
A colloid is a mixture in which one substance consisting of microscopically dispersed insoluble particles is suspended throughout another substance. Some definitions specify that the particles must be dispersed in a liquid, while others extend the definition to include substances like aerosols and gels. The term colloidal suspension refers unambiguously to the overall mixture. A colloid has a dispersed phase and a continuous phase. The dispersed phase particles have a diameter of approximately 1 nanometre to 1 micrometre.

An emulsion is a mixture of two or more liquids that are normally immiscible owing to liquid-liquid phase separation. Emulsions are part of a more general class of two-phase systems of matter called colloids. Although the terms colloid and emulsion are sometimes used interchangeably, emulsion should be used when both phases, dispersed and continuous, are liquids. In an emulsion, one liquid is dispersed in the other. Examples of emulsions include vinaigrettes, homogenized milk, liquid biomolecular condensates, and some cutting fluids for metal working.
In chemistry, a mixture is a material made up of two or more different chemical substances which are not chemically bonded. A mixture is the physical combination of two or more substances in which the identities are retained and are mixed in the form of solutions, suspensions and colloids.
In polymer chemistry, emulsion polymerization is a type of radical polymerization that usually starts with an emulsion incorporating water, monomers, and surfactants. The most common type of emulsion polymerization is an oil-in-water emulsion, in which droplets of monomer are emulsified in a continuous phase of water. Water-soluble polymers, such as certain polyvinyl alcohols or hydroxyethyl celluloses, can also be used to act as emulsifiers/stabilizers. The name "emulsion polymerization" is a misnomer that arises from a historical misconception. Rather than occurring in emulsion droplets, polymerization takes place in the latex/colloid particles that form spontaneously in the first few minutes of the process. These latex particles are typically 100 nm in size, and are made of many individual polymer chains. The particles are prevented from coagulating with each other because each particle is surrounded by the surfactant ('soap'); the charge on the surfactant repels other particles electrostatically. When water-soluble polymers are used as stabilizers instead of soap, the repulsion between particles arises because these water-soluble polymers form a 'hairy layer' around a particle that repels other particles, because pushing particles together would involve compressing these chains.

In chemistry, a suspension is a heterogeneous mixture of a fluid that contains solid particles sufficiently large for sedimentation. The particles may be visible to the naked eye, usually must be larger than one micrometer, and will eventually settle, although the mixture is only classified as a suspension when and while the particles have not settled out.

In industrial process engineering, mixing is a unit operation that involves manipulation of a heterogeneous physical system with the intent to make it more homogeneous. Familiar examples include pumping of the water in a swimming pool to homogenize the water temperature, and the stirring of pancake batter to eliminate lumps (deagglomeration).
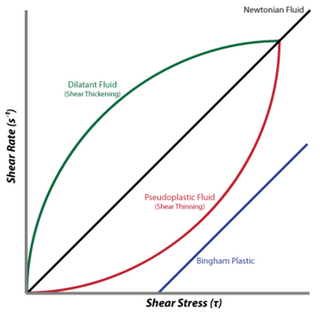
A dilatant material is one in which viscosity increases with the rate of shear strain. Such a shear thickening fluid, also known by the initialism STF, is an example of a non-Newtonian fluid. This behaviour is usually not observed in pure materials, but can occur in suspensions.
A sol is a colloidal suspension made out of tiny solid particles in a continuous liquid medium. Sols are stable and exhibit the Tyndall effect, which is the scattering of light by the particles in the colloid. Examples include amongst others blood, pigmented ink, cell fluids, paint, antacids and mud.
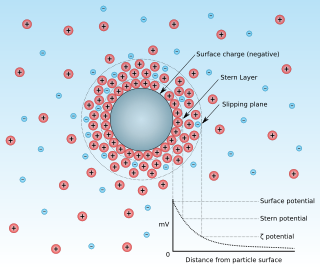
Zeta potential is the electrical potential at the slipping plane. This plane is the interface which separates mobile fluid from fluid that remains attached to the surface.

A nanoparticle or ultrafine particle is a particle of matter 1 to 100 nanometres (nm) in diameter. The term is sometimes used for larger particles, up to 500 nm, or fibers and tubes that are less than 100 nm in only two directions. At the lowest range, metal particles smaller than 1 nm are usually called atom clusters instead.

