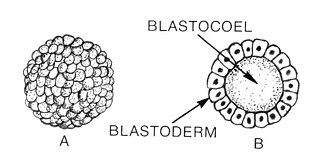
Blastulation is the stage in early animal embryonic development that produces the blastula. In mammalian development the blastula develops into the blastocyst with a differentiated inner cell mass and an outer trophectoderm. The blastula is a hollow sphere of cells known as blastomeres surrounding an inner fluid-filled cavity called the blastocoel. Embryonic development begins with a sperm fertilizing an egg cell to become a zygote, which undergoes many cleavages to develop into a ball of cells called a morula. Only when the blastocoel is formed does the early embryo become a blastula. The blastula precedes the formation of the gastrula in which the germ layers of the embryo form.

Drosophila embryogenesis, the process by which Drosophila embryos form, is a favorite model system for genetics and developmental biology. The study of its embryogenesis unlocked the century-long puzzle of how development was controlled, creating the field of evolutionary developmental biology. The small size, short generation time, and large brood size make it ideal for genetic studies. Transparent embryos facilitate developmental studies. Drosophila melanogaster was introduced into the field of genetic experiments by Thomas Hunt Morgan in 1909.
Segmentation in biology is the division of some animal and plant body plans into a linear series of repetitive segments that may or may not be interconnected to each other. This article focuses on the segmentation of animal body plans, specifically using the examples of the taxa Arthropoda, Chordata, and Annelida. These three groups form segments by using a "growth zone" to direct and define the segments. While all three have a generally segmented body plan and use a growth zone, they use different mechanisms for generating this patterning. Even within these groups, different organisms have different mechanisms for segmenting the body. Segmentation of the body plan is important for allowing free movement and development of certain body parts. It also allows for regeneration in specific individuals.

A morphogen is a substance whose non-uniform distribution governs the pattern of tissue development in the process of morphogenesis or pattern formation, one of the core processes of developmental biology, establishing positions of the various specialized cell types within a tissue. More specifically, a morphogen is a signaling molecule that acts directly on cells to produce specific cellular responses depending on its local concentration.
Hox genes, a subset of homeobox genes, are a group of related genes that specify regions of the body plan of an embryo along the head-tail axis of animals. Hox proteins encode and specify the characteristics of 'position', ensuring that the correct structures form in the correct places of the body. For example, Hox genes in insects specify which appendages form on a segment, and Hox genes in vertebrates specify the types and shape of vertebrae that will form. In segmented animals, Hox proteins thus confer segmental or positional identity, but do not form the actual segments themselves.

Ultrabithorax (Ubx) is a homeobox gene found in insects, and is used in the regulation of patterning in morphogenesis. There are many possible products of this gene, which function as transcription factors. Ubx is used in the specification of serially homologous structures, and is used at many levels of developmental hierarchies. In Drosophila melanogaster it is expressed in the third thoracic (T3) and first abdominal (A1) segments and represses wing formation. The Ubx gene regulates the decisions regarding the number of wings and legs the adult flies will have. The developmental role of the Ubx gene is determined by the splicing of its product, which takes place after translation of the gene. The specific splice factors of a particular cell allow the specific regulation of the developmental fate of that cell, by making different splice variants of transcription factors. In D. melanogaster, at least six different isoforms of Ubx exist.

Krüppel is a gap gene in Drosophila melanogaster, located on the 2R chromosome, which encodes a zinc finger C2H2 transcription factor. Gap genes work together to establish the anterior-posterior segment patterning of the insect through regulation of the transcription factor encoding pair rule genes. These genes in turn regulate segment polarity genes. Krüppel means "cripple" in German, named for the crippled appearance of mutant larvae, who have failed to develop proper thoracic and anterior segments in the abdominal region. Mutants can also have abdominal mirror duplications.
In the field of developmental biology, regional differentiation is the process by which different areas are identified in the development of the early embryo. The process by which the cells become specified differs between organisms.

The bicoid 3′-UTR regulatory element is an mRNA regulatory element that controls the gene expression of the bicoid protein in fruitfly Drosophila melanogaster.

A pair-rule gene is a type of gene involved in the development of the segmented embryos of insects. Pair-rule genes are expressed as a result of differing concentrations of gap gene proteins, which encode transcription factors controlling pair-rule gene expression. Pair-rule genes are defined by the effect of a mutation in that gene, which causes the loss of the normal developmental pattern in alternating segments.

