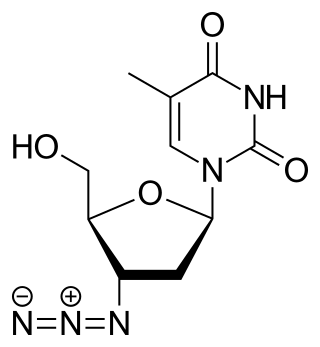
Zidovudine (ZDV), also known as azidothymidine (AZT), was the first antiretroviral medication used to prevent and treat HIV/AIDS. It is generally recommended for use in combination with other antiretrovirals. It may be used to prevent mother-to-child spread during birth or after a needlestick injury or other potential exposure. It is sold both by itself and together as lamivudine/zidovudine and abacavir/lamivudine/zidovudine. It can be used by mouth or by slow injection into a vein.
The management of HIV/AIDS normally includes the use of multiple antiretroviral drugs as a strategy to control HIV infection. There are several classes of antiretroviral agents that act on different stages of the HIV life-cycle. The use of multiple drugs that act on different viral targets is known as highly active antiretroviral therapy (HAART). HAART decreases the patient's total burden of HIV, maintains function of the immune system, and prevents opportunistic infections that often lead to death. HAART also prevents the transmission of HIV between serodiscordant same-sex and opposite-sex partners so long as the HIV-positive partner maintains an undetectable viral load.
Reverse-transcriptase inhibitors (RTIs) are a class of antiretroviral drugs used to treat HIV infection or AIDS, and in some cases hepatitis B. RTIs inhibit activity of reverse transcriptase, a viral DNA polymerase that is required for replication of HIV and other retroviruses.

Zalcitabine, also called dideoxycytidine, is a nucleoside analog reverse-transcriptase inhibitor (NRTI) sold under the trade name Hivid. Zalcitabine was the third antiretroviral to be approved by the Food and Drug Administration (FDA) for the treatment of HIV/AIDS. It is used as part of a combination regimen.

Stavudine (d4T), sold under the brand name Zerit among others, is an antiretroviral medication used to prevent and treat HIV/AIDS. It is generally recommended for use with other antiretrovirals. It may be used for prevention after a needlestick injury or other potential exposure. However, it is not a first-line treatment. It is given by mouth.

Lamivudine, commonly called 3TC, is an antiretroviral medication used to prevent and treat HIV/AIDS. It is also used to treat chronic hepatitis B when other options are not possible. It is effective against both HIV-1 and HIV-2. It is typically used in combination with other antiretrovirals such as zidovudine, dolutegravir, and abacavir. Lamivudine may be included as part of post-exposure prevention in those who have been potentially exposed to HIV. Lamivudine is taken by mouth as a liquid or tablet.

Abacavir, sold under the brand name Ziagen among others, is a medication used to treat HIV/AIDS. Similar to other nucleoside analog reverse-transcriptase inhibitors (NRTIs), abacavir is used together with other HIV medications, and is not recommended by itself. It is taken by mouth as a tablet or solution and may be used in children over the age of three months.

Emtricitabine, with trade name Emtriva, is a nucleoside reverse-transcriptase inhibitor (NRTI) for the prevention and treatment of HIV infection in adults and children. In 2019, it was the 494th most commonly prescribed medication in the United States, with more than 3 thousand prescriptions.

Nevirapine (NVP), sold under the brand name Viramune among others, is a medication used to treat and prevent HIV/AIDS, specifically HIV-1. It is generally recommended for use with other antiretroviral medications. It may be used to prevent mother to child spread during birth but is not recommended following other exposures. It is taken by mouth.

