Related Research Articles

The smallpox vaccine is the first vaccine to have been developed against a contagious disease. In 1796, British physician Edward Jenner demonstrated that an infection with the relatively mild cowpox virus conferred immunity against the deadly smallpox virus. Cowpox served as a natural vaccine until the modern smallpox vaccine emerged in the 20th century. From 1958 to 1977, the World Health Organization (WHO) conducted a global vaccination campaign that eradicated smallpox, making it the only human disease to be eradicated. Although routine smallpox vaccination is no longer performed on the general public, the vaccine is still being produced to guard against bioterrorism, biological warfare, and mpox.
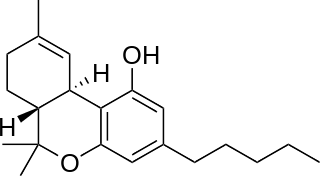
Nabiximols is a specific Cannabis extract that was approved in 2010 as a botanical drug in the United Kingdom. Nabiximols is sold as a mouth spray intended to alleviate neuropathic pain, spasticity, overactive bladder, and other symptoms of multiple sclerosis; it was developed by the UK company GW Pharmaceuticals. In 2019, it was proposed that following application of the spray, nabiximols is washed away from the oral mucosa by the saliva flow and ingested into the stomach, with subsequent absorption from the gastro-intestinal tract. Nabiximols is a combination drug standardized in composition, formulation, and dose. Its principal active components are the cannabinoids: tetrahydrocannabinol (THC) and cannabidiol (CBD). Each spray delivers a dose of 2.7 mg THC and 2.5 mg CBD.
Panitumumab, sold under the brand name Vectibix, is a fully human monoclonal antibody specific to the epidermal growth factor receptor.

Lubiprostone, sold under the brand name Amitiza among others, is a medication used in the management of chronic idiopathic constipation, predominantly irritable bowel syndrome-associated constipation in women and opioid-induced constipation. The drug is owned by Mallinckrodt and is marketed by Takeda Pharmaceutical Company.
Northwest Biotherapeutics is a development-stage American pharmaceutical company headquartered in Maryland that focuses on developing immunotherapies against different types of cancer. It was founded in 1996 by Alton L. Boynton.
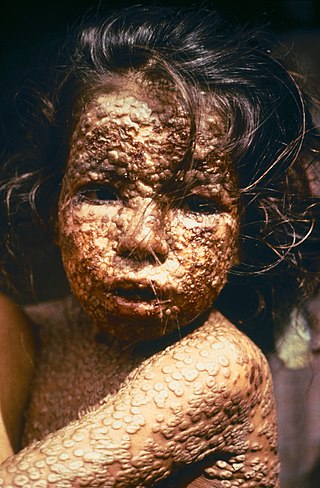
Smallpox was an infectious disease caused by variola virus which belongs to the genus Orthopoxvirus. The last naturally occurring case was diagnosed in October 1977, and the World Health Organization (WHO) certified the global eradication of the disease in 1980, making smallpox the only human disease to be eradicated.

Three Kingdoms is a 2010 Chinese television series based on the events in the late Eastern Han dynasty and the Three Kingdoms period. The plot is adapted from the 14th century historical novel Romance of the Three Kingdoms and other stories about the Three Kingdoms period. Directed by Gao Xixi, the series had a budget of over 160 million RMB and took five years of pre-production work. Shooting of the series commenced in October 2008, and it was released in China in May 2010.
Tanezumab is a monoclonal antibody against nerve growth factor as a treatment for pain via a novel mechanisms different from conventional pain-killer drugs. Tanezumab was discovered and developed by Rinat Neuroscience and was acquired by Pfizer in 2006.

Progressive vaccinia is a rare cutaneous condition caused by the vaccinia virus, characterized by painless but progressive necrosis and ulceration.
Elemenes are a group of closely related natural chemical compounds found in a variety of plants. The elemenes, which include α-, β-, γ-, and δ-elemene, are structural isomers of each other and are classified as sesquiterpenes. The elemenes contribute to the floral aromas of some plants, and are used as pheromones by some insects.
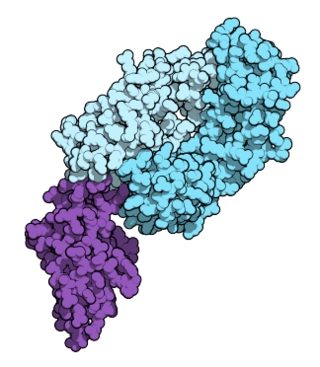
Nivolumab, sold under the brand name Opdivo, is a medication used to treat a number of types of cancer. This includes melanoma, lung cancer, malignant pleural mesothelioma, renal cell carcinoma, Hodgkin lymphoma, head and neck cancer, urothelial carcinoma, colon cancer, esophageal squamous cell carcinoma, liver cancer, gastric cancer, and esophageal or gastroesophageal junction (GEJ) cancer. It is used by slow injection into a vein.

