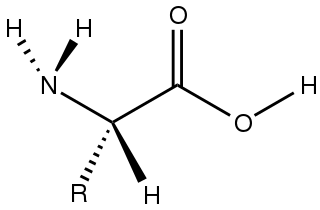
Amino acids are organic compounds that contain both amino and carboxylic acid functional groups. Although over 500 amino acids exist in nature, by far the most important are the 22 α-amino acids incorporated into proteins. Only these 22 appear in the genetic code of life.

Peptides are short chains of amino acids linked by peptide bonds. A polypeptide is a longer, continuous, unbranched peptide chain. Polypeptides that have a molecular mass of 10,000 Da or more are called proteins. Chains of fewer than twenty amino acids are called oligopeptides, and include dipeptides, tripeptides, and tetrapeptides.

In organic chemistry, a peptide bond is an amide type of covalent chemical bond linking two consecutive alpha-amino acids from C1 of one alpha-amino acid and N2 of another, along a peptide or protein chain.
Atrial natriuretic peptide (ANP) or atrial natriuretic factor (ANF) is a natriuretic peptide hormone secreted from the cardiac atria that in humans is encoded by the NPPA gene. Natriuretic peptides are a family of hormone/paracrine factors that are structurally related. The main function of ANP is causing a reduction in expanded extracellular fluid (ECF) volume by increasing renal sodium excretion. ANP is synthesized and secreted by cardiac muscle cells in the walls of the atria in the heart. These cells contain volume receptors which respond to increased stretching of the atrial wall due to increased atrial blood volume.
A signal peptide is a short peptide present at the N-terminus of most newly synthesized proteins that are destined toward the secretory pathway. These proteins include those that reside either inside certain organelles, secreted from the cell, or inserted into most cellular membranes. Although most type I membrane-bound proteins have signal peptides, most type II and multi-spanning membrane-bound proteins are targeted to the secretory pathway by their first transmembrane domain, which biochemically resembles a signal sequence except that it is not cleaved. They are a kind of target peptide.
The N-terminus (also known as the amino-terminus, NH2-terminus, N-terminal end or amine-terminus) is the start of a protein or polypeptide, referring to the free amine group (-NH2) located at the end of a polypeptide. Within a peptide, the amine group is bonded to the carboxylic group of another amino acid, making it a chain. That leaves a free carboxylic group at one end of the peptide, called the C-terminus, and a free amine group on the other end called the N-terminus. By convention, peptide sequences are written N-terminus to C-terminus, left to right (in LTR writing systems). This correlates the translation direction to the text direction, because when a protein is translated from messenger RNA, it is created from the N-terminus to the C-terminus, as amino acids are added to the carboxyl end of the protein.
Neuropeptides are chemical messengers made up of small chains of amino acids that are synthesized and released by neurons. Neuropeptides typically bind to G protein-coupled receptors (GPCRs) to modulate neural activity and other tissues like the gut, muscles, and heart.

In organic chemistry, peptide synthesis is the production of peptides, compounds where multiple amino acids are linked via amide bonds, also known as peptide bonds. Peptides are chemically synthesized by the condensation reaction of the carboxyl group of one amino acid to the amino group of another. Protecting group strategies are usually necessary to prevent undesirable side reactions with the various amino acid side chains. Chemical peptide synthesis most commonly starts at the carboxyl end of the peptide (C-terminus), and proceeds toward the amino-terminus (N-terminus). Protein biosynthesis in living organisms occurs in the opposite direction.
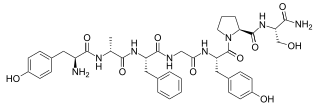
Dermorphin is a hepta-peptide first isolated from the skin of South American frogs belonging to the genus Phyllomedusa. The peptide is a natural opioid that binds as an agonist with high potency and selectivity to mu opioid receptors. Dermorphin is about 30–40 times more potent than morphine, but theoretically may be less likely to produce drug tolerance and addiction due to its high potency. The amino acid sequence of dermorphin is H-Tyr-D-Ala-Phe-Gly-Tyr-Pro-Ser-NH2.

MHC class I molecules are one of two primary classes of major histocompatibility complex (MHC) molecules and are found on the cell surface of all nucleated cells in the bodies of vertebrates. They also occur on platelets, but not on red blood cells. Their function is to display peptide fragments of proteins from within the cell to cytotoxic T cells; this will trigger an immediate response from the immune system against a particular non-self antigen displayed with the help of an MHC class I protein. Because MHC class I molecules present peptides derived from cytosolic proteins, the pathway of MHC class I presentation is often called cytosolic or endogenous pathway.

Antimicrobial peptides (AMPs), also called host defence peptides (HDPs) are part of the innate immune response found among all classes of life. Fundamental differences exist between prokaryotic and eukaryotic cells that may represent targets for antimicrobial peptides. These peptides are potent, broad spectrum antimicrobials which demonstrate potential as novel therapeutic agents. Antimicrobial peptides have been demonstrated to kill Gram negative and Gram positive bacteria, enveloped viruses, fungi and even transformed or cancerous cells. Unlike the majority of conventional antibiotics it appears that antimicrobial peptides frequently destabilize biological membranes, can form transmembrane channels, and may also have the ability to enhance immunity by functioning as immunomodulators.
The rubiscolins are a group of opioid peptides that are formed during digestion of the ribulose bisphosphate carboxylase/oxygenase (Rubisco) protein from spinach leaves. These peptides have much in common with the better-known gluten exorphins.

