Chromatography is a laboratory technique for the separation of a mixture.
The mixture is dissolved in a fluid called the mobile phase, which carries it through a structure holding another material called the stationary phase. The various constituents of the mixture travel at different speeds, causing them to separate. The separation is based on differential partitioning between the mobile and stationary phases. Subtle differences in a compound's partition coefficient result in differential retention on the stationary phase and thus affect the separation.

Size-exclusion chromatography (SEC), also known as molecular sieve chromatography, is a chromatographic method in which molecules in solution are separated by their size, and in some cases molecular weight. It is usually applied to large molecules or macromolecular complexes such as proteins and industrial polymers. Typically, when an aqueous solution is used to transport the sample through the column, the technique is known as gel-filtration chromatography, versus the name gel permeation chromatography, which is used when an organic solvent is used as a mobile phase. The chromatography column is packed with fine, porous beads which are composed of dextran polymers (Sephadex), agarose (Sepharose), or polyacrylamide. The pore sizes of these beads are used to estimate the dimensions of macromolecules. SEC is a widely used polymer characterization method because of its ability to provide good molar mass distribution (Mw) results for polymers.

High-performance liquid chromatography is a technique in analytical chemistry used to separate, identify, and quantify each component in a mixture. It relies on pumps to pass a pressurized liquid solvent containing the sample mixture through a column filled with a solid adsorbent material. Each component in the sample interacts slightly differently with the adsorbent material, causing different flow rates for the different components and leading to the separation of the components as they flow out of the column.

A polyhistidine-tag is an amino acid motif in proteins that consists of at least six histidine (His) residues, often at the N- or C-terminus of the protein. It is also known as hexa histidine-tag, 6xHis-tag, His6 tag and by the trademarked name His-tag. The tag was invented by Roche, although the use of histidines and its vectors are distributed by Qiagen. Various purification kits for histidine-tagged proteins are available from Qiagen, Sigma, Thermo Scientific, GE Healthcare, Macherey-Nagel, Cube Biotech, Clontech, Bio-Rad, and others.
Protein purification is a series of processes intended to isolate one or a few proteins from a complex mixture, usually cells, tissues or whole organisms. Protein purification is vital for the characterization of the function, structure and interactions of the protein of interest. The purification process may separate the protein and non-protein parts of the mixture, and finally separate the desired protein from all other proteins. Separation of one protein from all others is typically the most laborious aspect of protein purification. Separation steps usually exploit differences in protein size, physico-chemical properties, binding affinity and biological activity. The pure result may be termed protein isolate.
Affinity chromatography is a method of separating biochemical mixture based on a highly specific interaction between antigen and antibody, enzyme and substrate, receptor and ligand, or protein and nucleic acid. It is a type of chromatographic laboratory technique used for purifying biological molecules within a mixture by exploiting molecular properties.

Column chromatography in chemistry is a chromatography method used to isolate a single chemical compound from a mixture. Chromatography is able to separate substances based on differential adsorption of compounds to the adsorbent; compounds move through the column at different rates, allowing them to be separated into fractions. The technique is widely applicable, as many different adsorbents can be used with a wide range of solvents. The technique can be used on scales from micrograms up to kilograms. The main advantage of column chromatography is the relatively low cost and disposability of the stationary phase used in the process. The latter prevents cross-contamination and stationary phase degradation due to recycling. Column chromatography can be done using gravity to move the solvent, or using compressed gas to push the solvent through the column.

Ion exchange is an exchange of ions between two electrolytes or between an electrolyte solution and a complex. In most cases the term is used to denote the processes of purification, separation, and decontamination of aqueous and other ion-containing solutions with solid polymeric or mineralic "ion exchangers".

Ion chromatography is a chromatography process that separates ions and polar molecules based on their affinity to the ion exchanger. It works on almost any kind of charged molecule—including large proteins, small nucleotides, and amino acids. However, ion chromatography must be done in conditions that are one unit away from the isoelectric point of a protein.

