The National Pharmaceutical Product Index or NAPPI is a comprehensive database of pharmaceutical codes for medical products classification used in South Africa. Each product has a unique NAPPI code which enables electronic data interchange throughout the health care delivery chain.
NAPPI is governed by the NAPPI Advisory Board (NAB), a non-profit organisation representing hospitals, medical schemes, medical scheme administrators, and medical and dental associations. MediKredit is responsible for the management and maintenance of the NAPPI Product File.
Abbott Laboratories is an American multinational medical devices and health care company with headquarters in Abbott Park, Illinois, United States. The company was founded by Chicago physician Wallace Calvin Abbott in 1888 to formulate known drugs; today, it sells medical devices, diagnostics, branded generic medicines and nutritional products. It split off its research-based pharmaceuticals business into AbbVie in 2013.

Current good manufacturing practices (cGMP) are those conforming to the guidelines recommended by relevant agencies. Those agencies control the authorization and licensing of the manufacture and sale of food and beverages, cosmetics, pharmaceutical products, dietary supplements, and medical devices. These guidelines provide minimum requirements that a manufacturer must meet to assure that their products are consistently high in quality, from batch to batch, for their intended use. The rules that govern each industry may differ significantly; however, the main purpose of GMP is always to prevent harm from occurring to the end user. Additional tenets include ensuring the end product is free from contamination, that it is consistent in its manufacture, that its manufacture has been well documented, that personnel are well trained, and that the product has been checked for quality more than just at the end phase. GMP is typically ensured through the effective use of a quality management system (QMS).
The Pharmaceutical Benefits Scheme (PBS) is a program of the Australian Government that subsidises prescription medication for Australian citizens and permanent residents, as well as international visitors covered by a reciprocal health care agreement. The PBS is separate to the Medicare Benefits Schedule, a list of health care services that can be claimed under Medicare, Australia's universal health care insurance scheme.
Pharmaceutical marketing is a branch of marketing science and practice focused on the communication, differential positioning and commercialization of pharmaceutical products, like specialist drugs, biotech drugs and over-the-counter drugs. By extension, this definition is sometimes also used for marketing practices applied to nutraceuticals and medical devices.
Pharmacovigilance, also known as drug safety, is the pharmaceutical science relating to the "collection, detection, assessment, monitoring, and prevention" of adverse effects with pharmaceutical products. The etymological roots for the word "pharmacovigilance" are: pharmakon and vigilare. As such, pharmacovigilance heavily focuses on adverse drug reactions (ADR), which are defined as any response to a drug which is noxious and unintended, including lack of efficacy. Medication errors such as overdose, and misuse and abuse of a drug as well as drug exposure during pregnancy and breastfeeding, are also of interest, even without an adverse event, because they may result in an adverse drug reaction.
Pharmaceutical codes are used in medical classification to uniquely identify medication. They may uniquely identify an active ingredient, drug system in general, or a specific pharmaceutical product from a specific manufacturer.

Ritanserin, also known by its developmental code name R-55667, is a serotonin antagonist medication described as an anxiolytic, antidepressant, antiparkinsonian agent, and antihypertensive agent. It was chiefly investigated as a drug to treat insomnia, especially to enhance sleep quality by significantly increasing slow wave sleep by virtue of potent and concomitant 5HT2a and 5HT2c antagonism
Title 21 is the portion of the Code of Federal Regulations that governs food and drugs within the United States for the Food and Drug Administration (FDA), the Drug Enforcement Administration (DEA), and the Office of National Drug Control Policy (ONDCP).
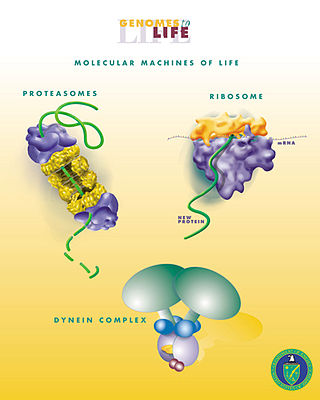
Biological engineering or bioengineering is the application of principles of biology and the tools of engineering to create usable, tangible, economically viable products. Biological engineering employs knowledge and expertise from a number of pure and applied sciences, such as mass and heat transfer, kinetics, biocatalysts, biomechanics, bioinformatics, separation and purification processes, bioreactor design, surface science, fluid mechanics, thermodynamics, and polymer science. It is used in the design of medical devices, diagnostic equipment, biocompatible materials, renewable energy, ecological engineering, agricultural engineering, process engineering and catalysis, and other areas that improve the living standards of societies.

The Association of the British Pharmaceutical Industry (ABPI) is the trade association for over 120 companies in the UK producing prescription medicines for humans, founded in 1891. It is the British equivalent of America's PhRMA; however, the member companies research, develop, manufacture and supply medicines prescribed for the National Health Service.
Pharmaceutical policy is a branch of health policy that deals with the development, provision and use of medications within a health care system. It embraces drugs, biologics, vaccines and natural health products.

Healthcare in Singapore is under the purview of the Ministry of Health of the Government of Singapore. It mainly consists of a government-run publicly funded universal healthcare system as well as a significant private healthcare sector. Financing of healthcare costs is done through a mixture of direct government subsidies, compulsory comprehensive savings, national healthcare insurance, and cost-sharing.
The certificate of pharmaceutical product is a certificate issued in the format recommended by the World Health Organization (WHO), which establishes the status of the pharmaceutical product and of the applicant for this certificate in the exporting country; it is often mentioned in conjunction with the electronic Common Technical Document (eCTD). A CPP is issued for a single product, because manufacturing arrangements and approved information for different pharmaceutical forms and strengths can vary. The CPP is mentioned in World Trade Organization documents, although the tightly regulated products are subject to bilateral trade agreements or regional trade agreements. The International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) has instituted standards for this purpose but it is unclear how the ex-ICH countries operate their health regulators.
The Queen's Award for Enterprise: Innovation (Technology) (2007) was awarded on 21 April 2007, by Queen Elizabeth II.
Adcock Ingram is a South African pharmaceutical manufacturer listed on the Johannesburg Stock Exchange. Adcock Ingram manufactures and markets healthcare products to both the private and public sectors.
Nappi may refer to:
This article summarizes healthcare in California.
Cannabis in Lesotho is illegal for any use, but largely tolerated. Cannabis is widely produced in the country, being the nation's most significant cash-crop. In the 2000s it was estimated that 70% of the cannabis in South Africa originated in Lesotho. In 2017 Lesotho became the first African nation to grant a license for the cultivation of medical cannabis.
The distribution of medications has special drug safety and security considerations. Some drugs require cold chain management in their distribution.