The alkali metals consist of the chemical elements lithium (Li), sodium (Na), potassium (K), rubidium (Rb), caesium (Cs), and francium (Fr). Together with hydrogen they constitute group 1, which lies in the s-block of the periodic table. All alkali metals have their outermost electron in an s-orbital: this shared electron configuration results in their having very similar characteristic properties. Indeed, the alkali metals provide the best example of group trends in properties in the periodic table, with elements exhibiting well-characterised homologous behaviour. This family of elements is also known as the lithium family after its leading element.

Electrochemistry is the branch of physical chemistry concerned with the relationship between electrical potential difference, as a measurable and quantitative phenomenon, and identifiable chemical change, with the potential difference as an outcome of a particular chemical change, or vice versa. These reactions involve electrons moving via an electronically-conducting phase between electrodes separated by an ionically conducting and electronically insulating electrolyte.
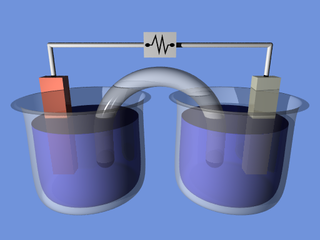
An electrochemical cell is a device that generates electrical energy from chemical reactions. Electrical energy can also be applied to these cells to cause chemical reactions to occur. Electrochemical cells which generate an electric current are called voltaic or galvanic cells and those that generate chemical reactions, via electrolysis for example, are called electrolytic cells.
An electrolyte is a medium containing ions that is electrically conducting through the movement of those ions, but not conducting electrons. This includes most soluble salts, acids, and bases dissolved in a polar solvent, such as water. Upon dissolving, the substance separates into cations and anions, which disperse uniformly throughout the solvent. Solid-state electrolytes also exist. In medicine and sometimes in chemistry, the term electrolyte refers to the substance that is dissolved.
Active transport is necessary for many essential physiological processes in living organisms, such as the uptake of nutrients, the removal of waste products, the regulation of ion concentrations, and the transmission of nerve impulses.

A pH meter is a scientific instrument that measures the hydrogen-ion activity in water-based solutions, indicating its acidity or alkalinity expressed as pH. The pH meter measures the difference in electrical potential between a pH electrode and a reference electrode, and so the pH meter is sometimes referred to as a "potentiometric pH meter". The difference in electrical potential relates to the acidity or pH of the solution. Testing of pH via pH meters (pH-metry) is used in many applications ranging from laboratory experimentation to quality control.

An automated analyser is a medical laboratory instrument designed to measure various substances and other characteristics in a number of biological samples quickly, with minimal human assistance. These measured properties of blood and other fluids may be useful in the diagnosis of disease.
In cellular biology, membrane transport refers to the collection of mechanisms that regulate the passage of solutes such as ions and small molecules through biological membranes, which are lipid bilayers that contain proteins embedded in them. The regulation of passage through the membrane is due to selective membrane permeability – a characteristic of biological membranes which allows them to separate substances of distinct chemical nature. In other words, they can be permeable to certain substances but not to others.

Nafion is a brand name for a sulfonated tetrafluoroethylene based fluoropolymer-copolymer discovered in the late 1960s by Dr. Walther Grot of DuPont. Nafion is a brand of the Chemours company. It is the first of a class of synthetic polymers with ionic properties that are called ionomers. Nafion's unique ionic properties are a result of incorporating perfluorovinyl ether groups terminated with sulfonate groups onto a tetrafluoroethylene (PTFE) backbone. Nafion has received a considerable amount of attention as a proton conductor for proton exchange membrane (PEM) fuel cells because of its excellent chemical and mechanical stability in the harsh conditions of this application.
A glass electrode is a type of ion-selective electrode made of a doped glass membrane that is sensitive to a specific ion. The most common application of ion-selective glass electrodes is for the measurement of pH. The pH electrode is an example of a glass electrode that is sensitive to hydrogen ions. Glass electrodes play an important part in the instrumentation for chemical analysis and physicochemical studies. The voltage of the glass electrode, relative to some reference value, is sensitive to changes in the activity of a certain type of ions.
Potassium selective electrodes are a type of ion selective electrode used in biochemical and biophysical research, where measurements of potassium concentration in an aqueous solution are required, usually on a real time basis.

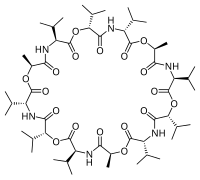
Valinomycin is a naturally occurring dodecadepsipeptide used in the transport of potassium and as an antibiotic. Valinomycin is obtained from the cells of several Streptomyces species, S. fulvissimus being a notable one.

