
Alkaloids are a class of basic, naturally occurring organic compounds that contain at least one nitrogen atom. This group also includes some related compounds with neutral and even weakly acidic properties. Some synthetic compounds of similar structure may also be termed alkaloids. In addition to carbon, hydrogen and nitrogen, alkaloids may also contain oxygen or sulfur. More rarely still, they may contain elements such as phosphorus, chlorine, and bromine.

Cocaine is a tropane alkaloid that acts as a central nervous system (CNS) stimulant. As an extract, it is mainly used recreationally, and often illegally for its euphoric and rewarding effects. It is also used in medicine by Indigenous South Americans for various purposes and rarely, but more formally, as a local anaesthetic or diagnostic tool by medical practitioners in more developed countries. It is primarily obtained from the leaves of two Coca species native to South America: Erythroxylum coca and E. novogranatense. After extraction from the plant, and further processing into cocaine hydrochloride, the drug is administered by being either snorted, applied topically to the mouth, or dissolved and injected into a vein. It can also then be turned into free base form, in which it can be heated until sublimated and then the vapours can be inhaled.

Tobacco smoke is a sooty aerosol produced by the incomplete combustion of tobacco during the smoking of cigarettes and other tobacco products. Temperatures in burning cigarettes range from about 400 °C between puffs to about 900 °C during a puff. During the burning of the cigarette tobacco, thousands of chemical substances are generated by combustion, distillation, pyrolysis and pyrosynthesis. Tobacco smoke is used as a fumigant and inhalant.

Hydroxylamine is an inorganic compound with the chemical formula NH2OH. The compound is in a form of a white hygroscopic crystals. Hydroxylamine is almost always provided and used as an aqueous solution. It is consumed almost exclusively to produce Nylon-6. The oxidation of NH3 to hydroxylamine is a step in biological nitrification.
In chemistry, a hydrochloride is an acid salt resulting, or regarded as resulting, from the reaction of hydrochloric acid with an organic base. An alternative name is chlorhydrate, which comes from French. An archaic alternative name is muriate, derived from hydrochloric acid's ancient name: muriatic acid.
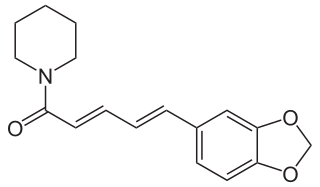
Piperine, possibly along with its isomer chavicine, is the compound responsible for the pungency of black pepper and long pepper. It has been used in some forms of traditional medicine.

Piperazine is an organic compound that consists of a six-membered ring containing two nitrogen atoms at opposite positions in the ring. Piperazine exists as small alkaline deliquescent crystals with a saline taste.
Triethylamine is the chemical compound with the formula N(CH2CH3)3, commonly abbreviated Et3N. It is also abbreviated TEA, yet this abbreviation must be used carefully to avoid confusion with triethanolamine or tetraethylammonium, for which TEA is also a common abbreviation. It is a colourless volatile liquid with a strong fishy odor reminiscent of ammonia. Like diisopropylethylamine (Hünig's base), triethylamine is commonly employed in organic synthesis, usually as a base.
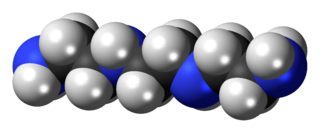
Triethylenetetramine (TETA and trien), also known as trientine (INN) when used medically, is an organic compound with the formula [CH2NHCH2CH2NH2]2. The pure freebase is a colorless oily liquid, but, like many amines, older samples assume a yellowish color due to impurities resulting from air-oxidation. It is soluble in polar solvents. The branched isomer tris(2-aminoethyl)amine and piperazine derivatives may also be present in commercial samples of TETA. The hydrochloride salts are used medically as a treatment for copper toxicity.

In chemistry, an amine oxide, also known as an amine N-oxide or simply N-oxide, is a chemical compound that has the chemical formula R3N+−O−. It contains a nitrogen-oxygen coordinate covalent bond with three additional hydrogen and/or substituent-groups attached to nitrogen. Sometimes it is written as R3N→O or, alternatively, as R3N=O.

