
Edward Teller was a Hungarian-American theoretical physicist who is known colloquially as "the father of the hydrogen bomb", although he did not care for the title, considering it to be in poor taste. Throughout his life, Teller was known both for his scientific ability and for his difficult interpersonal relations and volatile personality.
A timeline of atomic and subatomic physics.

Cyclobutadiene is an organic compound with the formula C4H4. It is very reactive owing to its tendency to dimerize. Although the parent compound has not been isolated, some substituted derivatives are robust and a single molecule of cyclobutadiene is quite stable. Since the compound degrades by a bimolecular process, the species can be observed by matrix isolation techniques at temperatures below 35 K. It is thought to adopt a rectangular structure.
Vibronic coupling in a molecule involves the interaction between electronic and nuclear vibrational motion. The term "vibronic" originates from the combination of the terms "vibrational" and "electronic", denoting the idea that in a molecule, vibrational and electronic interactions are interrelated and influence each other. The magnitude of vibronic coupling reflects the degree of such interrelation.
The Jahn–Teller effect is an important mechanism of spontaneous symmetry breaking in molecular and solid-state systems which has far-reaching consequences in different fields, and is responsible for a variety of phenomena in spectroscopy, stereochemistry, crystal chemistry, molecular and solid-state physics, and materials science. The effect is named for Hermann Arthur Jahn and Edward Teller, who first reported studies about it in 1937. The Jahn–Teller effect, and the related Renner–Teller effect, are discussed in Section 13.4 of the spectroscopy textbook by Bunker and Jensen.
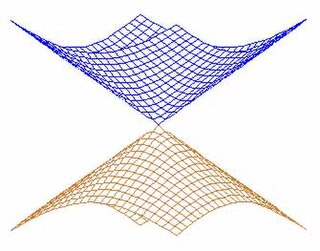
In quantum chemistry, a conical intersection of two or more potential energy surfaces is the set of molecular geometry points where the potential energy surfaces are degenerate (intersect) and the non-adiabatic couplings between these states are non-vanishing. In the vicinity of conical intersections, the Born–Oppenheimer approximation breaks down and the coupling between electronic and nuclear motion becomes important, allowing non-adiabatic processes to take place. The location and characterization of conical intersections are therefore essential to the understanding of a wide range of important phenomena governed by non-adiabatic events, such as photoisomerization, photosynthesis, vision and the photostability of DNA. The conical intersection involving the ground electronic state potential energy surface of the C6H3F3+ molecular ion is discussed in connection with the Jahn–Teller effect in Section 13.4.2 on pages 380-388 of the textbook by Bunker and Jensen.

Cobalt(III) fluoride is the inorganic compound with the formula CoF3. Hydrates are also known. The anhydrous compound is a hygroscopic brown solid. It is used to synthesize organofluorine compounds.

Chromium(II) chloride describes inorganic compounds with the formula CrCl2(H2O)n. The anhydrous solid is white when pure, however commercial samples are often grey or green; it is hygroscopic and readily dissolves in water to give bright blue air-sensitive solutions of the tetrahydrate Cr(H2O)4Cl2. Chromium(II) chloride has no commercial uses but is used on a laboratory-scale for the synthesis of other chromium complexes.
Copper proteins are proteins that contain one or more copper ions as prosthetic groups. Copper proteins are found in all forms of air-breathing life. These proteins are usually associated with electron-transfer with or without the involvement of oxygen (O2). Some organisms even use copper proteins to carry oxygen instead of iron proteins. A prominent copper proteins in humans is in cytochrome c oxidase (cco). The enzyme cco mediates the controlled combustion that produces ATP.

