Related Research Articles

In biochemistry, phosphorylation is the attachment of a phosphate group to a molecule or an ion. This process and its inverse, dephosphorylation, are common in biology. Protein phosphorylation often activates many enzymes.

Glucose 6-phosphate is a glucose sugar phosphorylated at the hydroxy group on carbon 6. This dianion is very common in cells as the majority of glucose entering a cell will become phosphorylated in this way.

The insulin receptor (IR) is a transmembrane receptor that is activated by insulin, IGF-I, IGF-II and belongs to the large class of receptor tyrosine kinase. Metabolically, the insulin receptor plays a key role in the regulation of glucose homeostasis; a functional process that under degenerate conditions may result in a range of clinical manifestations including diabetes and cancer. Insulin signalling controls access to blood glucose in body cells. When insulin falls, especially in those with high insulin sensitivity, body cells begin only to have access to lipids that do not require transport across the membrane. So, in this way, insulin is the key regulator of fat metabolism as well. Biochemically, the insulin receptor is encoded by a single gene INSR, from which alternate splicing during transcription results in either IR-A or IR-B isoforms. Downstream post-translational events of either isoform result in the formation of a proteolytically cleaved α and β subunit, which upon combination are ultimately capable of homo or hetero-dimerisation to produce the ≈320 kDa disulfide-linked transmembrane insulin receptor.
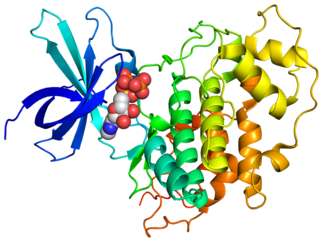
Glycogen synthase kinase 3 (GSK-3) is a serine/threonine protein kinase that mediates the addition of phosphate molecules onto serine and threonine amino acid residues. First discovered in 1980 as a regulatory kinase for its namesake, glycogen synthase (GS), GSK-3 has since been identified as a protein kinase for over 100 different proteins in a variety of different pathways. In mammals, including humans, GSK-3 exists in two isozymes encoded by two homologous genes GSK-3α (GSK3A) and GSK-3β (GSK3B). GSK-3 has been the subject of much research since it has been implicated in a number of diseases, including type 2 diabetes, Alzheimer's disease, inflammation, cancer, addiction and bipolar disorder.
Glycogenesis is the process of glycogen synthesis, in which glucose molecules are added to chains of glycogen for storage. This process is activated during rest periods following the Cori cycle, in the liver, and also activated by insulin in response to high glucose levels.

Edmond Henri Fischer was a Swiss-American biochemist. He and his collaborator Edwin G. Krebs were awarded the Nobel Prize in Physiology or Medicine in 1992 for describing how reversible phosphorylation works as a switch to activate proteins and regulate various cellular processes. From 2007 until 2014, he was the Honorary President of the World Cultural Council. At the time of his death at age 101 in 2021, he was the oldest living Nobel Prize laureate.

Glycogen phosphorylase is one of the phosphorylase enzymes. Glycogen phosphorylase catalyzes the rate-limiting step in glycogenolysis in animals by releasing glucose-1-phosphate from the terminal alpha-1,4-glycosidic bond. Glycogen phosphorylase is also studied as a model protein regulated by both reversible phosphorylation and allosteric effects.
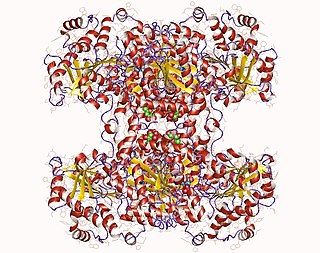
Glycogen synthase is a key enzyme in glycogenesis, the conversion of glucose into glycogen. It is a glycosyltransferase that catalyses the reaction of UDP-glucose and n to yield UDP and n+1.
The Division of Signal Transduction Therapy or DSTT is an organization managed by the University of Dundee, the Medical Research Council, and the pharmaceutical companies AstraZeneca, Boehringer Ingelheim, GlaxoSmithKline, Merck Serono, Janssen Pharmaceutica, and Pfizer. The purpose of the collaboration is to conduct cell signalling research and to encourage development of new drug treatments for global diseases such as cancer, rheumatoid arthritis, and Parkinson’s disease. Specifically the collaboration aims to target protein kinases and the ubiquitylation system in the development of these therapies. It is one of the largest ever collaborations between the commercial pharmaceutical industry and any academic research institute.
Nicholas Kester Tonks FRS is a professor in Cold Spring Harbor Laboratory. His research is mainly focused on studying the function and regulation of protein tyrosine phosphatases.