Glucagon-like peptide-1 (GLP-1) is a 30- or 31-amino-acid-long peptide hormone deriving from the tissue-specific posttranslational processing of the proglucagon peptide. It is produced and secreted by intestinal enteroendocrine L-cells and certain neurons within the nucleus of the solitary tract in the brainstem upon food consumption. The initial product GLP-1 (1–37) is susceptible to amidation and proteolytic cleavage, which gives rise to the two truncated and equipotent biologically active forms, GLP-1 (7–36) amide and GLP-1 (7–37). Active GLP-1 protein secondary structure includes two α-helices from amino acid position 13–20 and 24–35 separated by a linker region.
There are two known receptors for the vasoactive intestinal peptide (VIP) termed VPAC1 and VPAC2. These receptors bind both VIP and pituitary adenylate cyclase-activating polypeptide (PACAP) to some degree. Both receptors are members of the 7 transmembrane G protein-coupled receptor family.
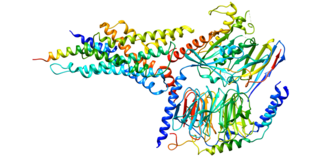
Glucagon-like peptide-2 receptor (GLP-2R) is a protein that in human is encoded by the GLP2R gene located on chromosome 17.
The relaxin receptors are a subclass of four closely related G protein-coupled receptors (GPCR) that bind relaxin peptide hormones.
Hemopressin (Hp) is an alpha hemoglobin fragment with the sequence PVNFKFLSH, originally identified in extracts of rat brain using an enzyme capture technique. It binds cannabinoid receptors, acting as an inverse agonist at CB1 receptors. Longer forms of hemopressin containing 2-3 additional amino acids on the N-terminus have been identified in extracts of mouse brain. These longer hemopressin peptides, named RVD-Hpα and VD-Hpα, bind to CB1 receptors and were originally reported to be agonists. In addition to the Hp peptides from alpha hemoglobin, a related peptide from beta hemoglobin has been found in mouse brain extracts; this peptide, named VD-Hpβ, is also an agonist at CB1 cannabinoid receptors. Hemopressin is not an endogenous peptide but rather an extraction artefact. The only endogenous peptide found endogenously at physiological conditions is RVD-hemopressin (pepcan-12), which has more recently been shown to be a negative allosteric modulator of CB1 receptors and positive allosteric modulator of CB2 receptors. RVD-hemopressin (pepcan-12) is generated from a pro-peptide called pepcan-23 and these peptides are exclusively found in noradrenergic neurons in the brain and in the adrenal medulla.
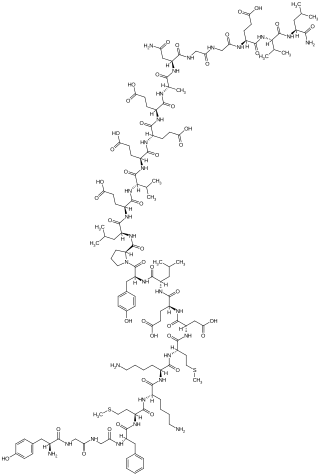
Amidorphin is an endogenous, C-terminally amidated, opioid peptide generated as a cleavage product of proenkephalin A in some mammalian species; in humans and most other species, the peptide is 1 residue longer and is not amidated. Amidorphin is widely distributed in the mammalian brain, with particularly high concentrations found in the striatum, and outside of the brain in adrenal medulla and posterior pituitary. The 26-residue peptide named amidorphin is found in several species including bovine, sheep, and pig. Humans and commonly studied lab animals produce a 27-residue peptide that does not have an amidated C-terminal residue; this is due to the absence of a Gly in the precursor sequence and replacement with Ala, which is not a substrate for the amidating enzyme. The properties of the 27-residue peptide are presumably similar to those of amidorphin, although this has not been adequately tested.

Brain natriuretic peptide (BNP), also known as B-type natriuretic peptide, is a hormone secreted by cardiomyocytes in the heart ventricles in response to stretching caused by increased ventricular blood volume. BNP is one of the three natriuretic peptides, in addition to atrial natriuretic peptide (ANP) and C-type natriuretic peptide ( CNP). BNP was first discovered in porcine brain tissue in 1988, which led to its initial naming as "brain natriuretic peptide", although subsequent research revealed that BNP is primarily produced and secreted by the ventricular myocardium in response to increased ventricular blood volume and stretching. To reflect its true source, BNP is now often referred to as "B-type natriuretic peptide" while retaining the same acronym.

EpiVacCorona is a peptide-based vaccine against COVID-19 developed by the Russian VECTOR Center of Virology. The lack of protective effectiveness of EpiVacCorona, which is still in use in Russia, has been reported in scientific literature and in the media. The vaccine consists of three chemically synthesized peptides that are conjugated to a large carrier protein. This protein is a fusion product of a viral nucleocapsid protein and a bacterial MBP protein. A phase III clinical trial to show whether or not the vaccine can protect people against COVID-19 was launched in November 2020 with more than three thousand participants. The conclusions and results of the trial have not been made public.













