| |
 | |
| Clinical data | |
|---|---|
| Dependence liability | None |
| Routes of administration | Oral |
| ATC code |
|
| Legal status | |
| Legal status |
|
| Pharmacokinetic data | |
| Metabolism | Hepatic and CYP3A & CYP2B |
| Identifiers | |
| |
| CAS Number | |
| PubChem CID | |
| IUPHAR/BPS | |
| DrugBank | |
| ChemSpider | |
| UNII | |
| KEGG | |
| ChEBI | |
| CompTox Dashboard (EPA) | |
| ECHA InfoCard | 100.112.565 |
| Chemical and physical data | |
| Formula | C35H52O4 |
| Molar mass | 536.797 g·mol−1 |
| 3D model (JSmol) | |
| Melting point | 79–80 °C (174–176 °F) |
| Solubility in water | 0.66 mg/mL (20 °C) |
| |
| |
| | |
Hyperforin is a phytochemical produced by some of the members of the plant genus Hypericum , notably Hypericum perforatum (St John's wort). [2] Hyperforin may be involved in the pharmacological effects of St. John's wort, [2] specifically in its antidepressant effects. [3] [4] [5] Meta-analyses of clinical trials suggest that H. perforatum is as effective as SSRIs for treating mild to moderate depression and is better tolerated, although findings are limited by short study durations.
Contents
- Occurrence
- Chemistry
- Pharmacokinetics
- Pharmacodynamics
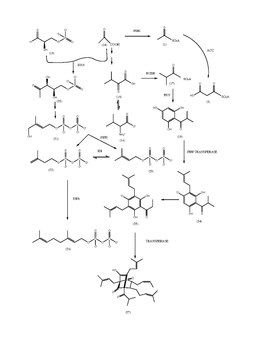
- Biosynthesis
- Antidepressant research
- See also
- References
- External links
Hyperforin is found in significant amounts only in H. perforatum, where it accumulates as a probable plant defense compound, with modern carbon dioxide extraction methods used to isolate it from mixtures containing related compounds like adhyperforin.