Indium selenide, an inorganic compound composed of indium and selenium, refers to:
Indium selenide, an inorganic compound composed of indium and selenium, refers to:

Indium is a chemical element; it has symbol In and atomic number 49. It is a silvery-white post-transition metal and one of the softest elements. Chemically, indium is similar to gallium and thallium, and its properties are largely intermediate between the two. It was discovered in 1863 by Ferdinand Reich and Hieronymous Theodor Richter by spectroscopic methods and named for the indigo blue line in its spectrum.
CIGS may refer to:
A selenide is a chemical compound containing a selenium with oxidation number of −2. Similar to sulfide, selenides occur both as inorganic compounds and as organic derivatives, which are called organoselenium compound.

An infrared detector is a detector that reacts to infrared (IR) radiation. The two main types of detectors are thermal and photonic (photodetectors).
Organoselenium chemistry is the science exploring the properties and reactivity of organoselenium compounds, chemical compounds containing carbon-to-selenium chemical bonds. Selenium belongs with oxygen and sulfur to the group 16 elements or chalcogens, and similarities in chemistry are to be expected. Organoselenium compounds are found at trace levels in ambient waters, soils and sediments.

Copper indium gallium (di)selenide (CIGS) is a I-III-VI2 semiconductor material composed of copper, indium, gallium, and selenium. The material is a solid solution of copper indium selenide (often abbreviated "CIS") and copper gallium selenide. It has a chemical formula of CuIn1−xGaxSe2, where the value of x can vary from 0 (pure copper indium selenide) to 1 (pure copper gallium selenide). CIGS is a tetrahedrally bonded semiconductor, with the chalcopyrite crystal structure, and a bandgap varying continuously with x from about 1.0 eV (for copper indium selenide) to about 1.7 eV (for copper gallium selenide).
Lead selenide (PbSe), or lead(II) selenide, a selenide of lead, is a semiconductor material. It forms cubic crystals of the NaCl structure; it has a direct bandgap of 0.27 eV at room temperature. A grey solid, it is used for manufacture of infrared detectors for thermal imaging. The mineral clausthalite is a naturally occurring lead selenide.
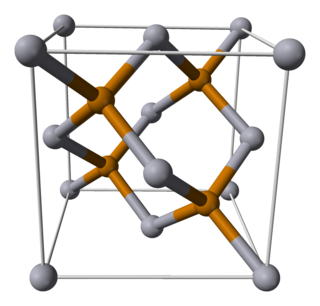
Mercury telluride (HgTe) is a binary chemical compound of mercury and tellurium. It is a semi-metal related to the II-VI group of semiconductor materials. Alternative names are mercuric telluride and mercury(II) telluride.
Indium(III) telluride (In2Te3) is a inorganic compound. A black solid, it is sometimes described as an intermetallic compound, because it has properties that are metal-like and salt like. It is a semiconductor that has attracted occasional interest for its thermoelectric and photovoltaic applications. No applications have been implemented commercially however.
In chemistry, the Grimm–Sommerfeld rule predicts that binary compounds with covalent character that have an average of 4 electrons per atom will have structures where both atoms are tetrahedrally coordinated. Examples are silicon carbide, the III-V semiconductors indium phosphide and gallium arsenide, the II-VI semiconductors, cadmium sulfide, cadmium selenide.

Selenium compounds are compounds containing the element selenium (Se). Among these compounds, selenium has various oxidation states, the most common ones being −2, +4, and +6. Selenium compounds exist in nature in the form of various minerals, such as clausthalite, guanajuatite, tiemannite, crookesite etc., and can also coexist with sulfide minerals such as pyrite and chalcopyrite. For many mammals, selenium compounds are essential. For example, selenomethionine and selenocysteine are selenium-containing amino acids present in the human body. Selenomethionine participates in the synthesis of selenoproteins. The reduction potential and pKa (5.47) of selenocysteine are lower than those of cysteine, making some proteins have antioxidant activity. Selenium compounds have important applications in semiconductors, glass and ceramic industries, medicine, metallurgy and other fields.
Indium(III) selenide is a compound of indium and selenium. It has potential for use in photovoltaic devices and has been the subject of extensive research. The two most common phases, α and β, have a layered structure, while γ has a "defect wurtzite structure." In all, five polymorphs are known: α, β, γ, δ, κ. The α-β phase transition is accompanied by a change in electrical conductivity. The band gap of γ-In2Se3 is approximately 1.9 eV.
The indium chalcogenides include all compounds of indium with the chalcogen elements, oxygen, sulfur, selenium and tellurium. (Polonium is excluded as little is known about its compounds with indium). The best-characterised compounds are the In(III) and In(II) chalcogenides e.g. the sulfides In2S3 and InS.
This group of compounds has attracted a lot of research attention because they include semiconductors, photovoltaics and phase-change materials. In many applications indium chalcogenides are used as the basis of ternary and quaternary compounds such as indium tin oxide, ITO and copper indium gallium selenide, CIGS.

Gallium(II) selenide (GaSe) is a chemical compound. It has a hexagonal layer structure, similar to that of GaS. It is a photoconductor, a second harmonic generation crystal in nonlinear optics, and has been used as a far-infrared conversion material at 14–31 THz and above.

A copper indium gallium selenide solar cell is a thin-film solar cell used to convert sunlight into electric power. It is manufactured by depositing a thin layer of copper indium gallium selenide solid solution on glass or plastic backing, along with electrodes on the front and back to collect current. Because the material has a high absorption coefficient and strongly absorbs sunlight, a much thinner film is required than of other semiconductor materials.
Solar Frontier Kabushiki Kaisha is a Japanese photovoltaic company that develops and manufactures thin film solar cells using CIGS technology. It is a fully owned subsidiary of Showa Shell Sekiyu and located in Minato, Tokyo, Japan. The company was founded in 2006 as Showa Shell Solar, and renamed Solar Frontier in April 2010.
Copper selenide is an inorganic binary compound between copper and selenium. The chemical formula depends on the ratio between the two elements, such as CuSe or Cu2Se.
Calcium selenide (CaSe) is a chemical compound consisting of the elements calcium and selenium in equal stoichiometric ratio.
Indium(II) selenide (InSe) is an inorganic compound composed of indium and selenium. It is a III-VI layered semiconductor. The solid has a structure consisting of two-dimensional layers bonded together only by van der Waals forces. Each layer has the atoms in the order Se-In-In-Se.