
Secretin is a hormone that regulates water homeostasis throughout the body and influences the environment of the duodenum by regulating secretions in the stomach, pancreas, and liver. It is a peptide hormone produced in the S cells of the duodenum, which are located in the intestinal glands. In humans, the secretin peptide is encoded by the SCT gene.

Somatostatin, also known as growth hormone-inhibiting hormone (GHIH) or by several other names, is a peptide hormone that regulates the endocrine system and affects neurotransmission and cell proliferation via interaction with G protein-coupled somatostatin receptors and inhibition of the release of numerous secondary hormones. Somatostatin inhibits insulin and glucagon secretion.

Parathyroid hormone-related protein (PTHrP) is a proteinaceous hormone and a member of the parathyroid hormone family secreted by mesenchymal stem cells. It is occasionally secreted by cancer cells. However, it also has normal functions in bone, teeth, vascular tissues and other tissues.

The glucocorticoid receptor also known as NR3C1 is the receptor to which cortisol and other glucocorticoids bind.

Relaxin is a protein hormone of about 6000 Da, first described in 1926 by Frederick Hisaw.

Agouti-signaling protein is a protein that in humans is encoded by the ASIP gene. It is responsible for the distribution of melanin pigment in mammals. Agouti interacts with the melanocortin 1 receptor to determine whether the melanocyte produces phaeomelanin, or eumelanin. This interaction is responsible for making distinct light and dark bands in the hairs of animals such as the agouti, which the gene is named after. In other species such as horses, agouti signalling is responsible for determining which parts of the body will be red or black. Mice with wildtype agouti will be grey-brown, with each hair being partly yellow and partly black. Loss of function mutations in mice and other species cause black fur coloration, while mutations causing expression throughout the whole body in mice cause yellow fur and obesity.

The adrenocorticotropic hormone receptor or ACTH receptor also known as the melanocortin receptor 2 or MC2 receptor is a type of melanocortin receptor (type 2) which is specific for ACTH. A G protein–coupled receptor located on the external cell plasma membrane, it is coupled to Gαs and upregulates levels of cAMP by activating adenylyl cyclase. The ACTH receptor plays a role in immune function and glucose metabolism.

The glucagon-like peptide-1 receptor (GLP1R) is a G protein-coupled receptor (GPCR) found on beta cells of the pancreas and on neurons of the brain. It is involved in the control of blood sugar level by enhancing insulin secretion. In humans it is synthesised by the gene GLP1R, which is present on chromosome 6. It is a member of the glucagon receptor family of GPCRs. GLP1R is composed of two domains, one extracellular (ECD) that binds the C-terminal helix of GLP-1, and one transmembrane (TMD) domain that binds the N-terminal region of GLP-1. In the TMD domain there is a fulcrum of polar residues that regulates the biased signaling of the receptor while the transmembrane helical boundaries and extracellular surface are a trigger for biased agonism.

Structures of GIPR human variant Sun, B., Kobilka, B.K., Sloop, K.W., Feng, D., Kobilka, T.S.
The relaxin receptors are a subclass of four closely related G protein-coupled receptors (GPCR) that bind relaxin peptide hormones.

Melanocortin 4 receptor (MC4R) is a melanocortin receptor that in humans is encoded by the MC4R gene. It encodes the MC4R protein, a G protein-coupled receptor (GPCR) that binds α-melanocyte stimulating hormone (α-MSH). In mouse models, MC4 receptors have been found to be involved in feeding behaviour, the regulation of metabolism, sexual behaviour, and male erectile function.

Relaxin/insulin-like family peptide receptor 3, also known as RXFP3, is a human G-protein coupled receptor.

Relaxin/insulin-like family peptide receptor 4, also known as RXFP4, is a human G-protein coupled receptor.

Relaxin/insulin-like family peptide receptor 2, also known as RXFP2, is a human G-protein coupled receptor.

Insulin-like 3 is a protein that in humans is encoded by the INSL3 gene.

Progesterone receptor membrane component 1 is a protein which co-purifies with progesterone binding proteins in the liver and ovary. In humans, the PGRMC1 protein is encoded by the PGRMC1 gene.

Ibutamoren is a potent, long-acting, orally-active, selective, and non-peptide agonist of the ghrelin receptor and a growth hormone secretagogue, mimicking the growth hormone (GH)-stimulating action of the endogenous hormone ghrelin. It has been shown to increase the secretion of several hormones including GH and insulin-like growth factor 1 (IGF-1) and produces sustained increases in the plasma levels of these hormones without affecting cortisol levels.

Relaxin-3 is a neuropeptide that was discovered in 2001, and which is highly conserved in species ranging from flies, fish, rodents and humans. Relaxin-3 is a member and ancestral gene of the relaxin family of peptides, which includes the namesake hormone relaxin which mediates peripheral actions during pregnancy and which was found to relax the pelvic ligament in guinea pigs almost a century ago. The cognate receptor for relaxin-3 is the G-protein coupled receptor RXFP3, however relaxin-3 is pharmacologically able to also cross react with RXFP1 and RXFP3.

Relaxin family peptide hormones in humans are represented by seven members: three relaxin-like (RLN) and four insulin-like (INSL) peptides: RLN1, RLN2, RNL3, INSL3, INSL4, INSL5, INSL6. This subdivision into two classes is based primarily on early findings, and does not reflect the evolutionary origins or physiological differences between peptides. For example, it is known that the genes coding for RLN3 and INSL5 arose from one ancestral gene, and INSL3 shares origin with RLN2 and its multiple duplicates: RLN1, INSL4, INSL6.
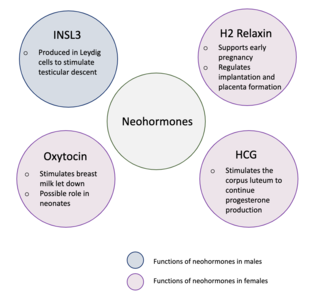
Neohormones are a group of recently evolved hormones primarily associated to the success of mammalian development. These hormones are specific to mammals and are not found in other vertebrates—this is because neohormones are evolved to enhance specific mammalian functions. In males, neohormones play important roles in regulating testicular descent and preparing the sperm for internal fertilisation. In females, neohormones are essential for regulating early pregnancy, mammary gland development lactation, and viviparity. Neohormones superimpose their actions on the hypothalamic-pituitary-gonadal axis and are not associated with other core bodily functions.























