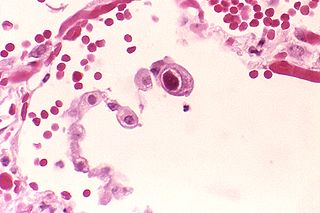
Cytomegalovirus (CMV) is a genus of viruses in the order Herpesvirales, in the family Herpesviridae, in the subfamily Betaherpesvirinae. Humans and other primates serve as natural hosts. The 11 species in this genus include human betaherpesvirus 5, which is the species that infects humans. Diseases associated with HHV-5 include mononucleosis and pneumonia. In the medical literature, most mentions of CMV without further specification refer implicitly to human CMV. Human CMV is the most studied of all cytomegaloviruses.
Virus latency is the ability of a pathogenic virus to lie dormant within a cell, denoted as the lysogenic part of the viral life cycle. A latent viral infection is a type of persistent viral infection which is distinguished from a chronic viral infection. Latency is the phase in certain viruses' life cycles in which, after initial infection, proliferation of virus particles ceases. However, the viral genome is not eradicated. The virus can reactivate and begin producing large amounts of viral progeny without the host becoming reinfected by new outside virus, and stays within the host indefinitely.

Human herpesvirus 6 (HHV-6) is the common collective name for human betaherpesvirus 6A (HHV-6A) and human betaherpesvirus 6B (HHV-6B). These closely related viruses are two of the nine known herpesviruses that have humans as their primary host.

Herpesviridae is a large family of DNA viruses that cause infections and certain diseases in animals, including humans. The members of this family are also known as herpesviruses. The family name is derived from the Greek word ἕρπειν, referring to spreading cutaneous lesions, usually involving blisters, seen in flares of herpes simplex 1, herpes simplex 2 and herpes zoster (shingles). In 1971, the International Committee on the Taxonomy of Viruses (ICTV) established Herpesvirus as a genus with 23 viruses among four groups. As of 2020, 115 species are recognized, all but one of which are in one of the three subfamilies. Herpesviruses can cause both latent and lytic infections.

Herpes simplex virus1 and 2, also known by their taxonomical names Human alphaherpesvirus 1 and Human alphaherpesvirus 2, are two members of the human Herpesviridae family, a set of viruses that produce viral infections in the majority of humans. Both HSV-1 and HSV-2 are very common and contagious. They can be spread when an infected person begins shedding the virus.

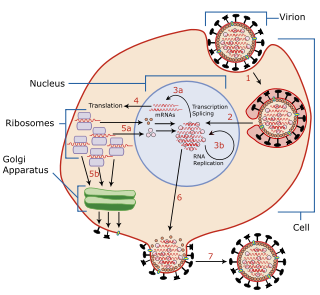
Viral entry is the earliest stage of infection in the viral life cycle, as the virus comes into contact with the host cell and introduces viral material into the cell. The major steps involved in viral entry are shown below. Despite the variation among viruses, there are several shared generalities concerning viral entry.

B-virus, Herpesvirus simiae, or Herpes virus B is the Simplexvirus infecting macaque monkeys. B virus is very similar to HSV-1, and as such, this neurotropic virus is not found in the blood.

Poliovirus receptor-related 2 (PVRL2), also known as nectin-2 and CD112, is a human plasma membrane glycoprotein.

Poliovirus receptor-related 1 (PVRL1), also known as nectin-1 and CD111 (formerly herpesvirus entry mediator C, HVEC) is a human protein of the immunoglobulin superfamily (IgSF), also considered a member of the nectins. It is a membrane protein with three extracellular immunoglobulin domains, a single transmembrane helix and a cytoplasmic tail. The protein can mediate Ca2+-independent cellular adhesion further characterizing it as IgSF cell adhesion molecule (IgSF CAM).

