
In chemistry, a ketone is a functional group with the structure RC(=O)R', where R and R' can be a variety of carbon-containing substituents. Ketones contain a carbonyl group (a carbon-oxygen double bond). The simplest ketone is acetone (R = R' = methyl), with the formula CH3C(O)CH3. Many ketones are of great importance in industry and in biology. Examples include many sugars (ketoses), many steroids (e.g., testosterone), and the solvent acetone.

A ketene is an organic compound of the form R′R″C=C=O, where R and R' are two arbitrary monovalent chemical groups. The name may also refer to the specific compound ethenone H
2C=C=O, the simplest ketene.
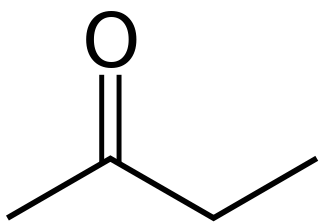
Butanone, also known as methyl ethyl ketone (MEK), is an organic compound with the formula CH3C(O)CH2CH3. This colorless liquid ketone has a sharp, sweet odor reminiscent of butterscotch and acetone It is produced industrially on a large scale, but occurs in nature only in trace amounts. It is partially soluble in water, and is commonly used as an industrial solvent. It is an isomer of another solvent, tetrahydrofuran.
The cumene process is an industrial process for synthesizing phenol and acetone from benzene and propylene. The term stems from cumene, the intermediate material during the process. It was invented by R. Ūdris and P. Sergeyev in 1942 (USSR)., and independently by Heinrich Hock in 1944

An oxime is a chemical compound belonging to the imines, with the general formula RR'C=NOH, where R is an organic side-chain and R' may be hydrogen, forming an aldoxime, or another organic group, forming a ketoxime. O-substituted oximes form a closely related family of compounds. Amidoximes are oximes of amides with general structure R1C(=NOH)NR2R3.
Mesitylene or 1,3,5-trimethylbenzene is a derivative of benzene with three methyl substituents positioned symmetrically around the ring. The other two isomeric trimethylbenzenes are 1,2,4-trimethylbenzene (pseudocumene) and 1,2,3-trimethylbenzene (hemimellitene). All three compounds have the formula C6H3(CH3)3, which is commonly abbreviated C6H3Me3. Mesitylene is a colourless liquid with sweet aromatic odor. It is a component of coal tar, which is its traditional source. It is a precursor to diverse fine chemicals. The mesityl group (Mes) is a substituent with the formula C6H2Me3 and is found in various other compounds.

A permanent marker or indelible marker is a type of marker pen that is used to create permanent or semi-permanent writing on an object. In general, the ink comprises a main carrier solvent, a glyceride, a pyrrolidone, a resin and a colorant, making it water resistant. It is capable of writing on a variety of surfaces from paper to metal to stone. They come in a variety of tip sizes, shapes, and colors. Like spray paint, these markers contain volatile organic compounds which evaporate to dry the ink. Due to compounds such as toluene and xylene often being present in permanent markers, they have a potential for abuse as a recreational drug.

A cyanohydrin is a functional group found in organic compounds in which a cyano and a hydroxy group are attached to the same carbon atom. The general formula is R2C(OH)CN, where R is H, alkyl, or aryl. Cyanohydrins are industrially important precursors to carboxylic acids and some amino acids. Cyanohydrins can be formed by the cyanohydrin reaction, which involves treating a ketone or an aldehyde with hydrogen cyanide (HCN) in the presence of excess amounts of sodium cyanide (NaCN) as a catalyst:

Acetone peroxide is an organic peroxide and a primary high explosive. It is produced by the reaction of acetone and hydrogen peroxide to yield a mixture of linear monomer and cyclic dimer, trimer, and tetramer forms. The trimer is known as triacetone triperoxide (TATP) or tri-cyclic acetone peroxide (TCAP). The dimer is known as diacetone diperoxide (DADP). Acetone peroxide takes the form of a white crystalline powder with a distinctive bleach-like odor or a fruit-like smell when pure, and can explode powerfully if subjected to heat, friction, static electricity, concentrated sulfuric acid, strong UV radiation or shock. Until about 2015 explosives detectors were not set to detect non-nitrogenous explosives as most explosives used were nitrogen-based. Nitrogen-free TATP has been used as the explosive of choice in several terrorist bomb attacks since 2001.
Acetamide (systematic name: ethanamide) is an organic compound with the formula CH3CONH2. It is the simplest amide derived from acetic acid. It finds some use as a plasticizer and as an industrial solvent. The related compound N,N-dimethylacetamide (DMA) is more widely used, but it is not prepared from acetamide. Acetamide can be considered an intermediate between acetone, which has two methyl (CH3) groups either side of the carbonyl (CO), and urea which has two amide (NH2) groups in those locations.
In chemistry, radical initiators are substances that can produce radical species under mild conditions and promote radical reactions. These substances generally possess weak bonds—bonds that have small bond dissociation energies. Radical initiators are utilized in industrial processes such as polymer synthesis. Typical examples are halogen molecules, azo compounds, and organic and inorganic peroxides.

Acetone, or propanone, is an organic compound with the formula (CH3)2CO. It is the simplest and smallest ketone. It is a colourless, highly volatile and flammable liquid with a characteristic pungent odour.

Bromoacetone is an organic compound with the formula CH3COCH2Br. This colorless liquid is a lachrymatory agent and a precursor to other organic compounds.
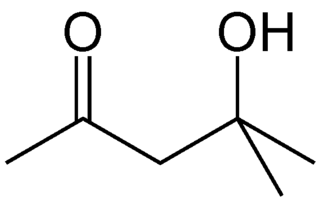
Diacetone alcohol is an organic compound with the formula CH3C(O)CH2C(OH)(CH3)2, sometimes called DAA. This colorless liquid is a common synthetic intermediate used for the preparation of other compounds, and is also used as a solvent.
Acetone cyanohydrin (ACH) is an organic compound used in the production of methyl methacrylate, the monomer of the transparent plastic polymethyl methacrylate (PMMA), also known as acrylic. It liberates hydrogen cyanide easily, so it is used as a source of such. For this reason, this cyanohydrin is also highly toxic.

Dimethyldioxirane (DMDO), also referred to as Murray's reagent in reference to Robert W. Murray, is a dioxirane derived from acetone and can be considered as a monomer of acetone peroxide. It is a powerful yet selective oxidizing agent which finds use in organic synthesis. It is known only in the form of a dilute solution, usually in acetone, and hence the properties of the pure material are largely unknown.
Isopropyl alcohol (IUPAC name propan-2-ol; commonly called isopropanol or 2-propanol) is a colorless, flammable chemical compound (chemical formula CH3CHOHCH3) with a strong odor. As an isopropyl group linked to a hydroxyl group, it is the simplest example of a secondary alcohol, where the alcohol carbon atom is attached to two other carbon atoms. It is a structural isomer of 1-propanol and ethyl methyl ether.
In industrial paper-making processes, organosolv is a pulping technique that uses an organic solvent to solubilise lignin and hemicellulose. It has been considered in the context of both pulp and paper manufacture and biorefining for subsequent conversion of cellulose to fuel ethanol. The process was invented by Theodor Kleinert in 1968 as an environmentally benign alternative to kraft pulping.
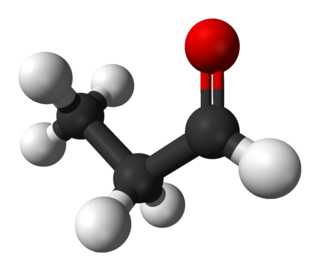
Propionaldehyde or propanal is the organic compound with the formula CH3CH2CHO. It is a saturated 3-carbon aldehyde and is a structural isomer of acetone. It is a colourless, flammable liquid with a slightly irritating, fruity odour.

Acetone oxime (acetoxime) is the organic compound with the formula (CH3)2CNOH. It is the simplest example of a ketoxime. It is a white crystalline solid that is soluble in water, ethanol, ether, chloroform, and ligroin. It is used as a reagent in organic synthesis.












