
Oct-4, also known as POU5F1, is a protein that in humans is encoded by the POU5F1 gene. Oct-4 is a homeodomain transcription factor of the POU family. It is critically involved in the self-renewal of undifferentiated embryonic stem cells. As such, it is frequently used as a marker for undifferentiated cells. Oct-4 expression must be closely regulated; too much or too little will cause differentiation of the cells.

X-inactivation is a process by which one of the copies of the X chromosome is inactivated in therian female mammals. The inactive X chromosome is silenced by being packaged into a transcriptionally inactive structure called heterochromatin. As nearly all female mammals have two X chromosomes, X-inactivation prevents them from having twice as many X chromosome gene products as males, who only possess a single copy of the X chromosome.

Sir Martin John EvansFLSW is an English biologist who, with Matthew Kaufman, was the first to culture mice embryonic stem cells and cultivate them in a laboratory in 1981. He is also known, along with Mario Capecchi and Oliver Smithies, for his work in the development of the knockout mouse and the related technology of gene targeting, a method of using embryonic stem cells to create specific gene modifications in mice. In 2007, the three shared the Nobel Prize in Physiology or Medicine in recognition of their discovery and contribution to the efforts to develop new treatments for illnesses in humans.

In molecular biology, miR-130 microRNA precursor is a small non-coding RNA that regulates gene expression. This microRNA has been identified in mouse, and in human. miR-130 appears to be vertebrate-specific miRNA and has now been predicted or experimentally confirmed in a range of vertebrate species. Mature microRNAs are processed from the precursor stem-loop by the Dicer enzyme. In this case, the mature sequence is excised from the 3' arm of the hairpin. It has been found that miR-130 is upregulated in a type of cancer called hepatocellular carcinoma. It has been shown that miR-130a is expressed in the hematopoietic stem/progenitor cell compartment but not in mature blood cells.

SRY -box 2, also known as SOX2, is a transcription factor that is essential for maintaining self-renewal, or pluripotency, of undifferentiated embryonic stem cells. Sox2 has a critical role in maintenance of embryonic and neural stem cells.

Shinya Yamanaka is a Japanese stem cell researcher and a Nobel Prize laureate. He is a professor and the director emeritus of Center for iPS Cell Research and Application, Kyoto University; as a senior investigator at the UCSF-affiliated Gladstone Institutes in San Francisco, California; and as a professor of anatomy at University of California, San Francisco (UCSF). Yamanaka is also a past president of the International Society for Stem Cell Research (ISSCR).

Xist is a non-coding RNA on the X chromosome of the placental mammals that acts as a major effector of the X-inactivation process. It is a component of the Xic – X-chromosome inactivation centre – along with two other RNA genes and two protein genes.
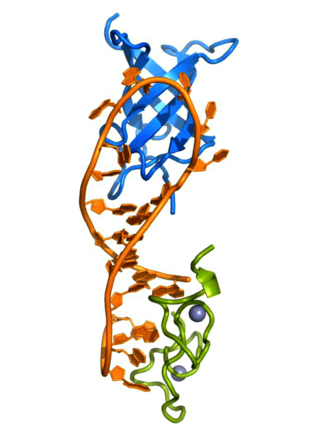
Lin-28 homolog A is a protein that in humans is encoded by the LIN28 gene.
Sally Temple is an American developmental neuroscientist in Albany, New York. She is a co-founder and scientific director for The Neural Stem Cell Institute and is a professor of Neuroscience and Neuropharmacology at Albany Medical College Temple is also the principal investigator in her laboratory that focuses on neural stem cells and therapies for neurological-related disorders

Cell potency is a cell's ability to differentiate into other cell types. The more cell types a cell can differentiate into, the greater its potency. Potency is also described as the gene activation potential within a cell, which like a continuum, begins with totipotency to designate a cell with the most differentiation potential, pluripotency, multipotency, oligopotency, and finally unipotency.

Ming-Ming Zhou is an American scientist who focuses on structural and chemical biology, NMR spectroscopy, and drug design. He is the Dr. Harold and Golden Lamport Professor and Chairman of the Department of Pharmacological Sciences. He is also the co-director of the Drug Discovery Institute at the Icahn School of Medicine at Mount Sinai and Mount Sinai Health System in New York City, as well as Professor of Sciences. Zhou is an elected fellow of the American Association for the Advancement of Science.
In molecular biology mir-367 microRNA is a short RNA molecule. MicroRNAs function to regulate the expression levels of other genes by several mechanisms.

Tsix is a non-coding RNA gene that is antisense to the Xist RNA. Tsix binds Xist during X chromosome inactivation. The name Tsix comes from the reverse of Xist, which stands for X-inactive specific transcript.

Dame Amanda Gay Fisher is a British cell biologist and Director of the Medical Research Council (MRC) London Institute of Medical Sciences at the Hammersmith Hospital campus of Imperial College London, where she is also a Professor leading the Institute of Clinical Sciences. She has made contributions to multiple areas of cell biology, including determining the function of several genes in HIV and describing the importance of a gene's location within the cell nucleus.

X chromosome inactivation (XCI) is the phenomenon that has been selected during the evolution to balance X-linked gene dosage between XX females and XY males.
Magdalena Żernicka-Goetz is a Polish-British developmental biologist. She is Professor of Mammalian Development and Stem Cell Biology in the Department of Physiology, Development and Neuroscience and Fellow of Sidney Sussex College, Cambridge. She also serves as Bren Professor of Biology and Biological Engineering at California Institute of Technology (Caltech).
Jeannie T. Lee is a Professor of Genetics at Harvard Medical School and the Massachusetts General Hospital, and a Howard Hughes Medical Institute Investigator. She is known for her work on X-chromosome inactivation and for discovering the functions of a new class of epigenetic regulators known as long noncoding RNAs (lncRNAs), including Xist and Tsix.
Emily Bernstein is a professor at Mount Sinai School of Medicine known for her research on RNA interference, epigenetics, and cancer, especially melanoma.
X chromosome reactivation (XCR) is the process by which the inactive X chromosome (the Xi) is re-activated in the cells of eutherian female mammals. Therian female mammalian cells have two X chromosomes, while males have only one, requiring X-chromosome inactivation (XCI) for sex-chromosome dosage compensation. In eutherians, XCI is the random inactivation of one of the X chromosomes, silencing its expression. Much of the scientific knowledge currently known about XCR comes from research limited to mouse models or stem cells.
Nissim Benvenisty is Professor of Genetics, the Herbert Cohn Chair in Cancer Research and the Director of “The Azrieli Center for Stem Cells and Genetic Research” at the Alexander Silberman Institute of Life Sciences, Hebrew University.