Herpesvirus entry mediator (HVEM), also known as tumor necrosis factor receptor superfamily member 14 (TNFRSF14), is a human cell surface receptor of the TNF-receptor superfamily.
Anthony (Tony) Charles Minson, PhD, FMedSci is a British virologist known for his work on the biology of herpesviruses, and a university administrator. He was the Senior Pro-Vice-Chancellor of the University of Cambridge from 2003 to 2009. He is emeritus professor of virology at the university's Department of Pathology and an emeritus fellow of Wolfson College.
Human betaherpesvirus 7 (HHV-7) is one of nine known members of the Herpesviridae family that infects humans. HHV-7 is a member of Betaherpesvirinae, a subfamily of the Herpesviridae that also includes HHV-6 and Cytomegalovirus. HHV-7 often acts together with HHV-6, and the viruses together are sometimes referred to by their genus, Roseolovirus. HHV-7 was first isolated in 1990 from CD4+ T cells taken from peripheral blood lymphocytes.
Deepak Shukla is an American molecular virologist with expertise in herpesviruses. He contributed to the discovery of HSV-1 entry receptors and establishing a link between the receptors and HSV-1 induced ocular diseases such as keratitis and retinitis. He has authored over 100 published papers and several book chapters on herpes viruses.

Herpesvirus glycoprotein B is a viral glycoprotein that is involved in the viral cell entry of Herpes simplex virus (HSV). Herpesviruses have a lipid bilayer, called the envelope, which contains twelve surface glycoproteins. For infectivity to be attained, the double stranded DNA genome of HSV must enter the host cell through means of fusion of its envelope with the cellular membrane or via endocytosis. Other viral glycoproteins involved in the process of viral cell entry include gC, gB, gD, gH, and gL, but only gC, gB, gD, and gH are required for the fusion of the HSV's envelope with the cellular membrane. It can be noted that all herpesviruses have glycoproteins gB, gH, and gL.
Herpes simplex research includes all medical research that attempts to prevent, treat, or cure herpes, as well as fundamental research about the nature of herpes. Examples of particular herpes research include drug development, vaccines and genome editing. HSV-1 and HSV-2 are commonly thought of as oral and genital herpes respectively, but other members in the herpes family include chickenpox (varicella/zoster), cytomegalovirus, and Epstein-Barr virus. There are many more virus members that infect animals other than humans, some of which cause disease in companion animals or have economic impacts in the agriculture industry.
David Mahan Knipe is the Higgins Professor of Microbiology and Molecular Genetics in the Department of Microbiology at the Harvard Medical School in Boston, Massachusetts and co-chief editor of the reference book Fields Virology. He returned to the Chair of the Program in Virology at Harvard Medical School in 2019, having previously held the position from 2004 through 2016 and served as interim Co-Chair of the Microbiology and Immunobiology Department from 2016 through 2018.
Almyra Oveta Fuller was an associate professor of microbiology and immunology at University of Michigan Medical School. She served as the director of the African Studies Center (ASC), faculty in the ASC STEM Initiative at the University of Michigan (U-M) and an adjunct professor at Payne Theological Seminary. Fuller was a virologist and specialized in research of Herpes simplex virus, as well as HIV/AIDS. Fuller and her research team discovered a B5 receptor, advancing the understanding of Herpes simplex virus and the cells it attacks.
Roselyn J. Eisenberg is a professor at The University of Pennsylvania and a member of the University's School of Veterinary Medicine and School of Dental Medicine. The majority of Eisenberg's research is focused on the herpes simplex virus and the poxvirus and how they enter into susceptible cells. She also studies glycoproteins, vaccines, virology and microbiology.
Human herpes viruses, also known as HHVs, are part of a family of DNA viruses that cause several diseases in humans. One of the most notable functions of this virus family is their ability to enter a latent phase and lay dormant within animals for extended periods of time. The mechanism that controls this is very complex because expression of viral proteins during latency is decreased a great deal, meaning that the virus must have transcription of its genes repressed. There are many factors and mechanisms that control this process and epigenetics is one way this is accomplished. Epigenetics refers to persistent changes in expression patterns that are not caused by changes to the DNA sequence. This happens through mechanisms such as methylation and acetylation of histones, DNA methylation, and non-coding RNAs (ncRNA). Altering the acetylation of histones creates changes in expression by changing the binding affinity of histones to DNA, making it harder or easier for transcription machinery to access the DNA. Methyl and acetyl groups can also act as binding sites for transcription factors and enzymes that further modify histones or alter the DNA itself.
Patricia Gail Spear is an American virologist. She is a professor emeritus of microbiology and immunology at Northwestern University in Evanston, Illinois. She is best known for her pioneering work studying the herpes simplex virus. Spear is a past president of the American Society for Virology and an elected member of the National Academy of Sciences.












