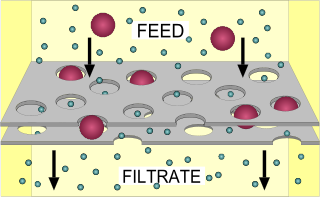
Filtration is a physical separation process that separates solid matter and fluid from a mixture using a filter medium that has a complex structure through which only the fluid can pass. Solid particles that cannot pass through the filter medium are described as oversize and the fluid that passes through is called the filtrate. Oversize particles may form a filter cake on top of the filter and may also block the filter lattice, preventing the fluid phase from crossing the filter, known as blinding. The size of the largest particles that can successfully pass through a filter is called the effective pore size of that filter. The separation of solid and fluid is imperfect; solids will be contaminated with some fluid and filtrate will contain fine particles. Filtration occurs both in nature and in engineered systems; there are biological, geological, and industrial forms.
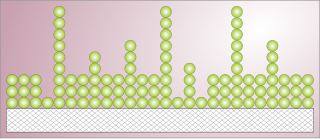
Adsorption is the adhesion of atoms, ions or molecules from a gas, liquid or dissolved solid to a surface. This process creates a film of the adsorbate on the surface of the adsorbent. This process differs from absorption, in which a fluid is dissolved by or permeates a liquid or solid. While adsorption does often precede absorption, which involves the transfer of the absorbate into the volume of the absorbent material, alternatively, adsorption is distinctly a surface phenomenon, wherein the adsorbate does not penetrate through the material surface and into the bulk of the adsorbent. The term sorption encompasses both adsorption and absorption, and desorption is the reverse of sorption.

Water purification is the process of removing undesirable chemicals, biological contaminants, suspended solids, and gases from water. The goal is to produce water that is fit for specific purposes. Most water is purified and disinfected for human consumption, but water purification may also be carried out for a variety of other purposes, including medical, pharmacological, chemical, and industrial applications. The history of water purification includes a wide variety of methods. The methods used include physical processes such as filtration, sedimentation, and distillation; biological processes such as slow sand filters or biologically active carbon; chemical processes such as flocculation and chlorination; and the use of electromagnetic radiation such as ultraviolet light.

Silica gel is an amorphous and porous form of silicon dioxide (silica), consisting of an irregular tridimensional framework of alternating silicon and oxygen atoms with nanometer-scale voids and pores. The voids may contain water or some other liquids, or may be filled by gas or vacuum. In the last case, the material is properly called silica xerogel.

Water treatment is any process that improves the quality of water to make it appropriate for a specific end-use. The end use may be drinking, industrial water supply, irrigation, river flow maintenance, water recreation or many other uses, including being safely returned to the environment. Water treatment removes contaminants and undesirable components, or reduces their concentration so that the water becomes fit for its desired end-use. This treatment is crucial to human health and allows humans to benefit from both drinking and irrigation use.
The Bayer process is the principal industrial means of refining bauxite to produce alumina (aluminium oxide) and was developed by Carl Josef Bayer. Bauxite, the most important ore of aluminium, contains only 30–60% aluminium oxide (Al2O3), the rest being a mixture of silica, various iron oxides, and titanium dioxide. The aluminium oxide must be further purified before it can be refined into aluminium.

Activated carbon, also called activated charcoal, is a form of carbon commonly used to filter contaminants from water and air, among many other uses. It is processed (activated) to have small, low-volume pores that greatly increase the surface area available for adsorption or chemical reactions that can be thought of as a microscopic "sponge" structure.. Activation is analogous to making popcorn from dried corn kernels: popcorn is light, fluffy, and its kernels have a high surface-area-to-volume ratio. Activated is sometimes replaced by active.

A diving air compressor is a breathing air compressor that can provide breathing air directly to a surface-supplied diver, or fill diving cylinders with high-pressure air pure enough to be used as a hyperbaric breathing gas. A low pressure diving air compressor usually has a delivery pressure of up to 30 bar, which is regulated to suit the depth of the dive. A high pressure diving compressor has a delivery pressure which is usually over 150 bar, and is commonly between 200 and 300 bar. The pressure is limited by an overpressure valve which may be adjustable.

Bone char is a porous, black, granular material produced by charring animal bones. Its composition varies depending on how it is made; however, it consists mainly of tricalcium phosphate 57–80%, calcium carbonate 6–10% and carbon 7–10%. It is primarily used for filtration and decolorisation.

