Related Research Articles

Antimony is a chemical element with the symbol Sb (from Latin stibium) and atomic number 51. A lustrous gray metalloid, it is found in nature mainly as the sulfide mineral stibnite (Sb2S3). Antimony compounds have been known since ancient times and were powdered for use as medicine and cosmetics, often known by the Arabic name kohl. The earliest known description of the metalloid in the West was written in 1540 by Vannoccio Biringuccio.

Astatine is a chemical element with the symbol At and atomic number 85. It is the rarest naturally occurring element in the Earth's crust, occurring only as the decay product of various heavier elements. All of astatine's isotopes are short-lived; the most stable is astatine-210, with a half-life of 8.1 hours. A sample of the pure element has never been assembled, because any macroscopic specimen would be immediately vaporized by the heat of its radioactivity.

Lead is a chemical element with the symbol Pb and atomic number 82. It is a heavy metal that is denser than most common materials. Lead is soft and malleable, and also has a relatively low melting point. When freshly cut, lead is a shiny gray with a hint of blue. It tarnishes to a dull gray color when exposed to air. Lead has the highest atomic number of any stable element and three of its isotopes are endpoints of major nuclear decay chains of heavier elements. Lead is toxic, even in small amounts, especially to children.

Tin is a chemical element with the symbol Sn and atomic number 50. A silvery-coloured metal, tin is soft enough to be cut with little force, and a bar of tin can be bent by hand with little effort. When bent, the so-called "tin cry" can be heard as a result of twinning in tin crystals; this trait is shared by indium, cadmium, zinc, and mercury in its solid state.

Fireworks are a class of low explosive pyrotechnic devices used for aesthetic and entertainment purposes. They are most commonly used in fireworks displays, combining a large number of devices in an outdoor setting. Such displays are the focal point of many cultural and religious celebrations.
A period 6 element is one of the chemical elements in the sixth row (or period) of the periodic table of the chemical elements, including the lanthanides. The periodic table is laid out in rows to illustrate recurring (periodic) trends in the chemical behaviour of the elements as their atomic number increases: a new row is begun when chemical behaviour begins to repeat, meaning that elements with similar behaviour fall into the same vertical columns. The sixth period contains 32 elements, tied for the most with period 7, beginning with caesium and ending with radon. Lead is currently the last stable element; all subsequent elements are radioactive. For bismuth, however, its only primordial isotope, 209Bi, has a half-life of more than 1019 years, over a billion times longer than the current age of the universe. As a rule, period 6 elements fill their 6s shells first, then their 4f, 5d, and 6p shells, in that order; however, there are exceptions, such as gold.

Pyrotechnics is the science and craft of creating such things as fireworks, safety matches, oxygen candles, explosive bolts and other fasteners, parts of automotive airbags, as well as gas-pressure blasting in mining, quarrying, and demolition. This trade relies upon self-contained and self-sustained exothermic chemical reactions to make heat, light, gas, smoke and/or sound. The name comes from the Greek words pyr ("fire") and tekhnikos.

A pnictogen is any of the chemical elements in group 15 of the periodic table. Group (V) is also known as the nitrogen group or nitrogen family. Group (V) consists of the elements nitrogen (N), phosphorus (P), arsenic (As), antimony (Sb), bismuth (Bi), and moscovium (Mc).

A firecracker is a small explosive device primarily designed to produce a large amount of noise, especially in the form of a loud bang, usually for celebration or entertainment; any visual effect is incidental to this goal. They have fuses, and are wrapped in a heavy paper casing to contain the explosive compound. Firecrackers, along with fireworks, originated in China.
The inert-pair effect is the tendency of the two electrons in the outermost atomic s-orbital to remain unshared in compounds of post-transition metals. The term inert-pair effect is often used in relation to the increasing stability of oxidation states that are two less than the group valency for the heavier elements of groups 13, 14, 15 and 16. The term "inert pair" was first proposed by Nevil Sidgwick in 1927. The name suggests that the outermost s electron pairs are more tightly bound to the nucleus in these atoms, and therefore more difficult to ionize or share.
Bismuth subsalicylate, sold generically as pink bismuth and under the brand names Pepto-Bismol and BisBacter, is an antacid medication used to treat temporary discomforts of the stomach and gastrointestinal tract, such as nausea, heartburn, indigestion, upset stomach, and diarrhea.