Protein phosphatase 1 (PP1) belongs to a certain class of phosphatases known as protein serine/threonine phosphatases. This type of phosphatase includes metal-dependent protein phosphatases (PPMs) and aspartate-based phosphatases. PP1 has been found to be important in the control of glycogen metabolism, muscle contraction, cell progression, neuronal activities, splicing of RNA, mitosis, cell division, apoptosis, protein synthesis, and regulation of membrane receptors and channels.

Vanadyl acetylacetonate is the chemical compound with the formula VO(acac)2, where acac– is the conjugate base of acetylacetone. It is a blue-green solid that dissolves in polar organic solvents. The coordination complex consists of the vanadyl group, VO2+, bound to two acac– ligands via the two oxygen atoms on each. Like other charge-neutral acetylacetonate complexes, it is not soluble in water.
Dario Renato Alessi is a French-born British biochemist, Director of the Medical Research Council Protein Phosphorylation and Ubiquitylation Unit and Professor of Signal Transduction, at the School of Life Sciences, University of Dundee.
David Barford is a British medical researcher and structural biologist at the MRC Laboratory of Molecular Biology Cambridge, UK.
The Portland Press Excellence in Science Award was an annual award instituted in 1964 to recognize notable research in any branch of biochemistry undertaken in the UK or Republic of Ireland. It was initially called the CIBA Medal and Prize, then the Novartis Medal and Prize. The prize consists of a medal and a £3000 cash award. The winner is invited to present a lecture at a Society conference and submit an article to one of the Society's publications. Notable recipients include the Nobel laureates John E. Walker, Paul Nurse, Sydney Brenner, César Milstein, Peter D. Mitchell, Rodney Porter, and John Cornforth.
The School of Life Sciences at the University of Dundee conducts research into the molecular and cellular mechanisms underlying human health and disease.

Helen Walden is an English structural biologist who received the Colworth medal from the Biochemical Society in 2015. She was awarded European Molecular Biology Organization (EMBO) membership in 2022. She is a Professor of Structural Biology at the University of Glasgow and has made significant contributions to the Ubiquitination field.

Claire Eyers is a British biological mass spectrometrist who is professor of biological mass spectrometry at the University of Liverpool, where she heads up the Centre for Proteome Research. Her research publications list her either as Claire E Haydon or Claire E Eyers.
Miratul Muqit FRSE FMedSci is a British neurologist and a Programme Lead at the MRC Protein Phosphorylation and Ubiquitylation Unit (MRCPPU) in the School of Life Sciences at the University of Dundee. His research focuses on the study of the PINK1 gene, mutations in which are a major cause of Parkinson's disease.

Hagit Eldar-Finkelman is an Israeli scientist and a principal investigator of an active research laboratory at the Sackler School of Medicine at Tel Aviv University. Eldar-Finkelman’s research is focused on the signal transduction field and drug development targeting protein kinases. She is well known for her pioneering work on the functions of GSK-3 and its contribution to diabetes and other pathogenies, including depressive behavior, Alzheimer’s diseases, and Huntington’s diseases. Novel findings also include the unique evolution of GSK-3 isozymes. Eldar-Finkelman is a leading figure in developing novel substrate competitive inhibitors (SCIs) for GSK-3 with significant benefits as drug candidates.
References
- ↑ Cohen, P (1979). "The hormonal control of glycogen metabolism in mammalian muscle by multivalent phosphorylation". Biochemical Society Transactions. 7 (3): 459–80. doi:10.1042/bst0070459. PMID 221283.
- ↑ Louis-Jeantet Prize
- ↑ "The Royal Society of Edinburgh Royal Medals" . Retrieved 8 June 2019.
- ↑ Arnold, Carrie (24 March 2009). "Profile of Phillip Cohen". Proceedings of the National Academy of Sciences. 106 (12): 4581–4583. Bibcode:2009PNAS..106.4581A. doi: 10.1073/pnas.0902168106 . ISSN 0027-8424. PMC 2660754 . PMID 19293376.
- ↑ "Philip Cohen". royalsociety.org. Retrieved 25 December 2023.
- ↑ "Principal Investigator | Philip Cohen | MRC PPU". www.ppu.mrc.ac.uk. Retrieved 25 December 2023.
- ↑ "cell.com" (PDF).
- ↑ "Principal Investigator | Philip Cohen". www.ppu.mrc.ac.uk. Retrieved 25 December 2023.
- ↑ "mrc.ukri.org".
- ↑ "Follow the money: Scotland funds a brain gain". Science|Business. Retrieved 25 December 2023.
- ↑ "Sir Philip Cohen, PhD". thevalleefoundation.org. Retrieved 25 December 2023.
- ↑ "Philip Cohen". The Conversation. 31 January 2017. Retrieved 25 December 2023.
- ↑ "Welcome to The Tricia Cohen Memorial Trust". The Tricia Cohen Memorial Trust. Retrieved 25 December 2023.
- ↑ "Excellence in Science Award". www.biochemistry.org. Retrieved 25 December 2023.
- ↑ "The Pfizer Award 1999 | MRC PPU". www.ppu.mrc.ac.uk. 31 October 1999. Retrieved 25 December 2023.
- ↑ admin (17 September 2017). "The Louis-Jeantet Prizes | Jeantet" . Retrieved 25 December 2023.
- ↑ "Sir Philip Cohen, PhD | The Vallee Foundation". thevalleefoundation.org. Retrieved 25 December 2023.
- ↑ "Bristol-Myers Squibb Award | MRC PPU". www.ppu.mrc.ac.uk. 1 October 2002. Retrieved 25 December 2023.
- ↑ "OUH - News archive". www.ous-research.no. Retrieved 25 December 2023.
- ↑ "Debrecen Award for Molecular Medicine 2004 | Faculty of Medicine". aok.unideb.hu. Retrieved 25 December 2023.
- ↑ "Philip Cohen Receives Prestigious Rolf Luft Award in Stockholm, Sweden | MRC PPU". www.ppu.mrc.ac.uk. 3 October 2006. Retrieved 25 December 2023.
- ↑ "Sir Philip Cohen, PhD | The Vallee Foundation". thevalleefoundation.org. Retrieved 25 December 2023.
- ↑ "Lifetime Achievement Award for Professor Sir Philip Cohen | Welcome to Bio-Dundee". www.biodundee.co.uk. Retrieved 25 December 2023.
- ↑ "Professor Sir Philip Cohen Receives MRC Millennium Medal". Proteomics & Metabolomics from Technology Networks. Retrieved 25 December 2023.
- ↑ De Meyts, Pierre (2000), Feingold, Kenneth R.; Anawalt, Bradley; Blackman, Marc R.; Boyce, Alison (eds.), "The Insulin Receptor and Its Signal Transduction Network", Endotext, South Dartmouth (MA): MDText.com, Inc., PMID 27512793 , retrieved 25 December 2023
- ↑ "Philip Cohen". royalsociety.org. Retrieved 25 December 2023.
- ↑ Embi, N.; Rylatt, D. B.; Cohen, P. (June 1980). "Glycogen synthase kinase-3 from rabbit skeletal muscle. Separation from cyclic-AMP-dependent protein kinase and phosphorylase kinase". European Journal of Biochemistry. 107 (2): 519–527. doi: 10.1111/j.1432-1033.1980.tb06059.x . ISSN 0014-2956. PMID 6249596.
- ↑ Alessi, Dario R.; James, Stephen R.; Downes, C.Peter; Holmes, Andrew B.; Gaffney, Piers R.J.; Reese, Colin B.; Cohen, Philip (April 1997). "Characterization of a 3-phosphoinositide-dependent protein kinase which phosphorylates and activates protein kinase Bα". Current Biology. 7 (4): 261–269. doi: 10.1016/s0960-9822(06)00122-9 . ISSN 0960-9822. PMID 9094314.