Related Research Articles

A vaccine is a biological preparation that provides active acquired immunity to a particular infectious or malignant disease. The safety and effectiveness of vaccines has been widely studied and verified. A vaccine typically contains an agent that resembles a disease-causing microorganism and is often made from weakened or killed forms of the microbe, its toxins, or one of its surface proteins. The agent stimulates the body's immune system to recognize the agent as a threat, destroy it, and recognize further and destroy any of the microorganisms associated with that agent that it may encounter in the future.

Influenza vaccines, also known as flu shots, are vaccines that protect against infection by influenza viruses. New versions of the vaccines are developed twice a year, as the influenza virus rapidly changes. While their effectiveness varies from year to year, most provide modest to high protection against influenza. The United States Centers for Disease Control and Prevention (CDC) estimates that vaccination against influenza reduces sickness, medical visits, hospitalizations, and deaths. Immunized workers who do catch the flu return to work half a day sooner on average. Vaccine effectiveness in those over 65 years old remains uncertain due to a lack of high-quality research.
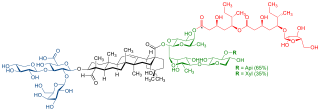
QS-21 is a purified plant extract used as a vaccine adjuvant. It is derived from the soap bark tree, which is native to the countries of Chile, Peru, and Bolivia. The crude drug is imported from Peru and Chile.

H5N1 clinical trials are clinical trials concerning H5N1 vaccines, which are intended to provide immunization to influenza A virus subtype H5N1. They are intended to discover pharmacological effects and identify any adverse reactions the vaccines may achieve in humans.
Agenus Inc. is a Lexington, Massachusetts-based biotechnology company focused on immunotherapy including immuno-oncology, a field that uses the immune system to control or cure cancer. The company is developing checkpoint modulators (CPMs), patient-specific anti-cancer vaccines, and adjuvants desugned for use with various vaccines. CPM development is a particularly fast-moving field, since early products have produced unprecedented clinical benefits for patients.
In immunology, an adjuvant is a substance that increases or modulates the immune response to a vaccine. The word "adjuvant" comes from the Latin word adiuvare, meaning to help or aid. "An immunologic adjuvant is defined as any substance that acts to accelerate, prolong, or enhance antigen-specific immune responses when used in combination with specific vaccine antigens."
Malaria vaccines are vaccines that prevent malaria, a mosquito-borne infectious disease which annually affects an estimated 247 million people worldwide and causes 619,000 deaths. The first approved vaccine for malaria is RTS,S, known by the brand name Mosquirix. As of April 2023, the vaccine has been given to 1.5 million children living in areas with moderate-to-high malaria transmission. It requires at least three doses in infants by age 2, and a fourth dose extends the protection for another 1–2 years. The vaccine reduces hospital admissions from severe malaria by around 30%.
Serum Institute of India (SII) is an Indian biotechnology and biopharmaceuticals company, based in Pune. It is the world's largest manufacturer of vaccines. It was founded by Cyrus Poonawalla in 1966 and is a part of Cyrus Poonawalla Group.
Immune stimulating complexes (ISCOMs) are spherical open cage-like structures (typically 40 nm in diameter) that are spontaneously formed when mixing together cholesterol, phospholipids and Quillaja saponins under a specific stoichiometry. The complex displays immune stimulating properties and is thus mainly used as a vaccine adjuvant in order to induce a stronger immune response and longer protection. A specific adjuvant based on ISCOM technology is Matrix-M.
A subunit vaccine is a vaccine that contains purified parts of the pathogen that are antigenic, or necessary to elicit a protective immune response. Subunit vaccine can be made from dissembled viral particles in cell culture or recombinant DNA expression, in which case it is a recombinant subunit vaccine.

Sir Adrian Vivian Sinton Hill, is an Irish vaccinologist, Director of the Jenner Institute and Lakshmi Mittal and Family Professor of Vaccinology at the University of Oxford, an honorary Consultant Physician in Infectious Diseases, and Fellow of Magdalen College, Oxford. Hill is a leader in the field of malaria vaccine development and was a co-leader of the research team which produced the Oxford–AstraZeneca COVID-19 vaccine, along with Professor Sarah Gilbert of the Jenner Institute and Professor Andrew Pollard of the Oxford Vaccine Group.
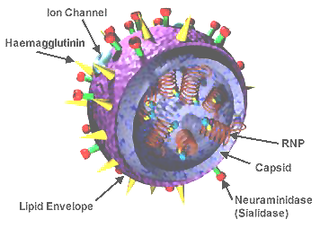
A universal flu vaccine is a flu vaccine that is effective against all influenza strains regardless of the virus sub type, antigenic drift or antigenic shift. Hence it should not require modification from year to year. As of 2021 no universal flu vaccine had been approved for general use, several were in development, and one was in clinical trial.

Dame Sarah Catherine Gilbert FRS is an English vaccinologist who is a Professor of Vaccinology at the University of Oxford and co-founder of Vaccitech. She specialises in the development of vaccines against influenza and emerging viral pathogens. She led the development and testing of the universal flu vaccine, which underwent clinical trials in 2011.