Ion chromatography separates ions and polar molecules based on their affinity to the ion exchanger. It works on almost any kind of charged molecule—including large proteins, small nucleotides, and amino acids. However, ion chromatography must be done in conditions that are one unit away from the isoelectric point of a protein.

Electrodialysis (ED) is used to transport salt ions from one solution through ion-exchange membranes to another solution under the influence of an applied electric potential difference. This is done in a configuration called an electrodialysis cell. The cell consists of a feed (dilute) compartment and a concentrate (brine) compartment formed by an anion exchange membrane and a cation exchange membrane placed between two electrodes. In almost all practical electrodialysis processes, multiple electrodialysis cells are arranged into a configuration called an electrodialysis stack, with alternating anion and cation-exchange membranes forming the multiple electrodialysis cells. Electrodialysis processes are different from distillation techniques and other membrane based processes in that dissolved species are moved away from the feed stream, whereas other processes move away the water from the remaining substances. Because the quantity of dissolved species in the feed stream is far less than that of the fluid, electrodialysis offers the practical advantage of much higher feed recovery in many applications.
In analytical chemistry, potentiometric titration is a technique similar to direct titration of a redox reaction. It is a useful means of characterizing an acid. No indicator is used; instead the electric potential is measured across the analyte, typically an electrolyte solution. To do this, two electrodes are used, an indicator electrode and a reference electrode. Reference electrodes generally used are hydrogen electrodes, calomel electrodes, and silver chloride electrodes. The indicator electrode forms an electrochemical half-cell with the interested ions in the test solution. The reference electrode forms the other half-cell.
A fluoride selective electrode is a type of ion selective electrode sensitive to the concentration of the fluoride ion. A common example is the lanthanum fluoride electrode.
Binding selectivity is defined with respect to the binding of ligands to a substrate forming a complex. Binding selectivity describes how a ligand may bind more preferentially to one receptor than another. A selectivity coefficient is the equilibrium constant for the reaction of displacement by one ligand of another ligand in a complex with the substrate. Binding selectivity is of major importance in biochemistry and in chemical separation processes.
Electrodeionization (EDI) is a water treatment technology that uses DC power, ion exchange membranes, and ion exchange resin to deionize water. EDI is usually a polishing treatment to reverse osmosis (RO). EDI differs from other RO polishing technologies such as chemically regenerated mixed beds, in that it is continuous and requires no chemical regeneration. EDI is sometimes referred to as continuous electrodeionization (CEDI) since the electric current regenerates the ion exchange resin mass continuously. The CEDI technique can achieve high purity, with product conductivity on the order of 0.1 μS/cm and sometimes with resistivity as high as 18.2 MΩ-cm. Electrodeionization (EDI) is a combination of three different processes: Electrolysis, where with a continuously applied electric DC current, both positive and negative ions are directed to the electrode that has opposing electrical charge; the electrical potential pulls anions and cations from diluting chambers, though cation or anion exchange membranes, into concentrating chambers. Ion exchange, where ion exchange resin makes up the diluting chambers, and the cations and anions are affixed to resin sites as water passes through the resin bed; and chemical regeneration, which occurs by water splitting. In a chemically regenerated mixed bed, the hydrogen (H+) of acid regenerates cation resin. The hydroxide (OH-) of sodium hydroxide (caustic soda) regenerates anion resin. In EDI, the electrical current causes water to split from H2O → H+ + OH-, regenerating the resin without the need for addition of external chemicals.
A biotransducer is the recognition-transduction component of a biosensor system. It consists of two intimately coupled parts; a bio-recognition layer and a physicochemical transducer, which acting together converts a biochemical signal to an electronic or optical signal. The bio-recognition layer typically contains an enzyme or another binding protein such as antibody. However, oligonucleotide sequences, sub-cellular fragments such as organelles and receptor carrying fragments, single whole cells, small numbers of cells on synthetic scaffolds, or thin slices of animal or plant tissues, may also comprise the bio-recognition layer. It gives the biosensor selectivity and specificity. The physicochemical transducer is typically in intimate and controlled contact with the recognition layer. As a result of the presence and biochemical action of the analyte, a physico-chemical change is produced within the biorecognition layer that is measured by the physicochemical transducer producing a signal that is proportionate to the concentration of the analyte. The physicochemical transducer may be electrochemical, optical, electronic, gravimetric, pyroelectric or piezoelectric. Based on the type of biotransducer, biosensors can be classified as shown to the right.
A polymer electrolyte is a polymer matrix capable of ion conduction. Much like other types of electrolyte—liquid and solid-state—polymer electrolytes aid in movement of charge between the anode and cathode of a cell. The use of polymers as an electrolyte was first demonstrated using dye-sensitized solar cells. The field has expanded since and is now primarily focused on the development of polymer electrolytes with applications in batteries, fuel cells, and membranes.