In colloidal chemistry, flocculation is a process by which colloidal particles come out of suspension to sediment in the form of floc or flake, either spontaneously or due to the addition of a clarifying agent. The action differs from precipitation in that, prior to flocculation, colloids are merely suspended, under the form of a stable dispersion and are not truly dissolved in solution.

Ostwald ripening is a phenomenon observed in solid solutions and liquid sols that involves the change of an inhomogeneous structure over time, in that small crystals or sol particles first dissolve and then redeposit onto larger crystals or sol particles.

Particle agglomeration refers to the formation of assemblages in a suspension and represents a mechanism leading to the functional destabilization of colloidal systems. During this process, particles dispersed in the liquid phase stick to each other, and spontaneously form irregular particle assemblages, flocs, or agglomerates. This phenomenon is also referred to as coagulation or flocculation and such a suspension is also called unstable. Particle agglomeration can be induced by adding salts or other chemicals referred to as coagulant or flocculant.

In polymer chemistry, suspension polymerization is a heterogeneous radical polymerization process that uses mechanical agitation to mix a monomer or mixture of monomers in a liquid phase, such as water, while the monomers polymerize, forming spheres of polymer. The monomer droplets are suspended in the liquid phase. The individual monomer droplets can be considered as undergoing bulk polymerization. The liquid phase outside these droplets help in better conduction of heat and thus tempering the increase in temperature.
Sedimentation potential occurs when dispersed particles move under the influence of either gravity or centrifugation or electricity in a medium. This motion disrupts the equilibrium symmetry of the particle's double layer. While the particle moves, the ions in the electric double layer lag behind due to the liquid flow. This causes a slight displacement between the surface charge and the electric charge of the diffuse layer. As a result, the moving particle creates a dipole moment. The sum of all of the dipoles generates an electric field which is called sedimentation potential. It can be measured with an open electrical circuit, which is also called sedimentation current.
A dispersant or a dispersing agent is a substance, typically a surfactant, that is added to a suspension of solid or liquid particles in a liquid to improve the separation of the particles and to prevent their settling or clumping.

A colloidal crystal is an ordered array of colloidal particles and fine grained materials analogous to a standard crystal whose repeating subunits are atoms or molecules. A natural example of this phenomenon can be found in the gem opal, where spheres of silica assume a close-packed locally periodic structure under moderate compression. Bulk properties of a colloidal crystal depend on composition, particle size, packing arrangement, and degree of regularity. Applications include photonics, materials processing, and the study of self-assembly and phase transitions.
Macroemulsions are dispersed liquid-liquid, thermodynamically unstable systems with particle sizes ranging from 1 to 100 μm, which, most often, do not form spontaneously. Macroemulsions scatter light effectively and therefore appear milky, because their droplets are greater than a wavelength of light. They are part of a larger family of emulsions along with miniemulsions. As with all emulsions, one phase serves as the dispersing agent. It is often called the continuous or outer phase. The remaining phase(s) are disperse or inner phase(s), because the liquid droplets are finely distributed amongst the larger continuous phase droplets. This type of emulsion is thermodynamically unstable, but can be stabilized for a period of time with applications of kinetic energy. Surfactants are used to reduce the interfacial tension between the two phases, and induce macroemulsion stability for a useful amount of time. Emulsions can be stabilized otherwise with polymers, solid particles or proteins.
Dispersion Technology Inc is a scientific instrument manufacturer located in Bedford Hills, New York. It was founded in 1996 by Philip Goetz and Dr. Andrei Dukhin. The company develops and sells analytical instruments intended for characterizing concentrated dispersions and emulsions, complying with the International Standards for acoustic particle sizing ISO 20998 and electroacoustic zeta potential measurement ISO 13099.
In polymer science, dispersion polymerization is a heterogeneous polymerization process carried out in the presence of a polymeric stabilizer in the reaction medium. Dispersion polymerization is a type of precipitation polymerization, meaning the solvent selected as the reaction medium is a good solvent for the monomer and the initiator, but is a non-solvent for the polymer. As the polymerization reaction proceeds, particles of polymer form, creating a non-homogeneous solution. In dispersion polymerization these particles are the locus of polymerization, with monomer being added to the particle throughout the reaction. In this sense, the mechanism for polymer formation and growth has features similar to that of emulsion polymerization. With typical precipitation polymerization, the continuous phase is the main locus of polymerization, which is the main difference between precipitation and dispersion.











