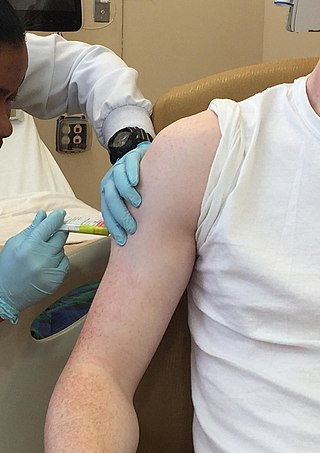
Clinical trials are prospective biomedical or behavioral research studies on human participants designed to answer specific questions about biomedical or behavioral interventions, including new treatments and known interventions that warrant further study and comparison. Clinical trials generate data on dosage, safety and efficacy. They are conducted only after they have received health authority/ethics committee approval in the country where approval of the therapy is sought. These authorities are responsible for vetting the risk/benefit ratio of the trial—their approval does not mean the therapy is 'safe' or effective, only that the trial may be conducted.
In a blind or blinded experiment, information which may influence the participants of the experiment is withheld until after the experiment is complete. Good blinding can reduce or eliminate experimental biases that arise from a participants' expectations, observer's effect on the participants, observer bias, confirmation bias, and other sources. A blind can be imposed on any participant of an experiment, including subjects, researchers, technicians, data analysts, and evaluators. In some cases, while blinding would be useful, it is impossible or unethical. For example, it is not possible to blind a patient to their treatment in a physical therapy intervention. A good clinical protocol ensures that blinding is as effective as possible within ethical and practical constraints.
Pharmacovigilance, also known as drug safety, is the pharmaceutical science relating to the "collection, detection, assessment, monitoring, and prevention" of adverse effects with pharmaceutical products. The etymological roots for the word "pharmacovigilance" are: pharmakon and vigilare. As such, pharmacovigilance heavily focuses on adverse drug reactions (ADR), which are defined as any response to a drug which is noxious and unintended, including lack of efficacy. Medication errors such as overdose, and misuse and abuse of a drug as well as drug exposure during pregnancy and breastfeeding, are also of interest, even without an adverse event, because they may result in an adverse drug reaction.

The United States Food and Drug Administration's Investigational New Drug (IND) program is the means by which a pharmaceutical company obtains permission to start human clinical trials and to ship an experimental drug across state lines before a marketing application for the drug has been approved. Regulations are primarily at 21 CFR 312. Similar procedures are followed in the European Union, Japan, and Canada due to regulatory harmonization efforts by the International Council for Harmonisation.
In the life sciences, a contract research organization (CRO) is a company that provides support to the pharmaceutical, biotechnology, and medical device industries in the form of research services outsourced on a contract basis. A CRO may provide such services as biopharmaceutical development, biological assay development, commercialization, clinical development, clinical trials management, pharmacovigilance, outcomes research, and real world evidence.
In a clinical trial, the investigators must specify inclusion and exclusion criteria for participation in the study.
In pharmaceuticals, an adverse event (AE) is any untoward medical occurrence in a patient or clinical investigation subject administered a pharmaceutical product and which does not necessarily have a causal relationship with this treatment. An adverse event can therefore be any unfavourable and unintended symptom or sign or disease temporally associated with the use of a medicinal (investigational) product, whether or not related to the medicinal (investigational) product.
In drug development and production, good clinical practice (GCP) is an international quality standard, which governments can then transpose into regulations for clinical trials involving human subjects. GCP follows the International Council for Harmonisation of Technical Requirements for Registration of Pharmaceuticals for Human Use (ICH), and enforces tight guidelines on ethical aspects of clinical research.
In medicine an intention-to-treat (ITT) analysis of the results of a randomized controlled trial is based on the initial treatment assignment and not on the treatment eventually received. ITT analysis is intended to avoid various misleading artifacts that can arise in intervention research such as non-random attrition of participants from the study or crossover. ITT is also simpler than other forms of study design and analysis, because it does not require observation of compliance status for units assigned to different treatments or incorporation of compliance into the analysis. Although ITT analysis is widely employed in published clinical trials, it can be incorrectly described and there are some issues with its application. Furthermore, there is no consensus on how to carry out an ITT analysis in the presence of missing outcome data.
