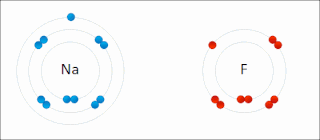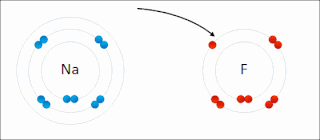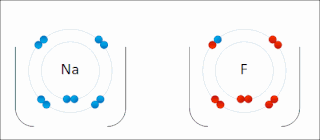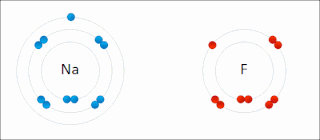
Ionic bonding is a type of chemical bonding that involves the electrostatic attraction between oppositely charged ions, or between two atoms with sharply different electronegativities, and is the primary interaction occurring in ionic compounds. It is one of the main types of bonding, along with covalent bonding and metallic bonding. Ions are atoms with an electrostatic charge. Atoms that gain electrons make negatively charged ions. Atoms that lose electrons make positively charged ions. This transfer of electrons is known as electrovalence in contrast to covalence. In the simplest case, the cation is a metal atom and the anion is a nonmetal atom, but these ions can be more complex, e.g. molecular ions like NH+
4 or SO2−
4. In simpler words, an ionic bond results from the transfer of electrons from a metal to a non-metal to obtain a full valence shell for both atoms.

In chemistry, a salt or ionic compound is a chemical compound consisting of an ionic assembly of positively charged cations and negatively charged anions, which results in a neutral compound with no net electric charge. The constituent ions are held together by electrostatic forces termed ionic bonds.
In organic chemistry, a carbanion is an anion in which carbon is negatively charged.

In organic chemistry, quaternary ammonium cations, also known as quats, are positively-charged polyatomic ions of the structure [NR4]+, where R is an alkyl group, an aryl group or organyl group. Unlike the ammonium ion and the primary, secondary, or tertiary ammonium cations, the quaternary ammonium cations are permanently charged, independent of the pH of their solution. Quaternary ammonium salts or quaternary ammonium compounds are salts of quaternary ammonium cations. Polyquats are a variety of engineered polymer forms which provide multiple quat molecules within a larger molecule.

Potassium superoxide is an inorganic compound with the formula KO2. It is a yellow paramagnetic solid that decomposes in moist air. It is a rare example of a stable salt of the superoxide anion. It is used as a CO2 scrubber, H2O dehumidifier, and O2 generator in rebreathers, spacecraft, submarines, and spacesuits.

Ozonide is the polyatomic anion O−3. Cyclic organic compounds formed by the addition of ozone to an alkene are also called ozonides.
Chemical nomenclature is a set of rules to generate systematic names for chemical compounds. The nomenclature used most frequently worldwide is the one created and developed by the International Union of Pure and Applied Chemistry (IUPAC).
In chemical nomenclature, the IUPAC nomenclature of inorganic chemistry is a systematic method of naming inorganic chemical compounds, as recommended by the International Union of Pure and Applied Chemistry (IUPAC). It is published in Nomenclature of Inorganic Chemistry. Ideally, every inorganic compound should have a name from which an unambiguous formula can be determined. There is also an IUPAC nomenclature of organic chemistry.

Tetramethylammonium hydroxide (TMAH or TMAOH) is a quaternary ammonium salt with molecular formula N(CH3)4+ OH−. It is commonly encountered in form of concentrated solutions in water or methanol. TMAH in solid state and its aqueous solutions are all colorless, but may be yellowish if impure. Although TMAH has virtually no odor when pure, samples often have a strong fishy smell due to presence of trimethylamine which is a common impurity. TMAH has several diverse industrial and research applications.

