
Serotonin–norepinephrine reuptake inhibitors (SNRIs) are a class of antidepressant medications used to treat major depressive disorder (MDD), anxiety disorders, social phobia, chronic neuropathic pain, fibromyalgia syndrome (FMS), and menopausal symptoms. Off-label uses include treatments for attention-deficit hyperactivity disorder (ADHD), obsessive–compulsive disorder (OCD), and migraine prevention. SNRIs are monoamine reuptake inhibitors; specifically, they inhibit the reuptake of serotonin and norepinephrine. These neurotransmitters are thought to play an important role in mood regulation. SNRIs can be contrasted with the selective serotonin reuptake inhibitors (SSRIs) and norepinephrine reuptake inhibitors (NRIs), which act upon single neurotransmitters.

Elan Corporation plc was a major drugs firm based in Dublin, Ireland, which had major interests in the United States. It was listed on the New York Stock Exchange as ELN, the Irish Stock Exchange as ELN.I, and the London Stock Exchange as ELN.L. In 2013, the company merged with Perrigo to form Perrigo Company PLC.
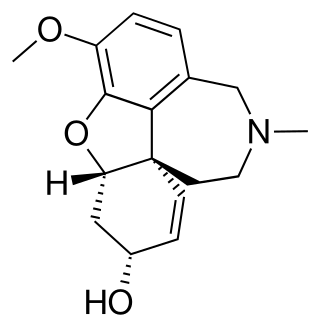
Galantamine is a type of acetylcholinesterase inhibitor. It is an alkaloid extracted from the bulbs and flowers of Galanthus nivalis, Galanthus caucasicus, Galanthus woronowii, and other members of the family Amaryllidaceae, such as Narcissus (daffodil), Leucojum aestivum (snowflake), and Lycoris including Lycoris radiata. It can also be produced synthetically.
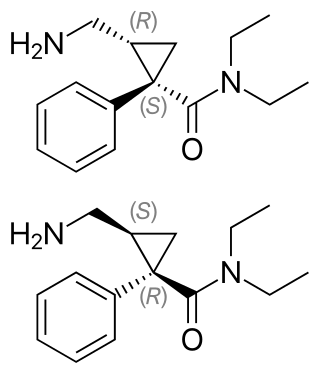
Milnacipran is a serotonin–norepinephrine reuptake inhibitor (SNRI) used in the clinical treatment of fibromyalgia. It is not approved for the clinical treatment of major depressive disorder in the US, but it is in other countries.

Huperzine A is a naturally-occurring sesquiterpene alkaloid compound found in the firmoss Huperzia serrata and in varying quantities in other food Huperzia species, including H. elmeri, H. carinat, and H. aqualupian. Huperzine A has been investigated as a treatment for neurological conditions such as Alzheimer's disease, but a 2013 meta-analysis of those studies concluded that they were of poor methodological quality and the findings should be interpreted with caution. Huperzine A inhibits the breakdown of the neurotransmitter acetylcholine (ACh) by the enzyme acetylcholinesterase. It is commonly available over the counter as a nutritional supplement and marketed as a memory and concentration enhancer.

Monoclonal antibodies (mAbs) have varied therapeutic uses. It is possible to create a mAb that binds specifically to almost any extracellular target, such as cell surface proteins and cytokines. They can be used to render their target ineffective, to induce a specific cell signal, to cause the immune system to attack specific cells, or to bring a drug to a specific cell type.

Latrepirdine is an antihistamine drug which has been used clinically in Russia since 1983.

Neramexane is a drug related to memantine, which acts as an NMDA antagonist and has neuroprotective effects. It is being developed for various possible applications, including treatment of tinnitus, Alzheimer's disease, drug addiction and as an analgesic. Animal studies have also suggested antidepressant and nootropic actions, so there are a wide range of potential applications this drug may be used for. It also acts as a nicotinic acetylcholine receptor antagonist.
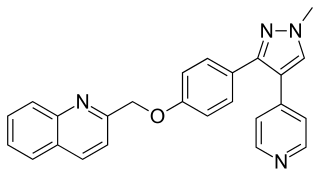
Mardepodect is a drug which was developed by Pfizer for the treatment of schizophrenia. It acts as a phosphodiesterase inhibitor selective for the PDE10A subtype. The PDE10A enzyme is expressed primarily in the brain, mostly in the striatum, nucleus accumbens and olfactory tubercle, and is thought to be particularly important in regulating the activity of dopamine-sensitive medium spiny neurons in the striatum which are known to be targets of conventional antipsychotic drugs. Older PDE10A inhibitors such as papaverine have been shown to produce antipsychotic effects in animal models, and more potent and selective PDE10A inhibitors are a current area of research for novel antipsychotic drugs which act through a different pathway to conventional dopamine or 5-HT2A antagonist drugs and may have a more favourable side effects profile. Mardepodect is currently one of the furthest advanced PDE10A inhibitors in development and has progressed through to Phase II clinical trials in humans. In 2017, development of mardepodect for the treatment of schizophrenia and Huntington's disease was discontinued.

