
Ductility is a mechanical property commonly described as a material's amenability to drawing. In materials science, ductility is defined by the degree to which a material can sustain plastic deformation under tensile stress before failure. Ductility is an important consideration in engineering and manufacturing. It defines a material's suitability for certain manufacturing operations and its capacity to absorb mechanical overload. Some metals that are generally described as ductile include gold and copper, while platinum is the most ductile of all metals in pure form. However, not all metals experience ductile failure as some can be characterized with brittle failure like cast iron. Polymers generally can be viewed as ductile materials as they typically allow for plastic deformation.

Polymer degradation is the reduction in the physical properties of a polymer, such as strength, caused by changes in its chemical composition. Polymers and particularly plastics are subject to degradation at all stages of their product life cycle, including during their initial processing, use, disposal into the environment and recycling. The rate of this degradation varies significantly; biodegradation can take decades, whereas some industrial processes can completely decompose a polymer in hours.

A material is brittle if, when subjected to stress, it fractures with little elastic deformation and without significant plastic deformation. Brittle materials absorb relatively little energy prior to fracture, even those of high strength. Breaking is often accompanied by a sharp snapping sound.

Hydrogen embrittlement (HE), also known as hydrogen-assisted cracking or hydrogen-induced cracking (HIC), is a reduction in the ductility of a metal due to absorbed hydrogen. Hydrogen atoms are small and can permeate solid metals. Once absorbed, hydrogen lowers the stress required for cracks in the metal to initiate and propagate, resulting in embrittlement. Hydrogen embrittlement occurs most notably in steels, as well as in iron, nickel, titanium, cobalt, and their alloys. Copper, aluminium, and stainless steels are less susceptible to hydrogen embrittlement.

A reactor pressure vessel (RPV) in a nuclear power plant is the pressure vessel containing the nuclear reactor coolant, core shroud, and the reactor core.

Stress corrosion cracking (SCC) is the growth of crack formation in a corrosive environment. It can lead to unexpected and sudden failure of normally ductile metal alloys subjected to a tensile stress, especially at elevated temperature. SCC is highly chemically specific in that certain alloys are likely to undergo SCC only when exposed to a small number of chemical environments. The chemical environment that causes SCC for a given alloy is often one which is only mildly corrosive to the metal. Hence, metal parts with severe SCC can appear bright and shiny, while being filled with microscopic cracks. This factor makes it common for SCC to go undetected prior to failure. SCC often progresses rapidly, and is more common among alloys than pure metals. The specific environment is of crucial importance, and only very small concentrations of certain highly active chemicals are needed to produce catastrophic cracking, often leading to devastating and unexpected failure.
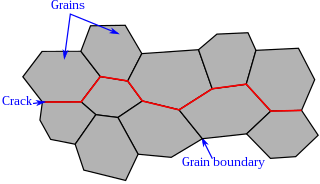
Intergranular fracture, intergranular cracking or intergranular embrittlement occurs when a crack propagates along the grain boundaries of a material, usually when these grain boundaries are weakened. The more commonly seen transgranular fracture, occurs when the crack grows through the material grains. As an analogy, in a wall of bricks, intergranular fracture would correspond to a fracture that takes place in the mortar that keeps the bricks together.
In metallurgy and materials science, annealing is a heat treatment that alters the physical and sometimes chemical properties of a material to increase its ductility and reduce its hardness, making it more workable. It involves heating a material above its recrystallization temperature, maintaining a suitable temperature for an appropriate amount of time and then cooling.
Liquid metal embrittlement is a phenomenon of practical importance, where certain ductile metals experience drastic loss in tensile ductility or undergo brittle fracture when exposed to specific liquid metals. Generally, tensile stress, either externally applied or internally present, is needed to induce embrittlement. Exceptions to this rule have been observed, as in the case of aluminium in the presence of liquid gallium. This phenomenon has been studied since the beginning of the 20th century. Many of its phenomenological characteristics are known and several mechanisms have been proposed to explain it. The practical significance of liquid metal embrittlement is revealed by the observation that several steels experience ductility losses and cracking during hot-dip galvanizing or during subsequent fabrication. Cracking can occur catastrophically and very high crack growth rates have been measured.
Radiation damage is the effect of ionizing radiation on physical objects including non-living structural materials. It can be either detrimental or beneficial for materials.
In geology, a deformation mechanism is a process occurring at a microscopic scale that is responsible for changes in a material's internal structure, shape and volume. The process involves planar discontinuity and/or displacement of atoms from their original position within a crystal lattice structure. These small changes are preserved in various microstructures of materials such as rocks, metals and plastics, and can be studied in depth using optical or digital microscopy.
Methods have been devised to modify the yield strength, ductility, and toughness of both crystalline and amorphous materials. These strengthening mechanisms give engineers the ability to tailor the mechanical properties of materials to suit a variety of different applications. For example, the favorable properties of steel result from interstitial incorporation of carbon into the iron lattice. Brass, a binary alloy of copper and zinc, has superior mechanical properties compared to its constituent metals due to solution strengthening. Work hardening has also been used for centuries by blacksmiths to introduce dislocations into materials, increasing their yield strengths.
Forensic polymer engineering is the study of failure in polymeric products. The topic includes the fracture of plastic products, or any other reason why such a product fails in service, or fails to meet its specification. The subject focuses on the material evidence from crime or accident scenes, seeking defects in those materials that might explain why an accident occurred, or the source of a specific material to identify a criminal. Many analytical methods used for polymer identification may be used in investigations, the exact set being determined by the nature of the polymer in question, be it thermoset, thermoplastic, elastomeric or composite in nature.