A cigarette filter, also known as a filter tip, is a component of a cigarette, along with cigarette paper, capsules and adhesives. Filters were introduced in the early 1950s.
Coca paste is a crude extract of the coca leaf which contains 40% to 91% cocaine freebase along with companion coca alkaloids and varying quantities of benzoic acid, methanol, and kerosene. In South America, coca paste, also known as cocaine base and, therefore, often confused with cocaine sulfate in North America, is relatively inexpensive and is widely used by low-income populations. The coca paste is smoked in tobacco or cannabis cigarettes and use has become widespread in several Latin American countries. Traditionally, coca paste has been relatively abundant in South American countries such as Colombia where it is processed into cocaine hydrochloride for distribution to the rest of the world. The caustic reactions associated with the local application of coca paste prevents its use by oral, intranasal, mucosal, intramuscular, intravenous or subcutaneous routes. Coca paste can only be smoked when combined with a combustible material such as tobacco or cannabis.
Black cocaine is a mixture of regular cocaine base or cocaine hydrochloride with various other substances. These other substances are added
Acid–base extraction is a subclass of liquid–liquid extractions and involves the separation of chemical species from other acidic or basic compounds. It is typically performed during the work-up step following a chemical synthesis to purify crude compounds and results in the product being largely free of acidic or basic impurities. A separatory funnel is commonly used to perform an acid-base extraction.

Etaqualone is a quinazolinone-class GABAergic and is an analogue of methaqualone that was developed in the 1960s and marketed mainly in France and some other European countries. It has sedative, hypnotic, muscle relaxant and central nervous system depressant properties resulting from its agonist activity at the β-subtype of the GABAA receptor, and was used for the treatment of insomnia.

N-Hydroxysuccinimide (NHS) is an organic compound with the formula (CH2CO)2NOH. It is a white solid that is used as a reagent for preparing active esters in peptide synthesis. It can be synthesized by heating succinic anhydride with hydroxylamine or hydroxylamine hydrochloride.

Desomorphine is a semi-synthetic opioid commercialized by Roche, with powerful, fast-acting effects, such as sedation and analgesia. It was first discovered and patented by a German team working for Knoll in 1920 but was not generally recognized. It was later synthesized in 1932 by Lyndon Frederick Small. Small also successfully patented it in 1934 in the United States. Desomorphine was used in Switzerland under the brand name Permonid and was described as having a fast onset and a short duration of action, with relatively little nausea compared to equivalent doses of morphine. Dose for dose it is eight to ten times more potent than morphine.

Crack cocaine, commonly known simply as crack, and also known as rock, is a free base form of the stimulant cocaine that can be smoked. Crack offers a short, intense high to smokers. The Manual of Adolescent Substance Abuse Treatment calls it the most addictive form of cocaine.

Methoxyamine is the organic compound with the formula CH3ONH2. Also called O-methylhydroxylamine, it is a colourless volatile liquid that is soluble in polar organic solvent and in water. It is a derivative of hydroxylamine with the hydroxyl hydrogen replaced by a methyl group. Alternatively, it can be viewed as a derivative of methanol with the hydroxyl hydrogen replaced by an amino group. It is an isomer of N-methylhydroxylamine and aminomethanol. It decomposes in an exothermic reaction (-56 kJ/mol) to methane and azanone unless stored as a hydrochloride salt.

Dioscorine is an alkaloid toxin isolated from the tubers of tropical yam on several continents. It has been used as a monkey poison in some African countries, and as an arrow poison to aid in hunting in several parts of Asia. It was first isolated from Dioscorea hirsute by Boorsma in 1894 and obtained in a crystalline form by Schutte in 1897, and has since been found in other Dioscorea species. Dioscorine is a neurotoxin that acts by blocking the nicotinic acetylcholine receptor. Dioscorine is generally isolated in tandem with other alkaloids such as dioscin but is usually the most potent toxin in the mixture. It is a convulsant, producing symptoms similar to picrotoxin, with which it shares a similar mechanism of action.