Gold(III) bromide is a dark-red to black crystalline solid. It has the empirical formula AuBr3, but exists primarily as a dimer with the molecular formula Au2Br6 in which two gold atoms are bridged by two bromine atoms. It is commonly referred to as gold(III) bromide, gold tribromide, and rarely but traditionally auric bromide, and sometimes as digold hexabromide. As is similar with the other gold halides, this compound is unique for being a coordination complex of a group 11 transition metal that is stable in an oxidation state of three whereas copper or silver complexes persist in oxidation states of one or two.
The Renner–Teller effect is observed in the spectra of molecules having electronic states that allow vibration through a linear configuration. For such molecules electronic states that are doubly degenerate at linearity will split into two close-lying nondegenerate states for non-linear configurations. As part of the Renner-Teller effect, the rovibronic levels of such a pair of states will be strongly Coriolis coupled by the rotational kinetic energy operator causing a breakdown of the Born-Oppenheimer approximation. This is to be contrasted with the Jahn-Teller effect which occurs for polyatomic molecules in electronic states that allow vibration through a symmetric nonlinear configuration, where the electronic state is degenerate, and which further involves a breakdown of the Born-Oppenheimer approximation but here caused by the vibrational kinetic energy operator.
Hermann Arthur Jahn was an English scientist of German origin. With Edward Teller, he identified the Jahn–Teller effect.
An electronic effect influences the structure, reactivity, or properties of molecule but is neither a traditional bond nor a steric effect. In organic chemistry, the term stereoelectronic effect is also used to emphasize the relation between the electronic structure and the geometry (stereochemistry) of a molecule.

Ferrous oxalate, or iron(II) oxalate, is an inorganic compound with the formula FeC2O4 · xH2O where x is typically 2. These are orange compounds, poorly soluble in water.
Lanthanum manganite is an inorganic compound with the formula LaMnO3, often abbreviated as LMO. Lanthanum manganite is formed in the perovskite structure, consisting of oxygen octahedra with a central Mn atom. The cubic perovskite structure is distorted into an orthorhombic structure by a strong Jahn–Teller distortion of the oxygen octahedra.
In solid state physics and physical chemistry, the fine electronic structure of a solid are the features of the electronic bands induced by intrinsic interactions between charge carriers. Valence and conduction bands split slightly compared to the difference between the various bands. Some mechanisms that allow it are angular momentum couplings, spin-orbit coupling, lattice distortions, and other interactions described by crystal field theory.
The pseudo Jahn–Teller effect (PJTE), occasionally also known as second-order JTE, is a direct extension of the Jahn–Teller effect (JTE) where spontaneous symmetry breaking in polyatomic systems occurs even in nondegenerate electronic states under the influence of sufficiently low-lying excited states of appropriate symmetry. "The pseudo Jahn–Teller effect is the only source of instability and distortions of high-symmetry configurations of polyatomic systems in nondegenerate states, and it contributes significantly to the instability in degenerate states".

Hexaphosphabenzene is a valence isoelectronic analogue of benzene and is expected to have a similar planar structure due to resonance stabilization. Although several other allotropes of phosphorus are stable, no evidence for the existence of P6 has been reported. Preliminary ab initio calculations on the trimerisation of P2 leading to the formation of the cyclic P6 were performed, and it was predicted that hexaphosphabenzene would decompose to free P2 with an energy barrier of 13−15.4 kcal mol−1, and would therefore not be observed in the uncomplexed state under normal experimental conditions. The presence of an added solvent, such as ethanol, might lead to the formation of intermolecular hydrogen bonds which may block the destabilizing interaction between phosphorus lone pairs and consequently stabilize P6. The moderate barrier suggests that hexaphosphabenzene could be synthesized from a [2+2+2] cycloaddition of three P2 molecules. Currently, this is a synthetic endeavour which remains to be conquered.

Mary Jane Shultz is an American professor and researcher in physical, environmental, materials and surface chemistry at Tufts University. Since 2013, she is also a visiting professor at the Chinese Academy of Sciences.

Isaac B. Bersuker is a Soviet-Moldоvan-American theoretical physicist and quantum chemist whose principal research is in chemical physics, solid-state physics, and theoretical chemistry. Known for his "life-long years of experience in theoretical chemistry" working on the electronic structure and properties of coordination compounds, Isaac B. Bersuker is “one of the most widely recognized authorities” in the theory of the Jahn-Teller effect (JTE) and the pseudo-Jahn-Teller effect (PJTE). His accomplishments include explaining the polarization of the atomic core in Rydberg atoms, the effect of tunneling splitting in molecules and solids with a strong JTE, and the discovery of the PJTE origin of ferroelectricity in cubic perovskites. Known as the leading expert in JTE and PJTE, Bersuker is the permanent Chairman of the International Steering Committee of the Jahn-Teller symposia. His present affiliation is with the Oden Institute for Computational Engineering and Science of the Department of Chemistry of the University of Texas at Austin.