Environmental Stress Cracking (ESC) is one of the most common causes of unexpected brittle failure of thermoplastic polymers known at present. According to ASTM D883, stress cracking is defined as "an external or internal crack in a plastic caused by tensile stresses less than its short-term mechanical strength". This type of cracking typically involves brittle cracking, with little or no ductile drawing of the material from its adjacent failure surfaces. Environmental stress cracking may account for around 15-30% of all plastic component failures in service. This behavior is especially prevalent in glassy, amorphous thermoplastics. Amorphous polymers exhibit ESC because of their loose structure which makes it easier for the fluid to permeate into the polymer. Amorphous polymers are more prone to ESC at temperature higher than their glass transition temperature (Tg) due to the increased free volume. When Tg is approached, more fluid can permeate into the polymer chains.
In polymers, such as plastics, thermal degradation refers to a type of polymer degradation where damaging chemical changes take place at elevated temperatures, without the simultaneous involvement of other compounds such as oxygen. Simply put, even in the absence of air, polymers will begin to degrade if heated high enough. It is distinct from thermal-oxidation, which can usually take place at less elevated temperatures.

In polymer chemistry photo-oxidation is the degradation of a polymer surface due to the combined action of light and oxygen. It is the most significant factor in the weathering of plastics. Photo-oxidation causes the polymer chains to break, resulting in the material becoming increasingly brittle. This leads to mechanical failure and, at an advanced stage, the formation of microplastics. In textiles the process is called phototendering.
Low hydrogen annealing, commonly known as "baking" is a heat treatment in metallurgy for the reduction or elimination of hydrogen in a material to prevent hydrogen embrittlement. Hydrogen embrittlement is the hydrogen-induced cracking of metals, particularly steel which results in degraded mechanical properties such as plasticity, ductility and fracture toughness at low temperature. Low hydrogen annealing is called a de-embrittlement process. Low hydrogen annealing is an effective method compared to alternatives such as electroplating the material with zinc to provide a barrier for hydrogen ingress which results in coating defects.
Polymer fracture is the study of the fracture surface of an already failed material to determine the method of crack formation and extension in polymers both fiber reinforced and otherwise. Failure in polymer components can occur at relatively low stress levels, far below the tensile strength because of four major reasons: long term stress or creep rupture, cyclic stresses or fatigue, the presence of structural flaws and stress-cracking agents. Formations of submicroscopic cracks in polymers under load have been studied by x ray scattering techniques and the main regularities of crack formation under different loading conditions have been analyzed. The low strength of polymers compared to theoretically predicted values are mainly due to the many microscopic imperfections found in the material. These defects namely dislocations, crystalline boundaries, amorphous interlayers and block structure can all lead to the non-uniform distribution of mechanical stress.
Metal-induced embrittlement (MIE) is the embrittlement caused by diffusion of metal, either solid or liquid, into the base material. Metal induced embrittlement occurs when metals are in contact with low-melting point metals while under tensile stress. The embrittler can be either solid (SMIE) or liquid. Under sufficient tensile stress, MIE failure occurs instantaneously at temperatures just above melting point. For temperatures below the melting temperature of the embrittler, solid-state diffusion is the main transport mechanism. This occurs in the following ways:













