
Caffeine is a central nervous system (CNS) stimulant of the methylxanthine class. It is mainly used as a eugeroic (wakefulness promoter) or as a mild cognitive enhancer to increase alertness and attentional performance. Caffeine acts by blocking binding of adenosine to the adenosine A1 receptor, which enhances release of the neurotransmitter acetylcholine. Caffeine has a three-dimensional structure similar to that of adenosine, which allows it to bind and block its receptors. Caffeine also increases cyclic AMP levels through nonselective inhibition of phosphodiesterase.

Stimulants are a class of drugs that increase the activity of the brain and the spinal cord. They are used for various purposes, such as enhancing alertness, attention, motivation, cognition, mood, and physical performance. Some of the most common stimulants are caffeine, nicotine, amphetamines, cocaine, and modafinil.

The adenosine receptors (or P1 receptors) are a class of purinergic G protein-coupled receptors with adenosine as the endogenous ligand. There are four known types of adenosine receptors in humans: A1, A2A, A2B and A3; each is encoded by a different gene.

Caffeine dependence is a condition characterized by a set of criteria including tolerance, withdrawal symptoms, persistent desire or unsuccessful efforts to control use, and continued use despite knowledge of adverse consequences attributed to caffeine. It can appear in physical dependence or psychological dependence, or both. Caffeine is one of the most common additives in many consumer products, including pills and beverages such as caffeinated alcoholic beverages, energy drinks, pain reliever medications, and colas. Caffeine is found naturally in plants such as coffee and tea and other plants. Studies have found that 89 percent of adults in the U.S. consume on average 200 mg of caffeine daily. One area of concern that has been presented is the relationship between pregnancy and caffeine consumption. Repeated caffeine doses of 100mg appeared to result in smaller size at birth in newborns. When looking at birth weight however, caffeine consumption did not appear to make an impact.

Paraxanthine, also known as 1,7-dimethylxanthine, is a metabolite of theophylline and theobromine, two well-known stimulants found in coffee, tea, and chocolate mainly in the form of caffeine. It is a member of the xanthine family of alkaloids, which includes theophylline, theobromine and caffeine.

A dopamine agonist(DA) is a compound that activates dopamine receptors. There are two families of dopamine receptors, D1-like and D2-like. They are all G protein-coupled receptors. D1- and D5-receptors belong to the D1-like family and the D2-like family includes D2, D3 and D4 receptors. Dopamine agonists are primarily used in the treatment of Parkinson's disease, and to a lesser extent, in hyperprolactinemia and restless legs syndrome. They are also used off-label in the treatment of clinical depression. The use of dopamine agonists is associated with impulse control disorders and dopamine agonist withdrawal syndrome (DAWS).
An anxiogenic or panicogenic substance is one that causes anxiety. This effect is in contrast to anxiolytic agents, which inhibits anxiety. Together these categories of psychoactive compounds may be referred to as anxiotropic compounds.

Fencamfamin (INN), also known as fencamfamine or by the brand names Glucoenergan and Reactivan, is a stimulant which was developed by Merck in the 1960s.
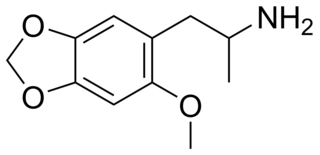
MMDA-2 (2-methoxy-4,5-methylenedioxyamphetamine) is a psychedelic drug of the amphetamine class. It is closely related to MMDA and MDA.

Metitepine, also known as methiothepin, is a drug described as a "psychotropic agent" of the tricyclic group which was never marketed. It acts as a non-selective antagonist of serotonin, dopamine, and adrenergic receptors and has antipsychotic properties.
A heteromer is something that consists of different parts; the antonym of homomeric. Examples are:

The cholecystokinin B receptor also known as CCKBR or CCK2 is a protein that in humans is encoded by the CCKBR gene.

Benocyclidine, also known as benzothiophenylcyclohexylpiperidine (BTCP), is a psychoactive recreational drug of the arylcyclohexylamine class which is related to phencyclidine (PCP). It was first described in a patent application naming Marc Caron and colleagues at Duke University in 1997.
Jane Stewart is a Canadian neuroscientist who has been active in the fields of psychology, psychiatry, and psychopharmacology. She is a professor emerita at Concordia University in Montreal, Canada.

Arylcyclohexylamines, also known as arylcyclohexamines or arylcyclohexanamines, are a chemical class of pharmaceutical, designer, and experimental drugs.

Melanocyte-inhibiting factor (also known as Pro-Leu-Gly-NH2, Melanostatin, MSH release–inhibiting hormone or MIF-1) is an endogenous peptide fragment derived from cleavage of the hormone oxytocin, but having generally different actions in the body. MIF-1 produces multiple effects, both blocking the effects of opioid receptor activation, while at the same time acting as a positive allosteric modulator of the D2 and D4 dopamine receptor subtypes, as well as inhibiting release of other neuropeptides such as alpha-MSH, and potentiating melatonin activity.

Levofenfluramine (INN), or (−)-3-trifluoromethyl-N-ethylamphetamine, also known as (−)-fenfluramine or (R)-fenfluramine, is a drug of the amphetamine family that, itself (i.e., in enantiopure form), was never marketed. It is the levorotatory enantiomer of fenfluramine, the racemic form of the compound, whereas the dextrorotatory enantiomer is dexfenfluramine. Both fenfluramine and dexfenfluramine are anorectic agents that have been used clinically in the treatment of obesity (and hence, levofenfluramine has been as well since it is a component of fenfluramine). However, they have since been discontinued due to reports of causing cardiovascular conditions such as valvular heart disease and pulmonary hypertension, adverse effects that are likely to be caused by excessive stimulation of 5-HT2B receptors expressed on heart valves.
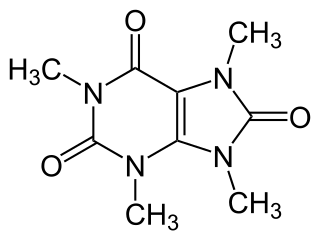
Theacrine, also known as 1,3,7,9-tetramethyluric acid, is a purine alkaloid found in Cupuaçu and in a Chinese tea known as kucha. It shows anti-inflammatory and analgesic effects and appears to affect adenosine signalling in a manner similar to caffeine. In kucha leaves, theacrine is synthesized from caffeine in what is thought to be a three-step pathway. Theacrine and caffeine are structurally similar.

Sheena Josselyn is a Canadian neuroscientist and a full professor of psychology and physiology at Hospital for Sick Children and The University of Toronto. Josselyn studies the neural basis of memory, specifically how the brain forms and stores memories in rodent models. She has made critical contributions to the field of Neuronal Memory Allocation and the study of engrams.

Bromerguride, also known as 2-bromolisuride, is an antidopaminergic and serotonergic agent of the ergoline group which was described as having atypical antipsychotic properties but was never marketed. It was the first antidopaminergic ergoline derivative to be discovered. The pharmacodynamic actions of bromerguride are said to be "reversed" relative to its parent compound lisuride, a dopaminergic agent.
















