
In pharmacology and toxicology, a route of administration is the way by which a drug, fluid, poison, or other substance is taken into the body.

A transdermal patch is a medicated adhesive patch that is placed on the skin to deliver a specific dose of medication through the skin and into the bloodstream. An advantage of a transdermal drug delivery route over other types of medication delivery is that the patch provides a controlled release of the medication into the patient, usually through either a porous membrane covering a reservoir of medication or through body heat melting thin layers of medication embedded in the adhesive. The main disadvantage to transdermal delivery systems stems from the fact that the skin is a very effective barrier; as a result, only medications whose molecules are small enough to penetrate the skin can be delivered by this method. The first commercially available prescription patch was approved by the U.S. Food and Drug Administration in December 1979. These patches administered scopolamine for motion sickness.
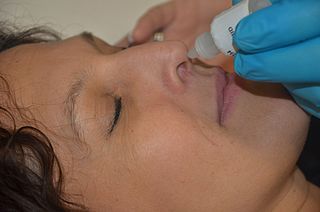
A topical medication is a medication that is applied to a particular place on or in the body. Most often topical medication means application to body surfaces such as the skin or mucous membranes to treat ailments via a large range of classes including creams, foams, gels, lotions, and ointments. Many topical medications are epicutaneous, meaning that they are applied directly to the skin. Topical medications may also be inhalational, such as asthma medications, or applied to the surface of tissues other than the skin, such as eye drops applied to the conjunctiva, or ear drops placed in the ear, or medications applied to the surface of a tooth. The word topical derives from Greek τοπικόςtopikos, "of a place".

An estrogen patch, or oestrogen patch, is a transdermal delivery system for estrogens such as estradiol and ethinylestradiol which can be used in menopausal hormone therapy, feminizing hormone therapy for transgender women, hormonal birth control, and other uses. Transdermal preparations of estrogen are metabolized differently than oral preparations. Transdermal estrogens avoid the first pass through the liver and thus potentially reduce the risk of blood clotting and stroke.
A metered-dose transdermal spray (MDTS) delivers a drug to the surface of the skin and is absorbed into the circulation on a sustained basis. It works in a similar manner to a transdermal patch or topical gel. The drug is delivered by a device placed gently against the skin and triggered, causing it to release a light spray containing a proprietary formulation of the drug that quickly dries on the skin to form an invisible drug depot. As it would be from a patch, the drug is then absorbed steadily for a predetermined amount of time.
Sonophoresis also known as phonophoresis, is a method that utilizes ultrasound to enhance the delivery of topical medications through the stratum corneum, to the epidermis and dermis. Sonophoresis allows for the enhancement of the permeability of the skin along with other modalities, such as iontophoresis, to deliver drugs with lesser side effects. Currently, sonophoresis is used widely in transdermal drug delivery, but has potential applications in other sectors of drug delivery, such as the delivery of drugs to the eye and brain.

Amcinonide is a topical glucocorticoid used to treat itching, redness and swelling associated with several dermatologic conditions such as atopic dermatitis and allergic contact dermatitis. Amcinonide can also be classified as a multi-functional small molecule corticosteroid, which has been approved by the FDA and is currently marketed as an ointment, lotion, or cream. It acts as both a transcription factor for responses to glucocorticoids and modulator for other transcription factors while also regulating phospholipase A2 activity.
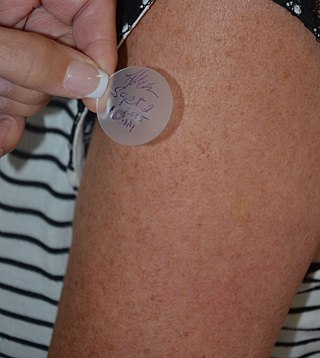
Transdermal is a route of administration wherein active ingredients are delivered across the skin for systemic distribution. Examples include transdermal patches used for medicine delivery. The drug is administered in the form of a patch or ointment that delivers the drug into the circulation for systemic effect.

Chlorphenesin carbamate is a centrally acting muscle relaxant used to treat muscle pain and spasms. Chlorphenesin carbamate is no longer used for this purpose in most developed nations due to the availability of much safer spasmolytics such as benzodiazepines.
Topical steroids are the topical forms of corticosteroids. Topical steroids are the most commonly prescribed topical medications for the treatment of rash and eczema. Topical steroids have anti-inflammatory properties and are classified based on their skin vasoconstrictive abilities. There are numerous topical steroid products. All the preparations in each class have the same anti-inflammatory properties but essentially differ in base and price.

The Hisamitsu Pharmaceutical Co., Inc., headquartered in Saga and Tokyo, is a Japanese multinational pharmaceutical corporation that develops and markets prescription and over-the-counter drug (OTC) products, especially external pain relieving products such as the transdermal patch. Hisamitsu has specialised in transdermal drug delivery system technology (TDDS) since the introduction of its original line of patches in 1903.