Solid-phase extraction (SPE) is a sample preparation process by which compounds that are dissolved or suspended in a liquid mixture are separated from other compounds in the mixture according to their physical and chemical properties. Analytical laboratories use solid phase extraction to concentrate and purify samples for analysis. Solid phase extraction can be used to isolate analytes of interest from a wide variety of matrices, including urine, blood, water, beverages, soil, and animal tissue.
Fast protein liquid chromatography (FPLC), is a form of liquid chromatography that is often used to analyze or purify mixtures of proteins. As in other forms of chromatography, separation is possible because the different components of a mixture have different affinities for two materials, a moving fluid and a porous solid. In FPLC the mobile phase is an aqueous solution, or "buffer". The buffer flow rate is controlled by a positive-displacement pump and is normally kept constant, while the composition of the buffer can be varied by drawing fluids in different proportions from two or more external reservoirs.
The stationary phase is a resin composed of beads, usually of cross-linked agarose, packed into a cylindrical glass or plastic column. FPLC resins are available in a wide range of bead sizes and surface ligands depending on the application.
Reversed-phase chromatography includes any chromatographic method that uses a hydrophobic stationary phase.
RPC refers to liquid chromatography.

Hydrophilic interaction chromatography is a variant of normal phase liquid chromatography that partly overlaps with other chromatographic applications such as ion chromatography and reversed phase liquid chromatography. HILIC uses hydrophilic stationary phases with reversed-phase type eluents. The name was suggested by Dr. Andrew Alpert in his 1990 paper on the subject. He described the chromatographic mechanism for it as liquid-liquid partition chromatography where analytes elute in order of increasing polarity, a conclusion supported by a review and re-evaluation of published data.
DNA separation by silica adsorption is a method of DNA separation that is based on DNA molecules binding to silica surfaces in the presence of certain salts and under certain pH conditions, usually conducted on a microchip coated in silica channels.

Diethylaminoethyl cellulose (DEAE-C) is a positively charged resin used in ion-exchange chromatography, a type of column chromatography, for the separation and purification of proteins and nucleic acids. Gel matrix beads are derivatized with diethylaminoethanol (DEAE) and lock negatively charged proteins or nucleic acids into the matrix. The proteins are released from the resin by increasing the salt concentration of the solvent or changing the pH of the solution as to change the charge on the protein.
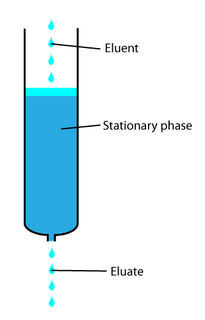
In analytical and organic chemistry, elution is the process of extracting one material from another by washing with a solvent; as in washing of loaded ion-exchange resins to remove captured ions.
Displacement chromatography is a chromatography technique in which a sample is placed onto the head of the column and is then displaced by a solute that is more strongly sorbed than the components of the original mixture. The result is that the components are resolved into consecutive “rectangular” zones of highly concentrated pure substances rather than solvent-separated “peaks”. It is primarily a preparative technique; higher product concentration, higher purity, and increased throughput may be obtained compared to other modes of chromatography.

Desalting and buffer exchange are methods to separate soluble macromolecules from smaller molecules (desalting) or replace the buffer system used for another one suitable for a downstream application. These methods are based on gel filtration chromatography, also called molecular sieve chromatography, which is a form of size-exclusion chromatography. Desalting and buffer exchange are two of the most common gel filtration chromatography applications, and they can be performed using the same resin.
Anion-exchange chromatography is a process that separates substances based on their charges using an ion-exchange resin containing positively charged groups, such as diethyl-aminoethyl groups (DEAE). In solution, the resin is coated with positively charged counter-ions (cations). Anion exchange resins will bind to negatively charged molecules, displacing the counter-ion. Anion exchange chromatography is commonly used to purify proteins, amino acids, sugars/carbohydrates and other acidic substances with a negative charge at higher pH levels. The tightness of the binding between the substance and the resin is based on the strength of the negative charge of the substance.