Gold compounds are compounds by the element gold (Au). Although gold is the most noble of the noble metals, it still forms many diverse compounds. The oxidation state of gold in its compounds ranges from −1 to +5, but Au(I) and Au(III) dominate its chemistry. Au(I), referred to as the aurous ion, is the most common oxidation state with soft ligands such as thioethers, thiolates, and organophosphines. Au(I) compounds are typically linear. A good example is Au(CN)−2, which is the soluble form of gold encountered in mining. The binary gold halides, such as AuCl, form zigzag polymeric chains, again featuring linear coordination at Au. Most drugs based on gold are Au(I) derivatives.
An alkalide is a chemical compound in which alkali metal atoms are anions with a charge or oxidation state of −1. Until the first discovery of alkalides in the 1970s, alkali metals were known to appear in salts only as cations with a charge or oxidation state of +1. These types of compounds are of theoretical interest due to their unusual stoichiometry and low ionization potentials. Alkalide compounds are chemically related to the electrides, salts in which trapped electrons are effectively the anions.
Arsenic trifluoride is a chemical compound of arsenic and fluorine with the chemical formula AsF3. It is a colorless liquid which reacts readily with water.
In condensed matter physics and inorganic chemistry, the cation-anion radius ratio can be used to predict the crystal structure of an ionic compound based on the relative size of its atoms. It is defined as the ratio of the ionic radius of the positively charged cation to the ionic radius of the negatively charged anion in a cation-anion compound. Anions are larger than cations. Large sized anions occupy lattice sites, while small sized cations are found in voids. In a given structure, the ratio of cation radius to anion radius is called the radius ratio. This is simply given by .
Tetramethylammonium pentafluoroxenate is a chemical compound with the chemical formula [N(CH3)4]+[XeF5]−. This salt consists of tetramethylammonium cations [N(CH3)4]+ and pentafluoroxenate(IV) anions [XeF5]−. The [XeF5]− ion was the first example of a pentagonal planar molecular geometry AX5E2 species. It was prepared by the reaction of [N(CH3)4]F with xenon tetrafluoride, [N(CH3)4]F being chosen because it can be prepared in anhydrous form and is readily soluble in organic solvents. The anion is planar, with the fluorine atoms in a slightly distorted pentagonal coordination. Other salts have been prepared with sodium, caesium and rubidium, and vibrational spectra show that these contain the same planar ion. The isolated anion has the point group of D5h.

An ion is an atom or molecule with a net electrical charge. The charge of an electron is considered to be negative by convention and this charge is equal and opposite to the charge of a proton, which is considered to be positive by convention. The net charge of an ion is not zero because its total number of electrons is unequal to its total number of protons.

A polonide is a chemical compound of the radioactive element polonium with any element less electronegative than polonium. Polonides are usually prepared by a direct reaction between the elements at temperatures of around 300–400 °C. They are amongst the most chemically stable compounds of polonium, and can be divided into two broad groups:

Caesium auride is the inorganic compound with the formula CsAu. It is the Cs+ salt of the unusual Au− anion.

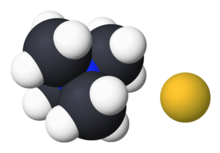
Tetramethylammonium (TMA) is the simplest quaternary ammonium cation. It has the chemical formula [Me4N]+ and consists of four methyl groups attached to a central nitrogen atom. The cation is isoelectronic with neopentane. It is positively-charged and can only be isolated in association with a counter-ion. Common salts include tetramethylammonium chloride and tetramethylammonium hydroxide. Tetramethylammonium salts are used in chemical synthesis and in pharmacological research. It confers no color to its salts.
Tetranitratoborate is an anion composed of boron with four nitrate groups. It has formula [B(NO3)4]−. It can form salts with large cations such as tetramethylammonium nitratoborate, or tetraethylammonium tetranitratoborate. The ion was first discovered by C. R. Guibert and M. D. Marshall in 1966 after failed attempts to make neutral (non-ionic) boron nitrate, B(NO3)3, which has resisted attempts to make it; if it exists, it is unstable above −78 °C.
The nitronickelates are a class of chemical compounds containing a nickel atom complexed by nitro groups, -NO2. Nickel can be in a +2 or +3 oxidation state. There can be five (pentanitronickelates), or six, (hexanitronickelates) nitro groups per nickel atom. They can be considered the double nitrites of nickel nitrite.