A transdermal analgesic or pain relief patch is a medicated adhesive patch used to relieve minor to severe pain. There are many types of analgesic patches based on the main ingredients in the patches. These include patches containing counterirritants, which are used to treat mild to moderate pain, and patches containing opioids such as buprenorphine and fentanyl, used to relieve moderate to severe pain. Fentanyl is often used for opioid-tolerant patients. Nitroglycerin, also known as glyceryl trinitrate (GTN), a medication used for heart failure, high blood pressure, anal fissures, painful periods, and to treat and prevent chest pain, can also be found in patches. Beyond these are patches that contain drugs such as diclofenac and lidocaine and various other drugs. The main purpose of transdermal analgesic patches are to administer drugs in a more viable way to patients, as opposed to oral consumption or intravenous administration such as an injection.
DDAIP is a pharmaceutical ingredient added to topical products to increase penetration through the skin. Chemically, DDAIP is an ester of N,N-dimethylalanine and dodecanol, although as of now the structural formula shows an ester with decanol (C10) instead. DDAIP is typically formulated as its hydrochloride salt (DDAIP.HCl). This salt is a white crystalline solid with a melting range of 88-93 °C and is an amphiphilic molecule with a pKa of 4.87 that is soluble in water up to about 40% w/v. DDAIP is proprietary to NexMed USA, a subsidiary of Apricus Biosciences.

Apricus Biosciences, Inc. is a San Diego based biopharmaceutical company advancing innovative medicines in urology and rheumatology.

Ethosomes are phospholipid nanovesicles used for dermal and transdermal delivery of molecules. Ethosomes were developed by Touitou et al.,1997, as additional novel lipid carriers composed of ethanol, phospholipids, and water. They are reported to improve the skin delivery of various drugs. Ethanol is an efficient permeation enhancer that is believed to act by affecting the intercellular region of the stratum corneum. Ethosomes are soft malleable vesicles composed mainly of phospholipids, ethanol, and water. These soft vesicles represent novel vesicles carriers for enhanced delivery through the skin. The size of the ethosomes vesicles can be modulated from tens of nanometers to microns.

Topical cream formulation is an emulsion semisolid dosage form that is used for skin external application. Most of the topical cream formulations contain more than 20 per cent of water and volatiles and/or less than 50 per cent of hydrocarbons, waxes, or polyethylene glycols as the vehicle for external skin application. In a topical cream formulation, ingredients are dissolved or dispersed in either a water-in-oil (W/O) emulsion or an oil-in-water (O/W) emulsion. The topical cream formulation has a higher content of oily substance than gel, but a lower content of oily ingredient than ointment. Therefore, the viscosity of topical cream formulation lies between gel and ointment. The pharmacological effect of the topical cream formulation is confined to the skin surface or within the skin. Topical cream formulation penetrates through the skin by transcellular route, intercellular route, or trans-appendageal route. Topical cream formulation is used for a wide range of diseases and conditions, including atopic dermatitis (eczema), psoriasis, skin infection, acne, and wart. Excipients found in a topical cream formulation include thickeners, emulsifying agents, preservatives, antioxidants, and buffer agents. Steps required to manufacture a topical cream formulation include excipient dissolution, phase mixing, introduction of active substances, and homogenization of the product mixture.
Topical drug delivery (TDD) is a route of drug administration that allows the topical formulation to be delivered across the skin upon application, hence producing a localized effect to treat skin disorders like eczema. The formulation of topical drugs can be classified into corticosteroids, antibiotics, antiseptics, and anti-fungal. The mechanism of topical delivery includes the diffusion and metabolism of drugs in the skin. Historically, topical route was the first route of medication used to deliver drugs in humans in ancient Egyptian and Babylonian in 3000 BCE. In these ancient cities, topical medications like ointments and potions were used on the skin. The delivery of topical drugs needs to pass through multiple skin layers and undergo pharmacokinetics, hence factor like dermal diseases minimize the bioavailability of the topical drugs. The wide use of topical drugs leads to the advancement in topical drug delivery. These advancements are used to enhance the delivery of topical medications to the skin by using chemical and physical agents. For chemical agents, carriers like liposomes and nanotechnologies are used to enhance the absorption of topical drugs. On the other hand, physical agents, like micro-needles is other approach for enhancement ofabsorption. Besides using carriers, other factors such as pH, lipophilicity, and drug molecule size govern the effectiveness of topical formulation.
Penetration enhancers are chemical compounds that can facilitate the penetration of active pharmaceutical ingredients (API) into or through the poorly permeable biological membranes. These compounds are used in some pharmaceutical formulations to enhance the penetration of APIs in transdermal drug delivery and transmucosal drug delivery. They typically penetrate into the biological membranes and reversibly decrease their barrier properties.
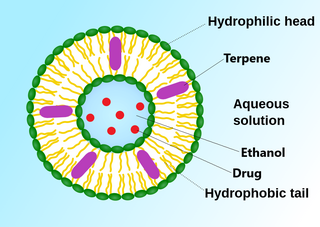
An invasome is a type of artificial vesicle nanocarrier that transport substances through the skin, the most superficial biological barrier. Vesicles are small particles surrounded by a lipid layer that can carry substances into and out of the cell. Artificial vesicles can be engineered to deliver drugs within the cell, with specific applications within transdermal drug delivery. However, the skin proves to be a barrier to effective penetration and delivery of drug therapies. Thus, invasomes are a new generation of vesicle with added structural components to assist with skin penetration.
Laser-assisted drug delivery (LADD) is a drug delivery technique commonly used in the dermatology field that involves lasers. As skin acts as a protective barrier to the environment, the absorption of topical products through the epidermis is limited; thus, different drug delivery modalities have been employed to improve the efficacy of these treatments. The use of lasers in LADD has been shown to enhance the penetration of drugs transdermal, leading to a higher absorption rate, limited systemic effects, and reduced duration of treatment. Although this technique has evolved in the past decade due to its efficacy through scientific research and clinical practice, there remain some limitations regarding the safety aspect that needs to be taken into consideration.












