Related Research Articles

The United Kingdom Atomic Energy Authority is a UK government research organisation responsible for the development of nuclear fusion power. It is an executive non-departmental public body of the Department for Business, Energy and Industrial Strategy (BEIS).

The Defence Science and Technology Laboratory (Dstl) is an executive agency of the Ministry of Defence of the United Kingdom. Its stated purpose is "to maximise the impact of science and technology for the defence and security of the UK". The agency is headed by Doug Umbers as its (interim) Chief Executive, with the board being chaired by Adrian Belton. Ministerial responsibility lies with the Minister for Defence Procurement.

The Vaccine and Infectious Disease Organization – International Vaccine Centre (VIDO-InterVac) is a research organization of the University of Saskatchewan that operates with financial support from the Government of Canada, the government of Saskatchewan, livestock industry councils and agencies, foundations and human and animal health companies. In addition to the 2,500,000 sq ft (230,000 m2) facility on campus, VIDO-InterVac also operates a 160-acre (0.6 km2) research station.
Serum Institute of India (SII) is an Indian biotechnology and biopharmaceuticals company. It is the world's largest manufacturer of vaccines located in the city of Pune, India and was founded by Cyrus Poonawalla in 1966. The company is a subsidiary of the holding company Poonawalla Investment and Industries.

The Biomedical Advanced Research and Development Authority(BARDA) is a U.S. Department of Health and Human Services (HHS) office responsible for the procurement and development of medical countermeasures, principally against bioterrorism, including chemical, biological, radiological and nuclear (CBRN) threats, as well as pandemic influenza and emerging diseases. BARDA was established in 2006 through the Pandemic and All-Hazards Preparedness Act (PAHPA) and reports to the Office of the Assistant Secretary for Preparedness and Response (ASPR). The office manages Project BioShield, which funds the research, development and stockpiling of vaccines and treatments that the government could use during public health emergencies such as chemical, biological, radiological or nuclear (CBRN) attacks.
Adrian Vivian Sinton Hill, is an Irish vaccinologist, Director of the Jenner Institute and Professor of Vaccinology and of Human Genetics at the University of Oxford, an honorary Consultant Physician in Infectious Diseases, and Fellow of Magdalen College, Oxford. Hill is a leader in the field of malaria vaccine development and was a co-leader of the research team which produced the Oxford–AstraZeneca COVID-19 vaccine, along with Professor Sarah Gilbert of the Jenner Institute and Professor Andrew Pollard of the Oxford Vaccine Group.
The Coalition for Epidemic Preparedness Innovations (CEPI) is a foundation that takes donations from public, private, philanthropic, and civil society organisations, to finance independent research projects to develop vaccines against emerging infectious diseases (EID).
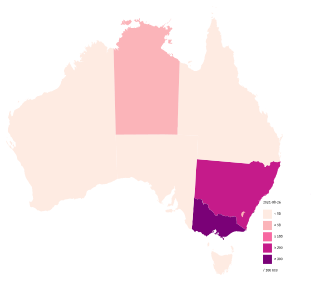
The COVID-19 pandemic in Australia is part of the ongoing worldwide pandemic of the coronavirus disease 2019 caused by severe acute respiratory syndrome coronavirus 2. The first confirmed case in Australia was identified on 25 January 2020, in Victoria, when a man who had returned from Wuhan, Hubei, China, tested positive for the virus.

A COVID‑19 vaccine is a vaccine intended to provide acquired immunity against severe acute respiratory syndrome coronavirus 2 (SARS‑CoV‑2), the virus that causes coronavirus disease 2019 (COVID‑19). Prior to the COVID‑19 pandemic, an established body of knowledge existed about the structure and function of coronaviruses causing diseases like severe acute respiratory syndrome (SARS) and Middle East respiratory syndrome (MERS). This knowledge accelerated the development of various vaccine platforms during early 2020. The initial focus of SARS-CoV-2 vaccines was on preventing symptomatic, often severe illness. On 10 January 2020, the SARS-CoV-2 genetic sequence data was shared through GISAID, and by 19 March, the global pharmaceutical industry announced a major commitment to address COVID-19. The COVID‑19 vaccines are widely credited for their role in reducing the spread, severity, and death caused by COVID-19.

Operation Warp Speed (OWS) was a public–private partnership initiated by the United States government to facilitate and accelerate the development, manufacturing, and distribution of COVID-19 vaccines, therapeutics, and diagnostics. The first news report of Operation Warp Speed was on April 29, 2020, and the program was officially announced on May 15, 2020. It was headed by Moncef Slaoui from May 2020 to January 2021 and by David A. Kessler from January to February 2021. At the end of February 2021, Operation Warp Speed was transferred into the responsibilities of the White House COVID-19 Response Team.