Entecavir, sold under the brand name Baraclude, is an antiviral medication used in the treatment of hepatitis B virus infection. In those with both HIV/AIDS and hepatitis B virus antiretroviral medication should also be used. Entecavir is taken by mouth as a tablet or solution.
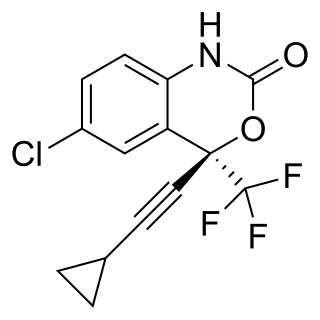
Efavirenz (EFV), sold under the brand names Sustiva among others, is an antiretroviral medication used to treat and prevent HIV/AIDS. It is generally recommended for use with other antiretrovirals. It may be used for prevention after a needlestick injury or other potential exposure. It is sold both by itself and in combination as efavirenz/emtricitabine/tenofovir. It is taken by mouth.

Abacavir/lamivudine/zidovudine, sold under the brand name Trizivir, is a fixed-dose combination antiretroviral medication for the treatment of HIV/AIDS. It contains three reverse transcriptase inhibitors patented by GlaxoSmithKline and marketed by a joint venture with Pfizer, ViiV Healthcare:

Efavirenz/emtricitabine/tenofovir, sold under the brand name Atripla among others, is a fixed-dose combination antiretroviral medication used to treat HIV/AIDS. It contains efavirenz, emtricitabine, and tenofovir disoproxil. It can be used by itself or together with other antiretroviral medications. It is taken by mouth.

Abacavir/lamivudine, sold under the brand name Kivexa among others, is a fixed-dose combination antiretroviral medication used to treat HIV/AIDS. It contains abacavir and lamivudine. It is generally recommended for use with other antiretrovirals. It is commonly used as part of the preferred treatment in children. It is taken by mouth as a tablet.
Discovery and development of nucleoside and nucleotide reverse-transcriptase inhibitors began in the 1980s when the AIDS epidemic hit Western societies. NRTIs inhibit the reverse transcriptase (RT), an enzyme that controls the replication of the genetic material of the human immunodeficiency virus (HIV). The first NRTI was zidovudine, approved by the U.S. Food and Drug Administration (FDA) in 1987, which was the first step towards treatment of HIV. Six NRTI agents and one NtRTI have followed. The NRTIs and the NtRTI are analogues of endogenous 2´-deoxy-nucleoside and nucleotide. Drug-resistant viruses are an inevitable consequence of prolonged exposure of HIV-1 to anti-HIV drugs.

Abacavir/dolutegravir/lamivudine, sold under the brand name Triumeq among others, is a fixed-dose combination antiretroviral medication for the treatment of HIV/AIDS. It is a combination of three medications with different and complementary mechanisms of action: abacavir, dolutegravir and lamivudine.
Lamivudine/nevirapine/stavudine (3TC/NVP/d4T) is a fixed-dose combination antiretroviral medication used to treat HIV/AIDS. It contains lamivudine, nevirapine, and stavudine. It is either used by itself or along with other antiretrovirals. It is taken by mouth twice a day.
Lamivudine/nevirapine/zidovudine (3TC/NVP/AZT) is a fixed-dose combination antiretroviral medication used to treat HIV/AIDS. It contains lamivudine, nevirapine, and zidovudine. It is either used by itself or along with other antiretrovirals. It is a recommended treatment in those who are pregnant. It is taken by mouth twice a day.

Bictegravir/emtricitabine/tenofovir alafenamide, sold under the brand name Biktarvy, is a fixed-dose combination antiretroviral medication for the treatment of HIV/AIDS. It contains bictegravir, a human immunodeficiency virus type 1 (HIV-1) integrase strand transfer inhibitor; emtricitabine, an HIV-1 nucleoside analog reverse transcriptase inhibitor; and tenofovir alafenamide, an HIV-1 nucleoside analog reverse transcriptase inhibitor.
Dolutegravir/lamivudine, sold under the brand name Dovato, is a fixed-dose combination antiretroviral medication for the treatment of HIV/AIDS. each pill contains 50 mg dolutegravir, as the salt, an integrase strand transfer inhibitor (INSTI), and 300 mg lamivudine, a nucleoside analogue reverse transcriptase inhibitor (NRTI). It is taken by mouth.














