
Brownian motion is the random motion of particles suspended in a medium.

In cell biology, the cytoplasm describes all material within a eukaryotic cell, enclosed by the cell membrane, except for the cell nucleus. The material inside the nucleus and contained within the nuclear membrane is termed the nucleoplasm. The main components of the cytoplasm are the cytosol, the organelles, and various cytoplasmic inclusions. The cytoplasm is about 80% water and is usually colorless.

A colloid is a mixture in which one substance consisting of microscopically dispersed insoluble particles is suspended throughout another substance. Some definitions specify that the particles must be dispersed in a liquid, while others extend the definition to include substances like aerosols and gels. The term colloidal suspension refers unambiguously to the overall mixture. A colloid has a dispersed phase and a continuous phase. The dispersed phase particles have a diameter of approximately 1 nanometre to 1 micrometre.

In chemistry, a suspension is a heterogeneous mixture of a fluid that contains solid particles sufficiently large for sedimentation. The particles may be visible to the naked eye, usually must be larger than one micrometer, and will eventually settle, although the mixture is only classified as a suspension when and while the particles have not settled out.

Brownian motors are nanoscale or molecular machines that use chemical reactions to generate directed motion in space. The theory behind Brownian motors relies on the phenomenon of Brownian motion, random motion of particles suspended in a fluid resulting from their collision with the fast-moving molecules in the fluid.
Sedimentation equilibrium in a suspension of different particles, such as molecules, exists when the rate of transport of each material in any one direction due to sedimentation equals the rate of transport in the opposite direction due to diffusion. Sedimentation is due to an external force, such as gravity or centrifugal force in a centrifuge.
A sol is a colloidal suspension made out of tiny solid particles in a continuous liquid medium. Sols are stable, so that they do not settle down when left undisturbed, and exhibit the Tyndall effect, which is the scattering of light by the particles in the colloid. The size of the particles can vary from 1 nm - 100 nm. Examples include amongst others blood, pigmented ink, cell fluids, paint, antacids and mud.

The Tyndall effect is light scattering by particles in a colloid such as a very fine suspension. Also known as Tyndall scattering, it is similar to Rayleigh scattering, in that the intensity of the scattered light is inversely proportional to the fourth power of the wavelength, so blue light is scattered much more strongly than red light. An example in everyday life is the blue colour sometimes seen in the smoke emitted by motorcycles, in particular two-stroke machines where the burnt engine oil provides these particles. The same effect can also be observed with tobacco smoke whose fine particles also preferentially scatter blue light.

Diffusion-limited aggregation (DLA) is the process whereby particles undergoing a random walk due to Brownian motion cluster together to form aggregates of such particles. This theory, proposed by T.A. Witten Jr. and L.M. Sander in 1981, is applicable to aggregation in any system where diffusion is the primary means of transport in the system. DLA can be observed in many systems such as electrodeposition, Hele-Shaw flow, mineral deposits, and dielectric breakdown.
In probability theory and statistics, diffusion processes are a class of continuous-time Markov process with almost surely continuous sample paths. Diffusion process is stochastic in nature and hence is used to model many real-life stochastic systems. Brownian motion, reflected Brownian motion and Ornstein–Uhlenbeck processes are examples of diffusion processes. It is used heavily in statistical physics, statistical analysis, information theory, data science, neural networks, finance and marketing.
In materials science, the sol–gel process is a method for producing solid materials from small molecules. The method is used for the fabrication of metal oxides, especially the oxides of silicon (Si) and titanium (Ti). The process involves conversion of monomers in solution into a colloidal solution (sol) that acts as the precursor for an integrated network of either discrete particles or network polymers. Typical precursors are metal alkoxides. Sol–gel process is used to produce ceramic nanoparticles.
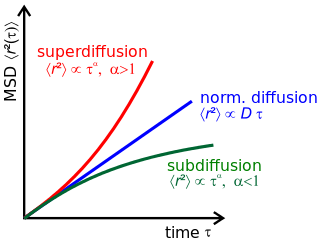
Anomalous diffusion is a diffusion process with a non-linear relationship between the mean squared displacement (MSD), , and time. This behavior is in stark contrast to Brownian motion, the typical diffusion process described by Einstein and Smoluchowski, where the MSD is linear in time.
An -superprocess, , within mathematics probability theory is a stochastic process on that is usually constructed as a special limit of near-critical branching diffusions.

Diffusiophoresis is the spontaneous motion of colloidal particles or molecules in a fluid, induced by a concentration gradient of a different substance. In other words, it is motion of one species, A, in response to a concentration gradient in another species, B. Typically, A is colloidal particles which are in aqueous solution in which B is a dissolved salt such as sodium chloride, and so the particles of A are much larger than the ions of B. But both A and B could be polymer molecules, and B could be a small molecule. For example, concentration gradients in ethanol solutions in water move 1 μm diameter colloidal particles with diffusiophoretic velocities of order 0.1 to 1 μm/s, the movement is towards regions of the solution with lower ethanol concentration. Both species A and B will typically be diffusing but diffusiophoresis is distinct from simple diffusion: in simple diffusion a species A moves down a gradient in its own concentration.

Particle size is a notion introduced for comparing dimensions of solid particles, liquid particles (droplets), or gaseous particles (bubbles). The notion of particle size applies to particles in colloids, in ecology, in granular material, and to particles that form a granular material.

Diffusion is the net movement of anything generally from a region of higher concentration to a region of lower concentration. Diffusion is driven by a gradient in Gibbs free energy or chemical potential. It is possible to diffuse "uphill" from a region of lower concentration to a region of higher concentration, as in spinodal decomposition. Diffusion is a stochastic process due to the inherent randomness of the diffusing entity and can be used to model many real-life stochastic scenarios. Therefore, diffusion and the corresponding mathematical models are used in several fields beyond physics, such as statistics, probability theory, information theory, neural networks, finance, and marketing.

A colloidal crystal is an ordered array of colloidal particles and fine grained materials analogous to a standard crystal whose repeating subunits are atoms or molecules. A natural example of this phenomenon can be found in the gem opal, where spheres of silica assume a close-packed locally periodic structure under moderate compression. Bulk properties of a colloidal crystal depend on composition, particle size, packing arrangement, and degree of regularity. Applications include photonics, materials processing, and the study of self-assembly and phase transitions.

In the physical sciences, a particle is a small localized object which can be described by several physical or chemical properties, such as volume, density, or mass. They vary greatly in size or quantity, from subatomic particles like the electron, to microscopic particles like atoms and molecules, to macroscopic particles like powders and other granular materials. Particles can also be used to create scientific models of even larger objects depending on their density, such as humans moving in a crowd or celestial bodies in motion.
Differential dynamic microscopy (DDM) is an optical technique that allows performing light scattering experiments by means of a simple optical microscope. DDM is suitable for typical soft materials such as for instance liquids or gels made of colloids, polymers and liquid crystals but also for biological materials like bacteria and cells.
An active Brownian particle (ABP) is a model of self-propelled motion in a dissipative environment. It is a nonequilibrium generalization of a Brownian particle.












