Related Research Articles

A ceramic is any of the various hard, brittle, heat-resistant, and corrosion-resistant materials made by shaping and then firing an inorganic, nonmetallic material, such as clay, at a high temperature. Common examples are earthenware, porcelain, and brick.

Piezoelectricity is the electric charge that accumulates in certain solid materials—such as crystals, certain ceramics, and biological matter such as bone, DNA, and various proteins—in response to applied mechanical stress. The word piezoelectricity means electricity resulting from pressure and latent heat. It is derived from Ancient Greek πιέζω (piézō) 'to squeeze or press', and ἤλεκτρον (ḗlektron) 'amber'. The German form of the word (Piezoelektricität) was coined in 1881 by the German physicist Wilhelm Gottlieb Hankel; the English word was coined in 1883.

Zirconium dioxide, sometimes known as zirconia, is a white crystalline oxide of zirconium. Its most naturally occurring form, with a monoclinic crystalline structure, is the mineral baddeleyite. A dopant stabilized cubic structured zirconia, cubic zirconia, is synthesized in various colours for use as a gemstone and a diamond simulant.

Strontium titanate is an oxide of strontium and titanium with the chemical formula SrTiO3. At room temperature, it is a centrosymmetric paraelectric material with a perovskite structure. At low temperatures it approaches a ferroelectric phase transition with a very large dielectric constant ~104 but remains paraelectric down to the lowest temperatures measured as a result of quantum fluctuations, making it a quantum paraelectric. It was long thought to be a wholly artificial material, until 1982 when its natural counterpart—discovered in Siberia and named tausonite—was recognised by the IMA. Tausonite remains an extremely rare mineral in nature, occurring as very tiny crystals. Its most important application has been in its synthesized form wherein it is occasionally encountered as a diamond simulant, in precision optics, in varistors, and in advanced ceramics.
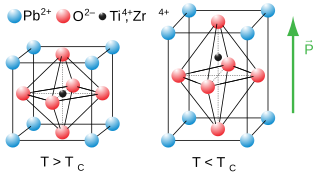
Lead zirconate titanate, also called lead zirconium titanate and commonly abbreviated as PZT, is an inorganic compound with the chemical formula Pb[ZrxTi1−x]O3(0 ≤ x ≤ 1). It is a ceramic perovskite material that shows a marked piezoelectric effect, meaning that the compound changes shape when an electric field is applied. It is used in a number of practical applications such as ultrasonic transducers and piezoelectric resonators. It is a white to off-white solid.
In electromagnetism, electrostriction is a property of all electrical non-conductors, or dielectrics, that causes them to change their shape under the application of an electric field. It is the dual property to magnetostriction.

A ferrite is a ceramic material made by mixing and firing iron(III) oxide with one or more additional metallic elements, such as strontium, barium, manganese, nickel, and zinc. They are ferrimagnetic, meaning they are attracted by magnetic fields and can be magnetized to become permanent magnets. Unlike other ferromagnetic materials, most ferrites are not electrically conductive, making them useful in applications like magnetic cores for transformers to suppress eddy currents. Ferrites can be divided into two families based on their resistance to being demagnetized.

Barium titanate (BTO) is an inorganic compound with chemical formula BaTiO3. Barium titanate appears white as a powder and is transparent when prepared as large crystals. It is a ferroelectric, pyroelectric, and piezoelectric ceramic material that exhibits the photorefractive effect. It is used in capacitors, electromechanical transducers and nonlinear optics.

A ceramic capacitor is a fixed-value capacitor where the ceramic material acts as the dielectric. It is constructed of two or more alternating layers of ceramic and a metal layer acting as the electrodes. The composition of the ceramic material defines the electrical behavior and therefore applications. Ceramic capacitors are divided into two application classes:
Lead scandium tantalate (PST) is a mixed oxide of lead, scandium, and tantalum. It has the formula Pb(Sc0.5Ta0.5)O3. It is a ceramic material with a perovskite structure, where the Sc and Ta atoms at the B site have an arrangement that is intermediate between ordered and disordered configurations, and can be fine-tuned with thermal treatment. It is ferroelectric at temperatures below 270 K (−3 °C; 26 °F), and is also piezoelectric. Like structurally similar lead zirconate titanate and barium strontium titanate, PST can be used for manufacture of uncooled focal plane array infrared imaging sensors for thermal cameras.
Lanthanum strontium cobalt ferrite (LSCF), also called lanthanum strontium cobaltite ferrite is a specific ceramic oxide derived from lanthanum cobaltite of the ferrite group. It is a phase containing lanthanum(III) oxide, strontium oxide, cobalt oxide and iron oxide with the formula La
xSr
1-xCo
yFe
1-yO
3, where 0.1≤x≤0.4 and 0.2≤y≤0.8.
The Burns temperature, Td, is the temperature where a ferroelectric material, previously in paraelectric state, starts to present randomly polarized nanoregions, that are polar precursor clusters. This behaviour is typical of several, but not all, ferroelectric materials, and was observed in lead titanate (PbTiO3), potassium niobate (KNbO3), lead lanthanum zirconate titanate (PLZT), lead magnesium niobate (PMN), lead zinc niobate (PZN), K2Sr4(NbO3)10, and strontium barium niobate (SBN), Na1/2Bi1/2O3 (NBT).

Abnormal or discontinuous grain growth, also referred to as exaggerated or secondary recrystallisation grain growth, is a grain growth phenomenon in which certain energetically favorable grains (crystallites) grow rapidly in a matrix of finer grains, resulting in a bimodal grain-size distribution.
Relaxor ferroelectrics are ferroelectric materials that exhibit high electrostriction. As of 2015, although they have been studied for over fifty years, the mechanism for this effect is still not completely understood, and is the subject of continuing research.
A complex oxide is a chemical compound that contains oxygen and at least two other elements. Complex oxide materials are notable for their wide range of magnetic and electronic properties, such as ferromagnetism, ferroelectricity, and high-temperature superconductivity. These properties often come from their strongly correlated electrons in d or f orbitals.

Kenji Uchino is an American electronics engineer, physicist, academic, inventor and industry executive. He is currently a professor of Electrical Engineering at Pennsylvania State University, where he also directs the International Center for Actuators and Transducers at Materials Research Institute. He is the former associate director at The US Office of Naval Research – Global Tokyo Office.
A process related to the sol-gel route is the Pechini, or liquid mix, process. An aqueous solution of suitable oxides or salts is mixed with an alpha-hydroxycarboxylic acid such as citric acid. Chelation, or the formation of complex ring-shaped compounds around the metal cations, takes place in the solution. A polyhydroxy alcohol is then added, and the liquid is heated to 150–250 °C (300–480 °F) to allow the chelates to polymerize, or form large, cross-linked networks. As excess water is removed by heating, a solid polymeric resin results. Eventually, at still higher temperatures of 500–900 °C (930–1,650 °F), the resin is decomposed or charred, and ultimately a mixed oxide is obtained. Particle size is extremely small, typically 20 to 50 nanometres, with intimate mixing taking place on the atomic scale.
Nickel niobate is a complex oxide which as a solid material has found potential applications in catalysis and lithium batteries.