
The boiling point of a substance is the temperature at which the vapor pressure of a liquid equals the pressure surrounding the liquid and the liquid changes into a vapor.
Saturation, saturated, unsaturation or unsaturated may refer to:

Humidity is the concentration of water vapour present in the air. Water vapour, the gaseous state of water, is generally invisible to the human eye. Humidity indicates the likelihood for precipitation, dew, or fog to be present. The amount of water vapour needed to achieve saturation increases as the temperature increases. As the temperature of a parcel of air decreases it will eventually reach the saturation point without adding or losing water mass. The amount of water vapour contained within a parcel of air can vary significantly. For example, a parcel of air near saturation may contain 28 grams of water per cubic metre of air at 30 °C, but only 8 grams of water per cubic metre of air at 8 °C.

Dew is water in the form of droplets that appears on thin, exposed objects in the morning or evening due to condensation.

The dew point is the temperature to which air must be cooled to become saturated with water vapor. When further cooled, the airborne water vapour will condense to form liquid water (dew). When air cools to its dew point through contact with a surface that is colder than the air, water will condense on the surface. When the temperature is below the freezing point of water, the dew point is called the frost point, as frost is formed rather than dew. The measurement of the dew point is related to humidity. A higher dew point means there is more moisture in the air.

Water vapor, water vapour or aqueous vapor is the gaseous phase of water. It is one state of water within the hydrosphere. Water vapor can be produced from the evaporation or boiling of liquid water or from the sublimation of ice. Water vapor is transparent, like most constituents of the atmosphere. Under typical atmospheric conditions, water vapor is continuously generated by evaporation and removed by condensation. It is less dense than most of the other constituents of air and triggers convection currents that can lead to clouds.

Relative humidity (RH) is the ratio of the partial pressure of water vapor to the equilibrium vapor pressure of water at a given temperature. Relative humidity depends on temperature and the pressure of the system of interest. The same amount of water vapor results in higher relative humidity in cool air than warm air. A related parameter is the dew point.
The lapse rate is the rate at which an atmospheric variable, normally temperature in Earth's atmosphere, falls with altitude. Lapse rate arises from the word lapse, in the sense of a gradual fall.

A hygrometer is an instrument used to measure the amount of humidity and water vapour in the atmosphere, in soil, or in confined spaces. Humidity measurement instruments usually rely on measurements of some other quantity such as temperature, pressure, mass, a mechanical or electrical change in a substance as moisture is absorbed. By calibration and calculation, these measured quantities can lead to a measurement of humidity. Modern electronic devices use temperature of condensation, or changes in electrical capacitance or resistance to measure humidity differences. The first crude hygrometer was invented by the Italian Renaissance polymath Leonardo da Vinci in 1480 and a more modern version was created by Swiss polymath Johann Heinrich Lambert in 1755. Later, in the year 1783, Swiss physicist and Geologist Horace Bénédict de Saussure invented the first hygrometer using human hair to measure humidity.

Colorfulness, chroma and saturation are attributes of perceived color relating to chromatic intensity. As defined formally by the International Commission on Illumination (CIE) they respectively describe three different aspects of chromatic intensity, but the terms are often used loosely and interchangeably in contexts where these aspects are not clearly distinguished. The precise meanings of the terms vary by what other functions they are dependent on.

Psychrometrics, psychrometry, and hygrometry are names for the field of engineering concerned with the physical and thermodynamic properties of gas-vapor mixtures. The term comes from the Greek psuchron (ψυχρόν) meaning "cold" and metron (μέτρον) meaning "means of measurement".

The wet-bulb temperature (WBT) is the temperature read by a thermometer covered in water-soaked cloth over which air is passed. At 100% relative humidity, the wet-bulb temperature is equal to the air temperature and it is lower at lower humidity. It is defined as the temperature of a parcel of air cooled to saturation by the evaporation of water into it, with the latent heat supplied by the parcel. A wet-bulb thermometer indicates a temperature close to the true (thermodynamic) wet-bulb temperature. The wet-bulb temperature is the lowest temperature that can be reached under current ambient conditions by the evaporation of water only.

An emagram is one of four thermodynamic diagrams used to display temperature lapse rate and moisture content profiles in the atmosphere. The emagram has axes of temperature (T) and pressure (p). In the emagram, the dry adiabats make an angle of about 45 degrees with the isobars, isotherms are vertical and isopleths of saturation mixing ratio are almost straight and vertical.

Thermodynamic diagrams are diagrams used to represent the thermodynamic states of a material and the consequences of manipulating this material. For instance, a temperature–entropy diagram may be used to demonstrate the behavior of a fluid as it is changed by a compressor.
Penitentes, or nieves penitentes, are snow formations found at high altitudes. They take the form of elongated, thin blades of hardened snow or ice, closely spaced and pointing towards the general direction of the sun.
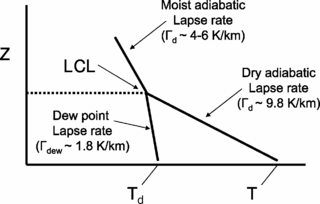
The lifted condensation level or lifting condensation level (LCL) is formally defined as the height at which the relative humidity (RH) of an air parcel will reach 100% with respect to liquid water when it is cooled by dry adiabatic lifting. The RH of air increases when it is cooled, since the amount of water vapor in the air remains constant, while the saturation vapor pressure decreases almost exponentially with decreasing temperature. If the air parcel is lifting further beyond the LCL, water vapor in the air parcel will begin condensing, forming cloud droplets. The LCL is a good approximation of the height of the cloud base which will be observed on days when air is lifted mechanically from the surface to the cloud base.
The vapour pressure of water is the pressure at which water vapour is in thermodynamic equilibrium with its condensed state. At higher pressures water would condense. The water vapour pressure is the partial pressure of water vapour in any gas mixture in equilibrium with solid or liquid water. As for other substances, water vapour pressure is a function of temperature and can be determined with the Clausius–Clapeyron relation.
The hydrocarbon dew point is the temperature at which the hydrocarbon components of any hydrocarbon-rich gas mixture, such as natural gas, will start to condense out of the gaseous phase. It is often also referred to as the HDP or the HCDP. The maximum temperature at which such condensation takes place is called the cricondentherm. The hydrocarbon dew point is a function of the gas composition as well as the pressure.

Surface weather observations are the fundamental data used for safety as well as climatological reasons to forecast weather and issue warnings worldwide. They can be taken manually, by a weather observer, by computer through the use of automated weather stations, or in a hybrid scheme using weather observers to augment the otherwise automated weather station. The ICAO defines the International Standard Atmosphere (ISA), which is the model of the standard variation of pressure, temperature, density, and viscosity with altitude in the Earth's atmosphere, and is used to reduce a station pressure to sea level pressure. Airport observations can be transmitted worldwide through the use of the METAR observing code. Personal weather stations taking automated observations can transmit their data to the United States mesonet through the Citizen Weather Observer Program (CWOP), the UK Met Office through their Weather Observations Website (WOW), or internationally through the Weather Underground Internet site. A thirty-year average of a location's weather observations is traditionally used to determine the station's climate. In the US a network of Cooperative Observers make a daily record of summary weather and sometimes water level information.
The Dew Tour is an extreme sports circuit organized by the Adventure Sports Group, a subsidiary of American Media, LLC.