Acetylcholinesterase inhibitors (AChEIs) also often called cholinesterase inhibitors, inhibit the enzyme acetylcholinesterase from breaking down the neurotransmitter acetylcholine into choline and acetate, thereby increasing both the level and duration of action of acetylcholine in the central nervous system, autonomic ganglia and neuromuscular junctions, which are rich in acetylcholine receptors. Acetylcholinesterase inhibitors are one of two types of cholinesterase inhibitors; the other being butyryl-cholinesterase inhibitors. Acetylcholinesterase is the primary member of the cholinesterase enzyme family.

PBT2 is a safe-for-human-use Zinc ionophore and an experimental drug candidate. It is a second-generation 8-hydroxyquinoline analog intended to be a successor to clioquinol and a potential treatment of Alzheimer's disease and Huntington's disease.

Rivastigmine is a cholinesterase inhibitor used for the treatment of mild to moderate Alzheimer's disease. The drug can be administered orally or via a transdermal patch; the latter form reduces the prevalence of side effects, which typically include nausea and vomiting.
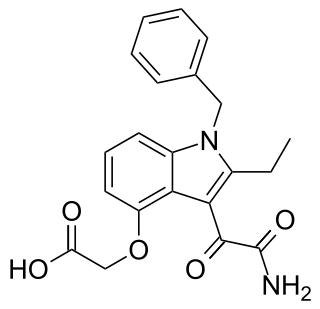
Varespladib is an inhibitor of the IIa, V, and X isoforms of secretory phospholipase A2 (sPLA2). The molecule acts as an anti-inflammatory agent by disrupting the first step of the arachidonic acid pathway of inflammation. From 2006 to 2012, varespladib was under active investigation by Anthera Pharmaceuticals as a potential therapy for several inflammatory diseases, including acute coronary syndrome and acute chest syndrome. The trial was halted in March 2012 due to inadequate efficacy. The selective sPLA2 inhibitor varespladib (IC50 value 0.009 μM in chromogenic assay, mole fraction 7.3X10-6) was studied in the VISTA-16 randomized clinical trial (clinicaltrials.gov Identifier: NCT01130246) and the results were published in 2014. The sPLA2 inhibition by varespladib in this setting seemed to be potentially harmful, and thus not a useful strategy for reducing adverse cardiovascular outcomes from acute coronary syndrome. Since 2016, scientific research has focused on the use of Varespladib as an inhibitor of snake venom toxins using various types of in vitro and in vivo models. Varespladib showed a significant inhibitory effect to snake venom PLA2 which makes it a potential first-line drug candidate in snakebite envenomation therapy. In 2019, the U.S. Food and Drug Administration (FDA) granted varespladib orphan drug status for its potential to treat snakebite.

Protein O-GlcNAc transferase also known as OGT or O-linked N-acetylglucosaminyltransferase is an enzyme that in humans is encoded by the OGT gene. OGT catalyzes the addition of the O-GlcNAc post-translational modification to proteins.

Protein O-GlcNAcase (EC 3.2.1.169, OGA, glycoside hydrolase O-GlcNAcase, O-GlcNAcase, BtGH84, O-GlcNAc hydrolase) is an enzyme with systematic name (protein)-3-O-(N-acetyl-D-glucosaminyl)-L-serine/threonine N-acetylglucosaminyl hydrolase. OGA is encoded by the OGA gene. This enzyme catalyses the removal of the O-GlcNAc post-translational modification in the following chemical reaction:
- [protein]-3-O-(N-acetyl-β-D-glucosaminyl)-L-serine + H2O ⇌ [protein]-L-serine + N-acetyl-D-glucosamine
- [protein]-3-O-(N-acetyl-β-D-glucosaminyl)-L-threonine + H2O ⇌ [protein]-L-threonine + N-acetyl-D-glucosamine

Saracatinib (AZD-0530) is an experimental drug being developed by AstraZeneca. It acts as a dual kinase inhibitor, with selective actions as a Src inhibitor and a Bcr-Abl tyrosine-kinase inhibitor.

Daridorexant, sold under the brand name Quviviq, is an orexin antagonist medication which is used for the treatment of insomnia. Daridorexant is taken by mouth.

Phenserine is a synthetic drug which has been investigated as a medication to treat Alzheimer's disease (AD), as the drug exhibits neuroprotective and neurotrophic effects.

Nirmatrelvir is an antiviral medication developed by Pfizer which acts as an orally active 3C-like protease inhibitor. It is part of a nirmatrelvir/ritonavir combination used to treat COVID-19 and sold under the brand name Paxlovid.

Buntanetap is an orally-administered small molecule inhibitor of several neurotoxic proteins that is under investigation in the treatment of Alzheimer's disease, frontotemporal dementia, chronic traumatic encephalopathy and Parkinson's disease. It is the (+) enantiomer of phenserine, as the (-) enantiomer also has unwanted anticholinergic effects. It is currently in phase III trials for the treatment of Parkinson's.




















