Related Research Articles

The design of experiments, also known as experiment design or experimental design, is the design of any task that aims to describe and explain the variation of information under conditions that are hypothesized to reflect the variation. The term is generally associated with experiments in which the design introduces conditions that directly affect the variation, but may also refer to the design of quasi-experiments, in which natural conditions that influence the variation are selected for observation.

An experiment is a procedure carried out to support or refute a hypothesis, or determine the efficacy or likelihood of something previously untried. Experiments provide insight into cause-and-effect by demonstrating what outcome occurs when a particular factor is manipulated. Experiments vary greatly in goal and scale but always rely on repeatable procedure and logical analysis of the results. There also exist natural experimental studies.

A placebo can be roughly defined as a sham medical treatment. Common placebos include inert tablets, inert injections, sham surgery, and other procedures.

A randomized controlled trial is a form of scientific experiment used to control factors not under direct experimental control. Examples of RCTs are clinical trials that compare the effects of drugs, surgical techniques, medical devices, diagnostic procedures, diets or other medical treatments.
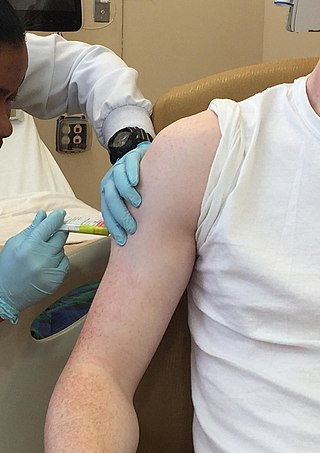
Clinical trials are prospective biomedical or behavioral research studies on human participants designed to answer specific questions about biomedical or behavioral interventions, including new treatments and known interventions that warrant further study and comparison. Clinical trials generate data on dosage, safety and efficacy. They are conducted only after they have received health authority/ethics committee approval in the country where approval of the therapy is sought. These authorities are responsible for vetting the risk/benefit ratio of the trial—their approval does not mean the therapy is 'safe' or effective, only that the trial may be conducted.
In a blind or blinded experiment, information which may influence the participants of the experiment is withheld until after the experiment is complete. Good blinding can reduce or eliminate experimental biases that arise from a participants' expectations, observer's effect on the participants, observer bias, confirmation bias, and other sources. A blind can be imposed on any participant of an experiment, including subjects, researchers, technicians, data analysts, and evaluators. In some cases, while blinding would be useful, it is impossible or unethical. For example, it is not possible to blind a patient to their treatment in a physical therapy intervention. A good clinical protocol ensures that blinding is as effective as possible within ethical and practical constraints.
Antihypertensives are a class of drugs that are used to treat hypertension. Antihypertensive therapy seeks to prevent the complications of high blood pressure, such as stroke, heart failure, kidney failure and myocardial infarction. Evidence suggests that reduction of the blood pressure by 5 mmHg can decrease the risk of stroke by 34% and of ischaemic heart disease by 21%, and can reduce the likelihood of dementia, heart failure, and mortality from cardiovascular disease. There are many classes of antihypertensives, which lower blood pressure by different means. Among the most important and most widely used medications are thiazide diuretics, calcium channel blockers, ACE inhibitors, angiotensin II receptor antagonists (ARBs), and beta blockers.
A nocebo effect is said to occur when negative expectations of the patient regarding a treatment cause the treatment to have a more negative effect than it otherwise would have. For example, when a patient anticipates a side effect of a medication, they can experience that effect even if the "medication" is actually an inert substance. The complementary concept, the placebo effect, is said to occur when positive expectations improve an outcome. The nocebo effect is also said to occur in someone who falls ill owing to the erroneous belief that they were exposed to a toxin, e.g. a physical phenomenon they believe is harmful, such as EM radiation.
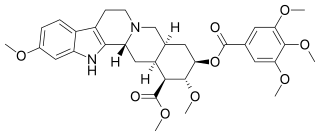
Reserpine is a drug that is used for the treatment of high blood pressure, usually in combination with a thiazide diuretic or vasodilator. Large clinical trials have shown that combined treatment with reserpine plus a thiazide diuretic reduces mortality of people with hypertension. Although the use of reserpine as a solo drug has declined since it was first approved by the FDA in 1955, the combined use of reserpine and a thiazide diuretic or vasodilator is still recommended in patients who do not achieve adequate lowering of blood pressure with first-line drug treatment alone. The reserpine-hydrochlorothiazide combo pill was the 17th most commonly prescribed of the 43 combination antihypertensive pills available In 2012.

A scientific control is an experiment or observation designed to minimize the effects of variables other than the independent variable. This increases the reliability of the results, often through a comparison between control measurements and the other measurements. Scientific controls are a part of the scientific method.