Jennerex Biotherapeutics, Inc. was an American private biopharmaceutical company that developed the oncolytic viruses JX-594 and JX-929 among others. By creating oncolytic viruses that can (1) kill tumor cells directly through lysis, (2) activate the immune system by delivering genes that encode immunostimulants and by overcoming tumor cell-induced immunological tolerance, and (3) reduce tumor nutrient supply through the destruction of blood vessels, Jennerex aimed to create a novel approach to treating and possibly curing cancer.
Vaccinia immune globulin (VIG) is made from the pooled blood of individuals who have been inoculated with the smallpox vaccine. The antibodies these individuals developed in response to the smallpox vaccine are removed and purified. This results in VIG. It can be administered intravenously. It is used to treat individuals who have developed progressive vaccinia after smallpox vaccination.

Setmelanotide, sold under the brand name Imcivree, is a medication used for the treatment of genetic obesity caused by a rare single-gene mutation.

ZF2001, trade-named Zifivax or ZF-UZ-VAC-2001, is an adjuvanted protein subunit COVID-19 vaccine developed by Anhui Zhifei Longcom in collaboration with the Institute of Microbiology at the Chinese Academy of Sciences. The vaccine candidate is in Phase III trials with 29,000 participants in China, Ecuador, Malaysia, Pakistan, and Uzbekistan.
Conbercept, sold under the commercial name Lumitin, is a novel vascular endothelial growth factor (VEGF) inhibitor used to treat neovascular age-related macular degeneration (AMD) and diabetic macular edema (DME). The anti-VEGF was approved for the treatment of neovascular AMD by the China State FDA (CFDA) in December 2013. As of December 2020, conbercept is undergoing phase III clinical trials through the U.S. Food and Drug Administration’s PANDA-1 and PANDA-2 development programs.

Soberana 02 or Soberana 2, technical name FINLAY-FR-2, is a COVID-19 vaccine produced by the Finlay Institute, a Cuban epidemiological research institute. The vaccine is known as PastoCovac in Iran, where it has been developed in collaboration with the Pasteur Institute of Iran.

SCB-2019 is a protein subunit COVID-19 vaccine developed by Clover Biopharmaceuticals using an adjuvant from Dynavax technologies. Positive results of Phase I trials for the vaccine were published in The Lancet and the vaccine completed enrollment of 29,000 participants in Phase II/III trials in July 2021. In September 2021, SCB-2019 announced Phase III results showing 67% efficacy against all cases of COVID-19 and 79% efficacy against all cases of the Delta variant. Additionally, the vaccine was 84% effective against moderate cases and 100% effective against hospitalization.

A viral vector vaccine is a vaccine that uses a viral vector to deliver genetic material (DNA) that can be transcribed by the recipient's host cells as mRNA coding for a desired protein, or antigen, to elicit an immune response. As of April 2021, six viral vector vaccines, four COVID-19 vaccines and two Ebola vaccines, have been authorized for use in humans.

AWcorna, originally termed ARCoV and also known as the Walvax COVID-19 vaccine, is an mRNA COVID-19 vaccine developed by Walvax Biotechnology, Suzhou Abogen Biosciences, and the PLA Academy of Military Science. In contrast to other mRNA COVID vaccines, such as those by Pfizer-BioNtech and Moderna, this vaccine primarily targets the Sars-CoV-2 receptor-binding domain of the spike protein, rather than the entire spike protein. It is approved for Phase III trials in China, Mexico, Indonesia, and Nepal.
References
- 1 2 Name, Drug (2 October 2021). "Analgecine - Vanway Pharmaceutical". AdisInsight.
29 Sep 2021 Discontinued - Phase-III for Neuropathic pain in China (Infusion)
- ↑ Dong, Jian; Tu, Hung-Ping; Ding, Wen-Yuan; Ding, Yue; Li, Jin; Zhang, Xue-Li; Fan, Cun-Yi; Dong, Hai-Long; Wu, Yi-Chia; Wang, Sebrina Su-Jung; Lin, Chen-Lung Steve (2016). "Analgecine, the extracts of Vaccinia-inoculated rabbit skin, effectively alleviates the chronic low back pain with little side effect – A randomized multi-center double-blind placebo-controlled phase 3 clinical trial". Contemporary Clinical Trials Communications. 2: 16–24. doi: 10.1016/j.conctc.2015.11.002 . ISSN 2451-8654. PMC 5935851 . PMID 29736443.