The Oxford–AstraZeneca COVID-19 vaccine, codenamed AZD1222, and sold under the brand names Covishield and Vaxzevria among others, is a viral vector vaccine for prevention of COVID-19. Developed by Oxford University and AstraZeneca, it is given by intramuscular injection, using as a vector the modified chimpanzee adenovirus ChAdOx1. Studies carried out in 2020 showed that the efficacy of the vaccine is 76.0% at preventing symptomatic COVID-19 beginning at 22 days following the first dose and 81.3% after the second dose. A study in Scotland found that, for symptomatic COVID-19 infection after the second dose, the vaccine is 81% effective against the Alpha variant, and 61% against the Delta variant.

BioNTech SE is a German biotechnology company based in Mainz that develops and manufactures active immunotherapies for patient-specific approaches to the treatment of diseases. It develops pharmaceutical candidates based on messenger ribonucleic acid (mRNA) for use as individualized cancer immunotherapies, as vaccines against infectious diseases and as protein replacement therapies for rare diseases, and also engineered cell therapy, novel antibodies and small molecule immunomodulators as treatment options for cancer.
The Vaccine Taskforce in the United Kingdom of Great Britain and Northern Ireland was set up in May 2020 by the Second Johnson ministry, in collaboration with Chief Scientific Advisor Patrick Vallance and Chief Medical Officer Professor Chris Whitty, in order to facilitate the path towards the introduction of a COVID-19 vaccine in the UK and its global distribution. The taskforce acts to coordinate the research efforts of government with industry, academics and funding agencies in order to be able to make timely decisions which expedite vaccine development and deployment.
COVID-19 Vaccines Global Access, abbreviated as COVAX, is a worldwide initiative aimed at equitable access to COVID-19 vaccines directed by Gavi, the Vaccine Alliance, the Coalition for Epidemic Preparedness Innovations (CEPI), and the World Health Organization (WHO). It is one of the three pillars of the Access to COVID-19 Tools Accelerator, an initiative begun in April 2020 by the WHO, the European Commission, and the government of France as a response to the COVID-19 pandemic. COVAX coordinates international resources to enable low-to-middle-income countries equitable access to COVID-19 tests, therapies, and vaccines. By 15 July 2020, 165 countries – representing 60% of the human population – had joined COVAX.

The COVID-19 vaccination programme in the United Kingdom is an ongoing mass immunisation campaign for COVID-19 during the pandemic in the United Kingdom. The UK's vaccination rollout was the world's first mass immunisation programme for the disease when it began on 8 December 2020 after Margaret Keenan received her first dose of two. As of 22 August 2021, 47,690,741 first doses and 41,810,753 second doses of a vaccine had been administered across the UK.

Valneva COVID-19 vaccine, also known as the VLA2001 and VLA2101, is a COVID-19 vaccine candidate developed by French biotechnology company Valneva SE in collaboration with American company Dynavax Technologies.

The Novavax COVID-19 vaccine, codenamed NVX-CoV2373, is a subunit COVID-19 vaccine candidate developed by Novavax and the Coalition for Epidemic Preparedness Innovations (CEPI), that is undergoing trials in India under the brand name Covovax. It requires two doses and is stable at 2 to 8 °C refrigerated temperatures.
The COVID-19 vaccination program in the Philippines is an ongoing mass immunization campaign against severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2), the virus that causes coronavirus disease 2019 (COVID-19), in response to the ongoing pandemic in the country.

As of 23 August 2021, 5 billion COVID-19 vaccine doses had been administered worldwide based on official reports from national health agencies collated by Our World in Data.

A dispute broke out in January 2021 between the European Commission and the pharmaceutical company AstraZeneca AB about the provision of COVID-19 vaccines during the COVID-19 pandemic, and, in February, spilled out into a dispute over Article 16 of the Northern Ireland Protocol. Vaccination proceeded apace in the UK but more slowly in the EU, and by the end of March 2021, over 30% of the UK population had received at least one dose of vaccine compared to about 8% of the EU population. This was partly due to limited availability of the AstraZeneca vaccine in the EU. The World Health Organization and the European Medicines Agency continued to state that the vaccine was safe and effective. However, a representative of the European Medicines Agency said in June that vaccines based on the mRNA technology should be preferred if available for all age groups, including for the over 60s.
References
- ↑ "New vaccines centre to protect UK from pandemic threats". University of Oxford. 3 December 2018. Retrieved 22 March 2021.
- 1 2 "Vaccines Manufacturing Innovation Centre, Oxfordshire, UK". www.pharmaceutical-technology.com. Retrieved 17 January 2021.
- ↑ "Vaccines Manufacturing and Innovation Centre to open 12 months ahead of schedule". GOV.UK. Retrieved 17 January 2021.
- ↑ "Government invests £93 million in new vaccine-manufacturing facility". ITV News. 17 May 2020. Retrieved 17 January 2021.
- ↑ "Extra £47.6 million for Vaccines Manufacturing and Innovation Centre". GOV.UK. 20 March 2021. Retrieved 21 March 2021.
- ↑ Weinfass, Ian (21 April 2020). "Vaccine-production facility build fast-tracked". Construction News. Retrieved 22 March 2021.
- ↑ "2020 Review". VMIC. 21 December 2020. Retrieved 22 March 2021.