An antibody is a protein that is produced in B cells and used by the immune system of humans and other vertebrate animals to identify a specific foreign object like a bacterium or a virus. Monoclonal antibodies are those that were produced in identical cells, often artificially, and so share the same target object. They have a wide range of applications including medical uses.

Monoclonal antibodies (mAbs) have varied therapeutic uses. It is possible to create a mAb that binds specifically to almost any extracellular target, such as cell surface proteins and cytokines. They can be used to render their target ineffective, to induce a specific cell signal, to cause the immune system to attack specific cells, or to bring a drug to a specific cell type.

In immunology, an idiotype is a shared characteristic between a group of immunoglobulin or T-cell receptor (TCR) molecules based upon the antigen binding specificity and therefore structure of their variable region. The variable region of antigen receptors of T cells (TCRs) and B cells (immunoglobulins) contain complementarity-determining regions (CDRs) with unique amino acid sequences. They define the surface and properties of the variable region, determining the antigen specificity and therefore the idiotope of the molecule. Immunoglobulins or TCRs with a shared idiotope are the same idiotype. Antibody idiotype is determined by:

Brigitte Alice Askonas was a British immunologist and a visiting professor at Imperial College London from 1995.
A rabbit hybridoma is a hybrid cell line formed by the fusion of an antibody producing rabbit B cell with a cancerous B-cell (myeloma).
Jerrold Schwaber was an American biologist and geneticist. In 1973 he described, with Edward Cohen, a method of producing antibodies involving human–mouse hybrid cells, or hybridomas. They fused "mouse myeloma cells secreting immunoglobulin of known specificity and human peripheral blood lymphocytes not secreting detectable immunoglobulin. The hybrid cells continued secretion of mouse immunoglobulin and initiate synthesis and secretion of human immunoglobulin." The antibody producing cells did not survive long and the antigens that the antibodies targeted remained unknown. In 1975, Georges Köhler, César Milstein, and Niels Kaj Jerne, succeeded in making hybridomas that made antibodies to known antigens and that were immortalized. They shared the Nobel Prize in Physiology or Medicine in 1984 for the discovery. His work in laying the foundation for modern monoclonal antibody technology is recognized.
Elizabeth Marian Press was a British immunologist, best known for her work with Rodney Porter on the structure of antibodies. She worked side by side with Porter for 25 years, at the National Institute for Medical Research, St Mary's Hospital and in the Medical Research Council Immunochemistry Unit, and played a major role in him being awarded the Nobel Prize in 1972.
Recombinant antibodies are antibody fragments produced by using recombinant antibody coding genes. They mostly consist of a heavy and light chain of the variable region of immunoglobulin. Recombinant antibodies have many advantages in both medical and research applications, which make them a popular subject of exploration and new production against specific targets. The most commonly used form is the single chain variable fragment (scFv), which has shown the most promising traits exploitable in human medicine and research. In contrast to monoclonal antibodies produced by hybridoma technology, which may lose the capacity to produce the desired antibody over time or the antibody may undergo unwanted changes, which affect its functionality, recombinant antibodies produced in phage display maintain high standard of specificity and low immunogenicity.

Terry William Pearson is a Canadian biochemist, immunologist, educator and biotechnology entrepreneur.











