Related Research Articles

Histidine (symbol His or H) is an essential amino acid that is used in the biosynthesis of proteins. It contains an α-amino group (which is in the protonated –NH3+ form under biological conditions), a carboxylic acid group (which is in the deprotonated –COO− form under biological conditions), and an imidazole side chain (which is partially protonated), classifying it as a positively charged amino acid at physiological pH. Initially thought essential only for infants, it has now been shown in longer-term studies to be essential for adults also. It is encoded by the codons CAU and CAC.

A protein complex or multiprotein complex is a group of two or more associated polypeptide chains. Protein complexes are distinct from multidomain enzymes, in which multiple catalytic domains are found in a single polypeptide chain.

Neurospora crassa is a type of red bread mold of the phylum Ascomycota. The genus name, meaning "nerve spore" in Greek, refers to the characteristic striations on the spores. The first published account of this fungus was from an infestation of French bakeries in 1843.

Amino acid synthesis is the set of biochemical processes by which the amino acids are produced. The substrates for these processes are various compounds in the organism's diet or growth media. Not all organisms are able to synthesize all amino acids. For example, humans can synthesize 11 of the 20 standard amino acids. These 11 are called the non-essential amino acids).

Lanosterol synthase is an oxidosqualene cyclase (OSC) enzyme that converts (S)-2,3-oxidosqualene to a protosterol cation and finally to lanosterol. Lanosterol is a key four-ringed intermediate in cholesterol biosynthesis. In humans, lanosterol synthase is encoded by the LSS gene.
Purine metabolism refers to the metabolic pathways to synthesize and break down purines that are present in many organisms.
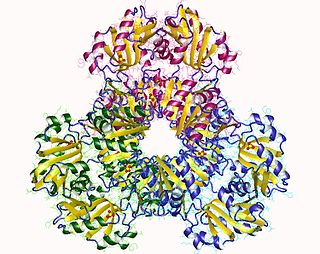
Ribose-phosphate diphosphokinase is an enzyme that converts ribose 5-phosphate into phosphoribosyl pyrophosphate (PRPP). It is classified under EC 2.7.6.1.

In enzymology, a phosphoribosylanthranilate isomerase (PRAI) is an enzyme that catalyzes the third step of the synthesis of the amino acid tryptophan.

Lysyl-tRNA synthetase is an enzyme that in humans is encoded by the KARS gene.

Aspartyl-tRNA synthetase, cytoplasmic is an enzyme that in humans is encoded by the DARS gene.

Aminoacyl tRNA synthetase complex-interacting multifunctional protein 2 is an enzyme that in humans is encoded by the AIMP2 gene.
The one gene–one enzyme hypothesis is the idea that genes act through the production of enzymes, with each gene responsible for producing a single enzyme that in turn affects a single step in a metabolic pathway. The concept was proposed by George Beadle and Edward Tatum in an influential 1941 paper on genetic mutations in the mold Neurospora crassa, and subsequently was dubbed the "one gene–one enzyme hypothesis" by their collaborator Norman Horowitz. In 2004, Horowitz reminisced that "these experiments founded the science of what Beadle and Tatum called 'biochemical genetics.' In actuality they proved to be the opening gun in what became molecular genetics and all the developments that have followed from that." The development of the one gene–one enzyme hypothesis is often considered the first significant result in what came to be called molecular biology. Although it has been extremely influential, the hypothesis was recognized soon after its proposal to be an oversimplification. Even the subsequent reformulation of the "one gene–one polypeptide" hypothesis is now considered too simple to describe the relationship between genes and proteins.

Ergothioneine is a naturally occurring amino acid and is a thiourea derivative of histidine, containing a sulfur atom on the imidazole ring. This compound occurs in relatively few organisms, notably actinomycetota, cyanobacteria, and certain fungi. Ergothioneine was discovered in 1909 and named after the ergot fungus from which it was first purified, with its structure being determined in 1911.

GCN2 is a serine/threonine-protein kinase that senses amino acid deficiency through binding to uncharged transfer RNA (tRNA). It plays a key role in modulating amino acid metabolism as a response to nutrient deprivation.
The term macromolecular assembly (MA) refers to massive chemical structures such as viruses and non-biologic nanoparticles, cellular organelles and membranes and ribosomes, etc. that are complex mixtures of polypeptide, polynucleotide, polysaccharide or other polymeric macromolecules. They are generally of more than one of these types, and the mixtures are defined spatially, and with regard to their underlying chemical composition and structure. Macromolecules are found in living and nonliving things, and are composed of many hundreds or thousands of atoms held together by covalent bonds; they are often characterized by repeating units. Assemblies of these can likewise be biologic or non-biologic, though the MA term is more commonly applied in biology, and the term supramolecular assembly is more often applied in non-biologic contexts. MAs of macromolecules are held in their defined forms by non-covalent intermolecular interactions, and can be in either non-repeating structures, or in repeating linear, circular, spiral, or other patterns. The process by which MAs are formed has been termed molecular self-assembly, a term especially applied in non-biologic contexts. A wide variety of physical/biophysical, chemical/biochemical, and computational methods exist for the study of MA; given the scale of MAs, efforts to elaborate their composition and structure and discern mechanisms underlying their functions are at the forefront of modern structure science.
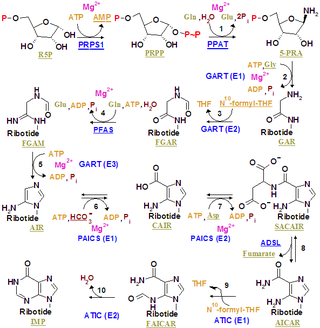
The purinosome is a putative multi-enzyme complex that carries out de novo purine biosynthesis within the cell. It is postulated to include all six of the human enzymes identified as direct participants in this ten-step biosynthetic pathway converting phosphoribosyl pyrophosphate to inosine monophosphate:
The frequency (frq) gene encodes the protein frequency (FRQ) that functions in the Neurospora crassa circadian clock. The FRQ protein plays a key role in circadian oscillator, serving to nucleate the negative element complex in the auto regulatory transcription-translation negative feedback-loop (TTFL) that is responsible for circadian rhythms in N. crassa. Similar rhythms are found in mammals, Drosophila and cyanobacteria. Recently, FRQ homologs have been identified in several other species of fungi. Expression of frq is controlled by the two transcription factors white collar-1 (WC-1) and white collar-2 (WC-2) that act together as the White Collar Complex (WCC) and serve as the positive element in the TTFL. Expression of frq can also be induced through light exposure in a WCC dependent manner. Forward genetics has generated many alleles of frq resulting in strains whose circadian clocks vary in period length.
White Collar-1 (wc-1) is a gene in Neurospora crassa encoding the protein WC-1. WC-1 has two separate roles in the cell. First, it is the primary photoreceptor for Neurospora and the founding member of the class of principle blue light photoreceptors in all of the fungi. Second, it is necessary for regulating circadian rhythms in FRQ. It is a key component of a circadian molecular pathway that regulates many behavioral activities, including conidiation. WC-1 and WC-2, an interacting partner of WC-1, comprise the White Collar Complex (WCC) that is involved in the Neurospora circadian clock. WCC is a complex of nuclear transcription factor proteins, and contains transcriptional activation domains, PAS domains, and zinc finger DNA-binding domains (GATA). WC-1 and WC-2 heterodimerize through their PAS domains to form the White Collar Complex (WCC).
The white collar--2 (wc-2) gene in Neurospora crassa encodes the protein White Collar-2 (WC-2). WC-2 is a GATA transcription factor necessary for blue light photoreception and for regulating circadian rhythms in Neurospora. In both contexts, WC-2 binds to its non-redundant counterpart White Collar-1 (WC-1) through PAS domains to form the White Collar Complex (WCC), an active transcription factor.
Transcription-translation coupling is a mechanism of gene expression regulation in which synthesis of an mRNA (transcription) is affected by its concurrent decoding (translation). In prokaryotes, mRNAs are translated while they are transcribed. This allows communication between RNA polymerase, the multisubunit enzyme that catalyzes transcription, and the ribosome, which catalyzes translation. Coupling involves both direct physical interactions between RNA polymerase and the ribosome, as well as ribosome-induced changes to the structure and accessibility of the intervening mRNA that affect transcription.
References
- ↑ Neuman, Nicole (2016). "The Complex Macromolecular Complex: Trends in Biochemical Sciences". Trends in Biochemical Sciences. 41 (1): 1–3. doi: 10.1016/j.tibs.2015.11.006 . PMID 26699226 . Retrieved 2018-07-11.
- 1 2 Ahmed A. Organization of the histidine-3 region of Neurospora. Mol Gen Genet. 1968;103(2):185-93. doi: 10.1007/BF00427145. PMID: 4306011