Acid test is a qualitative chemical or metallurgical assay which uses acid, or figuratively a definitive test for some attribute, e.g. of a person's character, or of the performance of a product.
Contents
Acid test or acid tests may also refer to:
Acid test is a qualitative chemical or metallurgical assay which uses acid, or figuratively a definitive test for some attribute, e.g. of a person's character, or of the performance of a product.
Acid test or acid tests may also refer to:

Limestone is a type of carbonate sedimentary rock which is the main source of the material lime. It is composed mostly of the minerals calcite and aragonite, which are different crystal forms of CaCO3. Limestone forms when these minerals precipitate out of water containing dissolved calcium. This can take place through both biological and nonbiological processes, though biological processes, such as the accumulation of corals and shells in the sea, have likely been more important for the last 540 million years. Limestone often contains fossils which provide scientists with information on ancient environments and on the evolution of life.

Marble is a metamorphic rock consisting of carbonate minerals that recrystallize under the influence of heat, pressure, and aqueous solutions (most commonly calcite (CaCO3) or dolomite (CaMg(CO3)2) and has a crystalline texture of varying thickness. Marble is typically not foliated (layered), although there are exceptions.
In chemistry, a salt is a chemical compound consisting of an ionic assembly of positively charged cations and negatively charged anions, which results in a compound with no net electric charge. A common example is table salt, with positively charged sodium ions and negatively charged chloride ions.

Aqua regia is a mixture of nitric acid and hydrochloric acid, optimally in a molar ratio of 1:3. Aqua regia is a fuming liquid. Freshly prepared aqua regia is colorless, but it turns yellow, orange or red within seconds from the formation of nitrosyl chloride and nitrogen dioxide. It was named by alchemists because it can dissolve the noble metals gold and platinum, though not all metals.
Benedict's reagent is a chemical reagent and complex mixture of sodium carbonate, sodium citrate, and copper(II) sulfate pentahydrate. It is often used in place of Fehling's solution to detect the presence of reducing sugars. The presence of other reducing substances also gives a positive result. Such tests that use this reagent are called the Benedict's tests. A positive test with Benedict's reagent is shown by a color change from clear blue to brick-red with a precipitate.

The lithology of a rock unit is a description of its physical characteristics visible at outcrop, in hand or core samples, or with low magnification microscopy. Physical characteristics include colour, texture, grain size, and composition. Lithology may refer to either a detailed description of these characteristics, or a summary of the gross physical character of a rock. Examples of lithologies in the second sense include sandstone, slate, basalt, or limestone.

Azurite is a soft, deep-blue copper mineral produced by weathering of copper ore deposits. During the early 19th century, it was also known as chessylite, after the type locality at Chessy-les-Mines near Lyon, France. The mineral, a basic carbonate with the chemical formula Cu3(CO3)2(OH)2, has been known since ancient times, and was mentioned in Pliny the Elder's Natural History under the Greek name kuanos (κυανός: "deep blue," root of English cyan) and the Latin name caeruleum. Copper (Cu2+) gives it its blue color.
The Leblanc process was an early industrial process for making soda ash used throughout the 19th century, named after its inventor, Nicolas Leblanc. It involved two stages: making sodium sulfate from sodium chloride, followed by reacting the sodium sulfate with coal and calcium carbonate to make sodium carbonate. The process gradually became obsolete after the development of the Solvay process.
An acid is any chemical compound that, when dissolved in water, gives a solution with a pH of less than 7.0.

Barium carbonate is the inorganic compound with the formula BaCO3. Like most alkaline earth metal carbonates, it is a white salt that is poorly soluble in water. It occurs as the mineral known as witherite. In a commercial sense, it is one of the most important barium compounds.

Dolomite (also known as dolomite rock, dolostone or dolomitic rock) is a sedimentary carbonate rock that contains a high percentage of the mineral dolomite, CaMg(CO3)2. It occurs widely, often in association with limestone and evaporites, though it is less abundant than limestone and rare in Cenozoic rock beds (beds less than about 66 million years in age). The first geologist to distinguish dolomite from limestone was Déodat Gratet de Dolomieu; a French mineralogist and geologist. He recognized and described the distinct characteristics of dolomite in the late 18th century, differentiating it from limestone. That's why the mineral was named after him.
Classical qualitative inorganic analysis is a method of analytical chemistry which seeks to find the elemental composition of inorganic compounds. It is mainly focused on detecting ions in an aqueous solution, therefore materials in other forms may need to be brought to this state before using standard methods. The solution is then treated with various reagents to test for reactions characteristic of certain ions, which may cause color change, precipitation and other visible changes.
HCL may refer to:

Calcareous is an adjective meaning "mostly or partly composed of calcium carbonate", in other words, containing lime or being chalky. The term is used in a wide variety of scientific disciplines.

Cleaning agents or hard-surface cleaners are substances used to remove dirt, including dust, stains, foul odors, and clutter on surfaces. Purposes of cleaning agents include health, beauty, removing offensive odor, and avoiding the spread of dirt and contaminants to oneself and others. Some cleaning agents can kill bacteria and clean at the same time. Others, called degreasers, contain organic solvents to help dissolve oils and fats.
An acid test is any qualitative chemical or metallurgical assay which uses acid; most commonly, and historically, the use of a strong acid to distinguish gold from base metals. Figuratively, acid test is any definitive test for some attribute, e.g. of a person's character, or of the performance of a product.
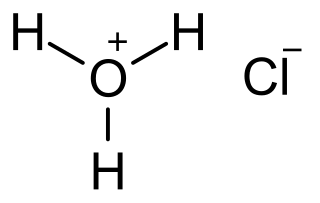
Hydrochloric acid, also known as muriatic acid or spirits of salt, is an aqueous solution of hydrogen chloride with the chemical formula HCl(aq). It is a colorless solution with a distinctive pungent smell. It is classified as a strong acid. It is a component of the gastric acid in the digestive systems of most animal species, including humans. Hydrochloric acid is an important laboratory reagent and industrial chemical.

A descaling agent or chemical descaler is a liquid chemical substance used to remove limescale from metal surfaces in contact with hot water, such as in boilers, water heaters, and kettles. Limescale is either white or brown in colour due to the presence of iron compounds. Glass surfaces may also exhibit scaling stains, as can many ceramic surfaces present in bathrooms and kitchen, and descaling agents can be used safely to remove those stains without affecting the substrate since both ceramics and glass are unreactive to most acids.
Samples used for radiocarbon dating must be handled carefully to avoid contamination. Not all material can be dated by this method; only samples containing organic matter can be tested: the date found will be the date of death of the plants or animals from which the sample originally came.