Heterogeneous catalysis is catalysis where the phase of catalysts differs from that of the reactants or products. The process contrasts with homogeneous catalysis where the reactants, products and catalyst exist in the same phase. Phase distinguishes between not only solid, liquid, and gas components, but also immiscible mixtures, or anywhere an interface is present.

Montmorillonite is a very soft phyllosilicate group of minerals that form when they precipitate from water solution as microscopic crystals, known as clay. It is named after Montmorillon in France. Montmorillonite, a member of the smectite group, is a 2:1 clay, meaning that it has two tetrahedral sheets of silica sandwiching a central octahedral sheet of alumina. The particles are plate-shaped with an average diameter around 1 μm and a thickness of 0.96 nm; magnification of about 25,000 times, using an electron microscope, is required to resolve individual clay particles. Members of this group include saponite, nontronite, beidellite, and hectorite.

A molecular sieve is a material with pores of uniform size. These pore diameters are similar in size to small molecules, and thus large molecules cannot enter or be adsorbed, while smaller molecules can. As a mixture of molecules migrates through the stationary bed of porous, semi-solid substance referred to as a sieve, the components of the highest molecular weight leave the bed first, followed by successively smaller molecules. Some molecular sieves are used in size-exclusion chromatography, a separation technique that sorts molecules based on their size. Another important use is as a desiccant. Most of molecular sieves are aluminosilicate zeolites with Si/Al molar ratio less than 2, but there are also examples of activated charcoal and silica gel.

Pressure swing adsorption (PSA) is a technique used to separate some gas species from a mixture of gases under pressure according to the species' molecular characteristics and affinity for an adsorbent material. It operates at near-ambient temperature and significantly differs from the cryogenic distillation commonly used to separate gases. Selective adsorbent materials are used as trapping material, preferentially adsorbing the target gas species at high pressure. The process then swings to low pressure to desorb the adsorbed gas.

Carbon filtering is a method of filtering that uses a bed of activated carbon to remove impurities from a fluid using adsorption.

Arsenic contamination of groundwater is a form of groundwater pollution which is often due to naturally occurring high concentrations of arsenic in deeper levels of groundwater. It is a high-profile problem due to the use of deep tube wells for water supply in the Ganges Delta, causing serious arsenic poisoning to large numbers of people. A 2007 study found that over 137 million people in more than 70 countries are probably affected by arsenic poisoning of drinking water. The problem became a serious health concern after mass poisoning of water in Bangladesh. Arsenic contamination of ground water is found in many countries throughout the world, including the US.
Iron oxide adsorption is a water treatment process that is used to remove arsenic from drinking water. Arsenic is a common natural contaminant of well water and is highly carcinogenic. Iron oxide adsorption treatment for arsenic in groundwater is a commonly practiced removal process which involves the chemical treatment of arsenic species such that they adsorb onto iron oxides and create larger particles that may be filtered out of the water stream.
Kimberlite tailings are waste materials left after the recovery of diamond from diamond source rock. The material has a particle size ranging from 20 mm to 75 micrometres and can be categorized as a soft aggregate. It contains mainly silica, alumina, iron oxide, and magnesia.

Nanosized aluminium oxide occurs in the form of spherical or nearly spherical nanoparticles, and in the form of oriented or undirected fibers.
Compressed air dryers are special types of filter systems that are specifically designed to remove the water that is inherent in compressed air. The compression of air raises its temperature and concentrates atmospheric contaminants, primarily water vapor, as resulting in air with elevated temperature and 100% relative humidity. As the compressed air cools down, water vapor condenses into the tank(s), pipes, hoses and tools connected downstream from the compressor which may be damaging. Therefore water vapor is removed from compressed air to prevent condensation from occurring and to prevent moisture from interfering in sensitive industrial processes.
Defluoridation is the downward adjustment of the level of fluoride in drinking water. Worldwide, fluoride is one of the most abundant anions present in groundwater. Fluoride is more present in groundwater than surface water mainly due to the leaching of minerals. Groundwater accounts for 98 percent of the earth's potable water. An excess of fluoride in drinking water causes dental fluorosis and skeletal fluorosis. The World Health Organization has recommended a guideline value of 1.5 mg/L as the concentration above which dental fluorosis is likely. Fluorosis is endemic in more than 20 developed and developing nations.