The number needed to treat (NNT) or number needed to treat for an additional beneficial outcome (NNTB) is an epidemiological measure used in communicating the effectiveness of a health-care intervention, typically a treatment with medication. The NNT is the average number of patients who need to be treated to prevent one additional bad outcome. It is defined as the inverse of the absolute risk reduction, and computed as , where is the incidence in the control (unexposed) group, and is the incidence in the treated (exposed) group. This calculation implicitly assumes monotonicity, that is, no individual can be harmed by treatment. The modern approach, based on counterfactual conditionals, relaxes this assumption and yields bounds on NNT.
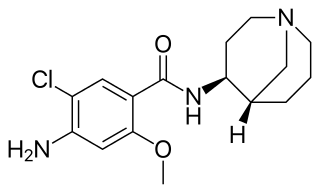
Renzapride is a prokinetic agent and antiemetic which acts as a full 5-HT4 agonist and partial 5-HT3 antagonist. It also functions as a 5-HT2B antagonist and has some affinity for the 5-HT2A and 5-HT2C receptors.
Random assignment or random placement is an experimental technique for assigning human participants or animal subjects to different groups in an experiment using randomization, such as by a chance procedure or a random number generator. This ensures that each participant or subject has an equal chance of being placed in any group. Random assignment of participants helps to ensure that any differences between and within the groups are not systematic at the outset of the experiment. Thus, any differences between groups recorded at the end of the experiment can be more confidently attributed to the experimental procedures or treatment.
Clinical study design is the formulation of trials and experiments, as well as observational studies in medical, clinical and other types of research involving human beings. The goal of a clinical study is to assess the safety, efficacy, and / or the mechanism of action of an investigational medicinal product (IMP) or procedure, or new drug or device that is in development, but potentially not yet approved by a health authority. It can also be to investigate a drug, device or procedure that has already been approved but is still in need of further investigation, typically with respect to long-term effects or cost-effectiveness.
In medicine, a crossover study or crossover trial is a longitudinal study in which subjects receive a sequence of different treatments. While crossover studies can be observational studies, many important crossover studies are controlled experiments, which are discussed in this article. Crossover designs are common for experiments in many scientific disciplines, for example psychology, pharmaceutical science, and medicine.

In fields such as epidemiology, social sciences, psychology and statistics, an observational study draws inferences from a sample to a population where the independent variable is not under the control of the researcher because of ethical concerns or logistical constraints. One common observational study is about the possible effect of a treatment on subjects, where the assignment of subjects into a treated group versus a control group is outside the control of the investigator. This is in contrast with experiments, such as randomized controlled trials, where each subject is randomly assigned to a treated group or a control group. Observational studies, for lacking an assignment mechanism, naturally present difficulties for inferential analysis.
A glossary of terms used in clinical research.

Placebo-controlled studies are a way of testing a medical therapy in which, in addition to a group of subjects that receives the treatment to be evaluated, a separate control group receives a sham "placebo" treatment which is specifically designed to have no real effect. Placebos are most commonly used in blinded trials, where subjects do not know whether they are receiving real or placebo treatment. Often, there is also a further "natural history" group that does not receive any treatment at all.

Ecopipam is a dopamine antagonist which is under development for the treatment of Lesch-Nyhan syndrome, Tourette's syndrome, speech disorders, and restless legs syndrome. It is taken by mouth.

Finerenone, sold under the brand name Kerendia and Firialta, is a medication used to reduce the risk of kidney function decline, kidney failure, cardiovascular death, non-fatal heart attacks, and hospitalization for heart failure in adults with chronic kidney disease associated with type 2 diabetes. Finerenone is a non-steroidal mineralocorticoid receptor antagonist (MRA). It is taken orally.
References
- 1 2 Hinkelmann, Klaus; Kempthorne, Oscar (2008). Design and Analysis of Experiments, Volume I: Introduction to Experimental Design (2nd ed.). Wiley. ISBN 978-0-471-72756-9. MR 2363107.
- 1 2 Bailey, R. A. (2008). Design of comparative experiments. Cambridge University Press. ISBN 978-0-521-68357-9. MR 2422352.
- 1 2 Seeley, Ellen W.; Grinspoon, Steven - Harvard Medical School (25 November 2016). "Chapter 2: Patient-Oriented Research". In David Robertson; Gordon H. Williams (eds.). Clinical and Translational Science: Principles of Human Research. Academic Press. p. 13, parag. 3 under "Clinical Trials". ISBN 978-0-12-802111-8 . Retrieved 2018-07-30.
- 1 2 Chaplin S (2006). "The placebo response: an important part of treatment". Prescriber. 17 (5): 16–22. doi: 10.1002/psb.344 . S2CID 72626022.
- 1 2 Everitt, B.S. (2002) The Cambridge Dictionary of Statistics, CUP. ISBN 0-521-81099-X (entry for control group)
- ↑ Neyman, Jerzy (1990) [1923], Dabrowska, Dorota M.; Speed, Terence P. (eds.), "On the application of probability theory to agricultural experiments: Essay on principles (Section 9)", Statistical Science, 5 (4): 465–472, doi: 10.1214/ss/1177012031 , MR 1092986
- ↑ Ian Hacking (September 1988). "Telepathy: Origins of Randomization in Experimental Design". Isis . 79 (3): 427–451. doi:10.1086/354775. S2CID 52201011.
- ↑ Stephen M. Stigler (November 1992). "A Historical View of Statistical Concepts in Psychology and Educational Research". American Journal of Education. 101 (1): 60–70. doi:10.1086/444032. S2CID 143685203.
- ↑ Trudy Dehue (December 1997). "Deception, Efficiency, and Random Groups: Psychology and the Gradual Origination of the Random Group Design" (PDF). Isis . 88 (4): 653–673. doi:10.1086/383850. PMID 9519574. S2CID 23526321.
- ↑ Tight Blood Pressure Control and Risk of Macrovascular and Microvascular Complications inType 2 Diabetes
- ↑ The ALLHAT Study