engrailed is a homeodomain transcription factor involved in many aspects of multicellular development. First known for its role in arthropod embryological development, working in consort with the Hox genes, engrailed has been found to be important in other areas of development. It has been identified in many bilaterians, including the arthropods, vertebrates, echinoderms, molluscs, nematodes, brachiopods, and polychaetes. It acts as a "selector" gene, conferring a specific identity to defined areas of the body, and co-ordinating the expression of downstream genes.
Orthodenticle (otd) is a homeobox gene found in Drosophila that regulates the development of anterior patterning, with particular involvement in the central nervous system function and eye development. It is located on the X chromosome. The gene is an ortholog of the human OTX1/OTX2 gene.
Homeotic genes are genes which regulate the development of anatomical structures in various organisms such as echinoderms, insects, mammals, and plants. Homeotic genes often encode transcription factor proteins, and these proteins affect development by regulating downstream gene networks involved in body patterning.
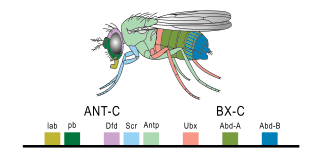
The Bithorax complex (BX-C) is one of two Drosophila melanogaster homeotic gene complexes, located on the right arm of chromosome 3. It is responsible for the differentiation of the posterior two-thirds of the fly by the regulation of three genes within the complex: Ultrabithorax (Ubx), abdominal A (abd-A), and Abdominal B (Abd-B).
A segmentation gene is a generic term for a gene whose function is to specify tissue pattern in each repeated unit of a segmented organism. Animals are constructed of segments; however, Drosophila segments also contain subdivided compartments. There are five gene classes which each contribute to the segmentation and development of the embryonic drosophila. These five gene classes include the coordinate gene, gap gene, pair-rule gene, segment polarity gene, and homeotic gene. In embryonic drosophila, the pair-rule gene defines odd-skipped and even-skipped genes as parasegments, showing 7 stripes in the embryo. In the next gene class, segment polarity gene, individual segments each have their own anterior and posterior pole, resulting in 14 segments. In the fruit fly Drosophila melanogaster, segment polarity genes help to define the anterior and posterior polarities within each embryonic parasegment by regulating the transmission of signals via the Wnt signaling pathway and Hedgehog signaling pathway. Segment polarity genes are expressed in the embryo following expression of the gap genes and pair-rule genes. The most commonly cited examples of these genes are engrailed and gooseberry in Drosophila melanogaster. The segment polarity is the last step in embryonic development and a repeated pattern where each half of each segment is deleted and a mirror-image is duplicated and reversed to replace that half segment; thus, forming a pattern element.
Staufen is a protein product of a maternally expressed gene first identified in Drosophila melanogaster. The protein has been implicated in helping regulate genes important in determination of gradients that set up the anterior posterior axis such as bicoid and oskar. Staufen proteins, abbreviated Stau, are necessary for cell localization during the oogenesis and zygotic development. It is involved in targeting of the messenger RNA encoding these genes to the correct pole of the egg cell.
Vasa is an RNA binding protein with an ATP-dependent RNA helicase that is a member of the DEAD box family of proteins. The vasa gene is essential for germ cell development and was first identified in Drosophila melanogaster, but has since been found to be conserved in a variety of vertebrates and invertebrates including humans. The Vasa protein is found primarily in germ cells in embryos and adults, where it is involved in germ cell determination and function, as well as in multipotent stem cells, where its exact function is unknown.
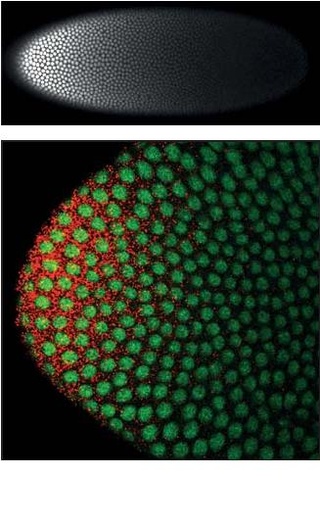
Homeotic protein bicoid is encoded by the bcd maternal effect gene in Drosophilia. Homeotic protein bicoid concentration gradient patterns the anterior-posterior (A-P) axis during Drosophila embryogenesis. Bicoid was the first protein demonstrated to act as a morphogen. Although bicoid is important for the development of Drosophila and other higher dipterans, it is absent from most other insects, where its role is accomplished by other genes.
Evx1 is a mammalian gene located downstream of the HoxA cluster, which encodes for a homeobox transcription factor. Evx1 is a homolog of even-skipped (eve), which is a pair-rule gene that regulates body segmentation in Drosophila. The expression of Evx1 is developmentally regulated, displaying a biphasic expression pattern with peak expression in the primitive streak during gastrulation and in interneurons during neural development. Evx1 has been shown to regulate anterior-posterior patterning during gastrulation by acting as a downstream effector of the Wnt and BMP signalling pathways. It is also a critical regulator of interneuron identity.
Hunchback is a maternal effect gene and gap gene found in the egg and zygotic genome of Drosophila embryogenesis. It is activated by the maternal Bicoid morphogen gradient and determines the anterior-posterior axis within the syncytial blastoderm. Hunchback is a Zinc finger transcription factor that coordinates the expression of other gap genes such as Krüppel, knirps, and giant.











