 | |
 | |
| Combination of | |
|---|---|
| Ethinylestradiol | Estrogen |
| Cyproterone acetate | Progestogen; Antiandrogen |
| Clinical data | |
| Trade names | Diane, Diane-35, others |
| Other names | EE/CPA; Co-cyprindiol; SHB 209 AB; SHB 209 AE; SH-81041 |
| Routes of administration | By mouth |
| Drug class | Estrogen; Progestogen; Antiandrogen |
| ATC code | |
| Legal status | |
| Legal status | |
| Identifiers | |
| CAS Number | |
| PubChem CID | |
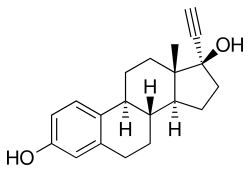
Ethinylestradiol/cyproterone acetate (EE/CPA), also known as co-cyprindiol and sold under the brand names Diane and Diane-35 among others, is a combination of ethinylestradiol (EE), an estrogen, and cyproterone acetate (CPA), a progestin and antiandrogen, which is used as a birth control pill to prevent pregnancy in women. [2] It is also used to treat androgen-dependent conditions in women such as acne, seborrhea, excessive facial/body hair growth, scalp hair loss, and high androgen levels associated with Polycystic ovary syndrome. [3] [4] [5] [6] [7] [8] The medication is taken by mouth once daily for 21 days, followed by a 7-day free interval. [2]
