Naphthol may refer to:
Naphthol may refer to:

Carbaryl is a chemical in the carbamate family used chiefly as an insecticide. It is a white crystalline solid previously sold under the brand name Sevin, which was a trademark of the Bayer Company. The Sevin trademark has since been acquired by GardenTech, which has eliminated carbaryl from most Sevin formulations. Union Carbide discovered carbaryl and introduced it commercially in 1958. Bayer purchased Aventis CropScience in 2002, a company that included Union Carbide pesticide operations. Carbaryl was the third-most-used insecticide in the United States for home gardens, commercial agriculture, and forestry and rangeland protection. As a veterinary drug, it is known as carbaril (INN).

Molisch's test is a sensitive chemical test, named after Austrian botanist Hans Molisch, for the presence of carbohydrates, based on the dehydration of the carbohydrate by sulfuric acid or hydrochloric acid to produce an aldehyde, which condenses with two molecules of a phenol, resulting in a violet ring.
1,1′-Bi-2-naphthol (BINOL) is an organic compound that is often used as a ligand for transition-metal catalysed asymmetric synthesis. BINOL has axial chirality and the two enantiomers can be readily separated and are stable toward racemisation. The specific rotation of the two enantiomers is 35.5° (c = 1 in THF), with the R enantiomer being the dextrorotary one. BINOL is a precursor for another chiral ligand called BINAP. The volumetric mass density of the two enantiomers is 0.62 g cm−3.
Oxidative coupling in chemistry is a coupling reaction of two molecular entities through an oxidative process. Usually oxidative couplings are catalysed by a transition metal complex like in classical cross-coupling reactions, although the underlying mechanism is different due to the oxidation process that requires an external oxidant. Many such couplings utilize dioxygen as the stoichiometric oxidant but proceed by electron transfer.
In organic chemistry, an azo coupling is an organic reaction between a diazonium compound and another aromatic compound that produces an azo compound. In this electrophilic aromatic substitution reaction, the aryldiazonium cation is the electrophile and the activated arene is a nucleophile. In most cases, including the examples below, the diazonium compound is also aromatic.
The Bucherer carbazole synthesis is a chemical reaction used to synthesize carbazoles from naphthols and aryl hydrazines using sodium bisulfite. The reaction is named after Hans Theodor Bucherer.

2-Naphthylamine is one of two isomeric aminonaphthalenes, compounds with the formula C10H7NH2. It is a colorless solid, but samples take on a reddish color in air because of oxidation. It was formerly used to make azo dyes, but it is a known carcinogen and has largely been replaced by less toxic compounds.
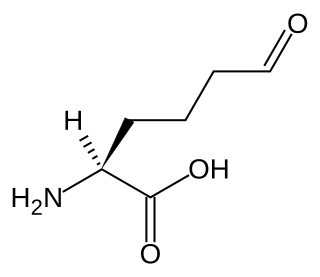
Allysine is a derivative of lysine that features a formyl group in place of the terminal amine. The free amino acid does not exist, but the allysine residue does. It is produced by aerobic oxidation of lysine residues by the enzyme lysyl oxidase. The transformation is an example of a post-translational modification. The semi-aldehyde form exists in equilibrium with a cyclic derivative.

2-Naphthol, or β-naphthol, is a fluorescent colorless (or occasionally yellow) crystalline solid with the formula C10H7OH. It is an isomer of 1-naphthol, differing by the location of the hydroxyl group on the naphthalene ring. The naphthols are naphthalene homologues of phenol, but more reactive. Both isomers are soluble in simple alcohols, ethers, and chloroform. 2-Naphthol is a widely used intermediate for the production of dyes and other compounds.
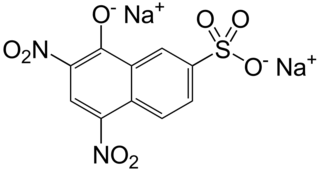
Naphthol yellow S is an organic compound that is a dye. It is a derivative of 1-naphthol. At one time it was a popular food colorant but it was delisted in 1959 in the U.S.
Acid yellow may refer to:

Acid Orange 7, also known as 2-naphthol orange is an azo dye. It is used for dyeing wool.
1-Naphthol, or α-naphthol, is a fluorescent organic compound with the formula C10H7OH. It is a white solid. It is an isomer of 2-naphthol differing by the location of the hydroxyl group on the naphthalene ring. The naphthols are naphthalene homologues of phenol, with the hydroxyl group being more reactive than in the phenols. Both isomers are soluble in simple alcohols, ethers, and chloroform. They are precursors to a variety of useful compounds. Naphthols are used as biomarkers for livestock and humans exposed to polycyclic aromatic hydrocarbons.

Naphthol Green B is a coordination complex of iron that is used as a dye.
C10H8O may refer to:
Voges–Proskauer or VP is a test used to detect acetoin in a bacterial broth culture. The test is performed by adding alpha-naphthol and potassium hydroxide to the Voges-Proskauer broth, which is a glucose-phosphate broth that has been inoculated with bacteria. A cherry red color indicates a positive result, while a yellow-brown color indicates a negative result.
Venkatagiri Sari is a sari style woven in Venkatagiri of Tirupati district in the Indian state of Andhra Pradesh. It was registered as one of the geographical indication from Andhra Pradesh by Geographical Indications of Goods Act, 1999. Venkatagiri saris are known for their fine weaving. These style of saris can also be found in the villages of Sengunthapuram, Variyankaval, Elaiyur, Kallathur, Andimadam and Marudhur villages.

4-Amino-2-methyl-1-naphthol is a menadione analog. Its hydrochloride (HCl) salt is often called vitamin K5. The HCl salt has been used as a medicine for vitamin K deficiency under tradenames such as Synkamin, which was sold by Parke-Davis, but has since been discontinued.
The molecular formula C11H11NO (molar mass: 173.211 g/mol, exact mass: 173.0841 u) may refer to:

4-amino-3-methyl-1-naphthol is a synthetic menadione analog. It is also known as vitamin K7, and was named as such in 1950 when it was recognized as a compound with vitamin K activity.