The oligodynamic effect is a biocidal effect of metals, especially heavy metals, that occurs even in low concentrations.

Bismuth(III) oxide is perhaps the most industrially important compound of bismuth. It is also a common starting point for bismuth chemistry. It is found naturally as the mineral bismite (monoclinic) and sphaerobismoite, but it is usually obtained as a by-product of the smelting of copper and lead ores. Dibismuth trioxide is commonly used to produce the "Dragon's eggs" effect in fireworks, as a replacement of red lead.
A pyrotechnic colorant is a chemical compound which causes a flame to burn with a particular color. These are used to create the colors in pyrotechnic compositions like fireworks and colored fires. The color-producing species are usually created from other chemicals during the reaction. Metal salts are commonly used; elemental metals are used rarely.

Bismuth subcarbonate (BiO)2CO3, sometimes written Bi2O2(CO3) is a chemical compound of bismuth containing both oxide and carbonate anions. Bismuth is in the +3 oxidation state. Bismuth subcarbonate occurs naturally as the mineral bismutite. Its structure consists of Bi–O layers and CO3 layers and is related to kettnerite, CaBi(CO3)OF. It is light-sensitive.

Bismuth is a chemical element with the symbol Bi and atomic number 83. It is a post-transition metal and one of the pnictogens, with chemical properties resembling its lighter group 15 siblings arsenic and antimony. Elemental bismuth occurs naturally, and its sulfide and oxide forms are important commercial ores. The free element is 86% as dense as lead. It is a brittle metal with a silvery-white color when freshly produced. Surface oxidation generally gives samples of the metal a somewhat rosy cast. Further oxidation under heat can give bismuth a vividly iridescent appearance due to thin-film interference. Bismuth is both the most diamagnetic element and one of the least thermally conductive metals known.

Antimony sulfate, Sb2(SO4)3, is a hygroscopic salt formed by reacting antimony or its compounds with hot sulfuric acid. It is used in doping of semiconductors and in the production of explosives and fireworks.

The metallic elements in the periodic table located between the transition metals to their left and the chemically weak nonmetallic metalloids to their right have received many names in the literature, such as post-transition metals, poor metals, other metals, p-block metals and chemically weak metals. The most common name, post-transition metals, is generally used in this article.

Heavy metals are generally defined as metals with relatively high densities, atomic weights, or atomic numbers. The criteria used, and whether metalloids are included, vary depending on the author and context. In metallurgy, for example, a heavy metal may be defined on the basis of density, whereas in physics the distinguishing criterion might be atomic number, while a chemist would likely be more concerned with chemical behaviour. More specific definitions have been published, none of which have been widely accepted. The definitions surveyed in this article encompass up to 96 out of the 118 known chemical elements; only mercury, lead and bismuth meet all of them. Despite this lack of agreement, the term is widely used in science. A density of more than 5 g/cm3 is sometimes quoted as a commonly used criterion and is used in the body of this article.

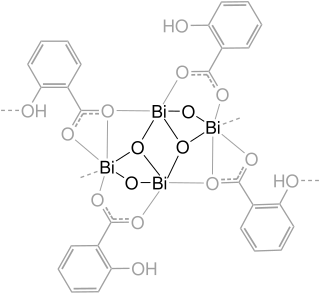
Bismuth compounds are compounds containing the element bismuth (Bi). Bismuth forms trivalent and pentavalent compounds, the trivalent ones being more common. Many of its chemical properties are similar to those of arsenic and antimony, although they are less toxic than derivatives of those lighter elements.
References
- ↑ Linda D. Hall (2006). Brands and Their Companies: Consumer Products and Their Manufacturers with Addresses and Phone Numbers. Thomson Gale. ISBN 978-0-7876-8951-3.
- ↑ "Firework Effect - Dragon Eggs" (video). youtube.com. October 20, 2000.
- ↑ Chris Mocella; John A. Conkling (15 January 2019). Chemistry of Pyrotechnics: Basic Principles and Theory, Third Edition. CRC Press. pp. 51–. ISBN 978-1-351-62655-2.