In drug development and medical device development the Investigator's Brochure (IB) is a comprehensive document summarizing the body of information about an investigational product obtained during a drug trial. The IB is a document of critical importance throughout the drug development process and is updated with new information as it becomes available. The purpose of the IB is to compile data relevant to studies of the IP in human subjects gathered during preclinical and other clinical trials.
In epidemiology, reporting bias is defined as "selective revealing or suppression of information" by subjects. In artificial intelligence research, the term reporting bias is used to refer to people's tendency to under-report all the information available.
In natural and social science research, a protocol is most commonly a predefined procedural method in the design and implementation of an experiment. Protocols are written whenever it is desirable to standardize a laboratory method to ensure successful replication of results by others in the same laboratory or by other laboratories. Additionally, and by extension, protocols have the advantage of facilitating the assessment of experimental results through peer review. In addition to detailed procedures, equipment, and instruments, protocols will also contain study objectives, reasoning for experimental design, reasoning for chosen sample sizes, safety precautions, and how results were calculated and reported, including statistical analysis and any rules for predefining and documenting excluded data to avoid bias.
A Clinical Research Coordinator (CRC) is a person responsible for conducting clinical trials using good clinical practice (GCP) under the auspices of a Principal Investigator (PI).
Clinical quality management systems (CQMS) are systems used in the life sciences sector designed to manage quality management best practices throughout clinical research and clinical study management. A CQMS system is designed to manage all of the documents, activities, tasks, processes, quality events, relationships, audits and training that must be administered and controlled throughout the life of a clinical trial. The premise of a CQMS is to bring together the activities led by two sectors of clinical research, Clinical Quality and Clinical Operations, to facilitate cross-functional activities to improve efficiencies and transparency and to encourage the use of risk mitigation and risk management practices at the clinical study level.
A glossary of terms used in clinical research.
Patient recruitment is the process of finding and enrolling suitable participants for clinical trials. It is a crucial aspect of drug development and medical research, as it affects the validity, reliability, and generalizability of the results. Patient recruitment can also be challenging, time-consuming, and costly, involving various ethical, regulatory, and logistical issues.
In order to comply with government regulatory requirements pertinent to clinical trials, every organization involved in clinical trials must maintain and store certain documents, images and content related to the clinical trial. Depending on the regulatory jurisdiction, this information may be stored in the trial master file or TMF, which today takes the form of an electronic trial master file (eTMF). The International Conference on Harmonization (ICH) published a consolidated guidance for industry on Good Clinical Practice in 1996 with the objective of providing a unified standard for the European Union, Japan, and the United States of America to facilitate mutual acceptance of clinical data by the regulatory authorities in those jurisdictions. This guidance document established the requirement across all ICH regions to establish trial master files containing essential documents that individually and collectively permit evaluation of the conduct of a trial and the quality of the data produced.[2] In some jurisdictions, for example the USA, there is no specific requirement for a trial master file. However, if the regulatory authority requires ICH GCP to be followed, then there is consequently a requirement to create and maintain a trial master file.[2]
The phases of clinical research are the stages in which scientists conduct experiments with a health intervention to obtain sufficient evidence for a process considered effective as a medical treatment. For drug development, the clinical phases start with testing for drug safety in a few human subjects, then expand to many study participants to determine if the treatment is effective. Clinical research is conducted on drug candidates, vaccine candidates, new medical devices, and new diagnostic assays.
Guidances for statistics in regulatory affairs refers to specific documents or guidelines that provide instructions, recommendations, and standards pertaining to the application of statistical methodologies and practices within the regulatory framework of industries such as pharmaceuticals and medical devices. These guidances serve as a reference for statisticians, researchers, and professionals involved in designing, conducting, analyzing, and reporting studies and trials in compliance with regulatory requirements. These documents embody the prevailing perspectives of regulatory agencies on specific subjects. It is worth noting that in the United States, the term "Guidances" is used, while in Europe, the term "Guidelines" is employed.