Vaxart, Inc. is an American biotechnology company focused on the discovery, development, and commercialization of oral recombinant vaccines administered using temperature-stable tablets that can be stored and shipped without refrigeration, eliminating the need for needle injection. Its development programs for oral vaccine delivery include prophylactic, enteric-coated tablet vaccines for inhibiting norovirus, seasonal influenza, respiratory syncytial virus, and human papillomavirus. It was founded in 2004 by Sean Tucker.
ChAdOx1 is an adenoviral vector for vaccines that was developed by the Jenner Institute, University of Oxford. The vector is a chimpanzee adenovirus modified to avoid its replication.

The Novavax COVID-19 vaccine, sold under the brand names Nuvaxovid and Covovax, among others, is a subunit COVID-19 vaccine developed by Novavax and the Coalition for Epidemic Preparedness Innovations (CEPI).

Shabir Ahmed Madhi is a South African physician who is professor of vaccinology and director of the South African Medical Research Council Respiratory and Meningeal Pathogens Research Unit at the University of the Witwatersrand, and National Research Foundation/Department of Science and Technology Research Chair in Vaccine Preventable Diseases. In January 2021, he was appointed Dean of the Faculty of Health Sciences at the University of the Witwateratand.

SCB-2019 is a protein subunit COVID-19 vaccine developed by Clover Biopharmaceuticals using an adjuvant from Dynavax technologies. Positive results of Phase I trials for the vaccine were published in The Lancet and the vaccine completed enrollment of 29,000 participants in Phase II/III trials in July 2021. In September 2021, SCB-2019 announced Phase III results showing 67% efficacy against all cases of COVID-19 and 79% efficacy against all cases of the Delta variant. Additionally, the vaccine was 84% effective against moderate cases and 100% effective against hospitalization.

COVID-19 vaccine clinical research uses clinical research to establish the characteristics of COVID-19 vaccines. These characteristics include efficacy, effectiveness, and safety. As of November 2022, 40 vaccines are authorized by at least one national regulatory authority for public use:
References
- 1 2 Datoo, Mehreen S.; Natama, Magloire H.; Somé, Athanase; Traoré, Ousmane; Rouamba, Toussaint; Bellamy, Duncan; Yameogo, Prisca; Valia, Daniel; Tegneri, Moubarak; Ouedraogo, Florence; Soma, Rachidatou (2021-05-15). "Efficacy of a low-dose candidate malaria vaccine, R21 in adjuvant Matrix-M, with seasonal administration to children in Burkina Faso: a randomised controlled trial". The Lancet. 397 (10287): 1809–1818. doi:10.1016/S0140-6736(21)00943-0. ISSN 0140-6736. PMC 8121760 . PMID 33964223.
- 1 2 3 Magnusson, Sofia E.; Reimer, Jenny M.; Karlsson, Karin H.; Lilja, Lena; Bengtsson, Karin Lövgren; Stertman, Linda (2013-03-25). "Immune enhancing properties of the novel Matrix-M adjuvant leads to potentiated immune responses to an influenza vaccine in mice". Vaccine. 31 (13): 1725–1733. doi:10.1016/j.vaccine.2013.01.039. ISSN 0264-410X. PMID 23384754.
- 1 2 "NanoFlu is on the FDA's Vaccine Fast-Track". www.precisionvaccinations.com. Retrieved 2021-10-27.
- ↑ "US Patent for Highly efficient influenza matrix (M1) proteins Patent (Patent # 10,544,399 issued January 28, 2020) - Justia Patents Search". patents.justia.com. Retrieved 2022-07-21.
- 1 2 3 Satyanarayana, Megha (2021-08-17). "A new, powerful malaria vaccine may be on the horizon". cen.acs.org. Retrieved 2021-10-27.
- ↑ "Is Old Vaccine Technology the Key to Hesitancy?". www.medpagetoday.com. 2021-06-17. Retrieved 2021-10-28.
- ↑ King2021-02-04T14:10:00+00:00, Anthony. "Covid vaccination efforts bolstered by trial results from J&J and Novavax". Chemistry World. Retrieved 2021-10-28.
- ↑ Milicic, Anita (24 March 2021). "Adjuvants: the unsung heroes of vaccines". The Conversation. Retrieved 2021-10-28.
- ↑ "Moths and tree bark: How the Novavax vaccine works". www.nebraskamed.com. Retrieved 2021-10-27.
- ↑ Bonam, Srinivasa Reddy; Partidos, Charalambos D.; Halmuthur, Sampath Kumar M.; Muller, Sylviane (September 2017). "An Overview of Novel Adjuvants Designed for Improving Vaccine Efficacy". Trends in Pharmacological Sciences. 38 (9): 771–793. doi:10.1016/j.tips.2017.06.002. ISSN 1873-3735. PMID 28668223. S2CID 205409092.
- 1 2 "Malaria vaccine becomes first to achieve WHO-specified 75 percent efficacy goal". ScienceDaily. Retrieved 2021-10-27.
- 1 2 Borrell, Brendan (2020-10-21). "The Tree That Could Help Stop the Pandemic". The Atlantic. Retrieved 2021-10-26.
- ↑ Tribble, Sarah Jane (2021-07-20). "Novavax's effort to vaccinate the world, from zero to not quite warp speed". Quartz. Retrieved 2023-04-13.