An iodide ion is the ion I−. Compounds with iodine in formal oxidation state −1 are called iodides. This page is for the iodide ion and its salts, not organoiodine compounds. In everyday life, iodide is most commonly encountered as a component of iodized salt, which many governments mandate. Worldwide, iodine deficiency affects two billion people and is the leading preventable cause of intellectual disability.

Potassium tetraiodomercurate(II) is an inorganic compound consisting of potassium cations and the tetraiodomercurate(II) anion. It is mainly used as Nessler's reagent, a 0.09 mol/L solution of potassium tetraiodomercurate(II) (K2[HgI4]) in 2.5 mol/L potassium hydroxide, used to detect ammonia.
Classical qualitative inorganic analysis is a method of analytical chemistry which seeks to find the elemental composition of inorganic compounds. It is mainly focused on detecting ions in an aqueous solution, therefore materials in other forms may need to be brought to this state before using standard methods. The solution is then treated with various reagents to test for reactions characteristic of certain ions, which may cause color change, precipitation and other visible changes.

Mercury(I) chloride is the chemical compound with the formula Hg2Cl2. Also known as the mineral calomel (a rare mineral) or mercurous chloride, this dense white or yellowish-white, odorless solid is the principal example of a mercury(I) compound. It is a component of reference electrodes in electrochemistry.
Mercury sulfide, mercuric sulfide, mercury sulphide, or mercury(II) sulfide is a chemical compound composed of the chemical elements mercury and sulfur. It is represented by the chemical formula HgS. It is virtually insoluble in water.
The Old Nassau reaction or Halloween reaction is a chemical clock reaction in which a clear solution turns orange and then black. This reaction was discovered by two undergraduate students at Princeton University researching the inhibition of the iodine clock reaction (or Landolt reaction) by Hg2+, resulting in the formation of orange HgI2. Orange and black are the school colors of Princeton University, and "Old Nassau" is a nickname for Princeton, named for its historic administration building, Nassau Hall.

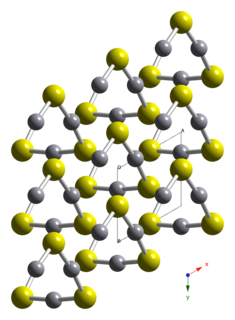
Mercury(II) iodide is a chemical compound with the molecular formula HgI2. It is typically produced synthetically but can also be found in nature as the extremely rare mineral coccinite. Unlike the related mercury(II) chloride it is hardly soluble in water (<100 ppm).

Mercury(II) thiocyanate (Hg(SCN)2) is an inorganic chemical compound, the coordination complex of Hg2+ and the thiocyanate anion. It is a white powder. It will produce a large, winding “snake” when ignited, an effect known as the Pharaoh's serpent.
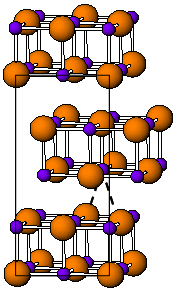
Thallium(I) iodide is a chemical compound with the formula TlI. It is unusual in being one of the few water-insoluble metal iodides, along with AgI, CuI, SnI2, SnI4, PbI2 and HgI2.
Mercury(I) sulfate, commonly called mercurous sulphate (UK) or mercurous sulfate (US) is the chemical compound Hg2SO4. Mercury(I) sulfate is a metallic compound that is white, pale yellow or beige powder. It is a metallic salt of sulfuric acid formed by replacing both hydrogen atoms with mercury(I). It is highly toxic; it could be fatal if inhaled, ingested, or absorbed by skin.
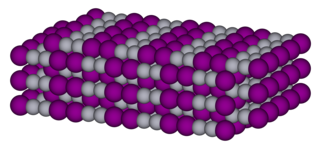
Mercury(I) iodide is a chemical compound of mercury and iodine. The chemical formula is Hg2I2. It is photosensitive and decomposes easily to mercury and HgI2.

Mercury(I) bromide or mercurous bromide is the chemical compound composed of mercury and bromine with the formula Hg2Br2. It changes color from white to yellow when heated and fluoresces a salmon color when exposed to ultraviolet light. It has applications in acousto-optical devices.

Mercury(I) fluoride or mercurous fluoride is the chemical compound composed of mercury and fluorine with the formula Hg2F2. It consists of small yellow cubic crystals, which turn black when exposed to light.

Mercury is a chemical element with symbol Hg and atomic number 80. It is commonly known as quicksilver and was formerly named hydrargyrum. A heavy, silvery d-block element, mercury is the only metallic element that is liquid at standard conditions for temperature and pressure; the only other element that is liquid under these conditions is the halogen bromine, though metals such as caesium, gallium, and rubidium melt just above room temperature.
Mercury(I) nitrate is a chemical compound with the formula Hg2(NO3)2. It is used in the preparation of other mercury(I) compounds, and is toxic.

Coccinite is a rare mercury iodide mineral with chemical formula of HgI2, mercury(II) iodide.
in addition to its discovery locality of Casas Viejas, Mexico, it has also been reported from Broken Hill, New South Wales, and from a uranium mine in Thuringia and old mercury workings in the Rhineland-Palatinate in Germany. At the Thuringia deposit the mineral occurs as a sublimation product resulting from fires associated with pyrite bearing, graptolitic slate.
Mercury(II) hydroxide or mercury dihydroxide is an inorganic metal hydroxide with the chemical formula HgH2O2 or more commonly written as Hg(OH)2.
Mayer's reagent is an alkaloidal precipitating reagent used for the detection of alkaloids in natural products. Mayer’s reagent is freshly prepared by dissolving a mixture of mercuric chloride and of potassium iodide in water. Most alkaloids are precipitated from neutral or slightly acidic solution by Mayer’s reagent to give a cream coloured precipitate. This test was invented by the German Chemist, Julius Robert Von Mayer (1814–1878).

Mercury(II) hydride is an inorganic compound with the chemical formula HgH
2. It is both thermodynamically and kinetically unstable at ambient temperature, and as such, little is known about its bulk properties. However, it known as a white, crystalline solid, which is kinetically stable at temperatures below −125 °C (−193 °F), which was synthesised for the first time in 1951.











