Related Research Articles

An electrode is an electrical conductor used to make contact with a nonmetallic part of a circuit. Electrodes are essential parts of batteries that can consist of a variety of materials depending on the type of battery.

A lithium-ion or Li-ion battery is a type of rechargeable battery that uses the reversible intercalation of Li+ ions into electronically conducting solids to store energy. In comparison with other commercial rechargeable batteries, Li-ion batteries are characterized by higher specific energy, higher energy density, higher energy efficiency, a longer cycle life, and a longer calendar life. Also noteworthy is a dramatic improvement in lithium-ion battery properties after their market introduction in 1991: within the next 30 years, their volumetric energy density increased threefold while their cost dropped tenfold.

Nanobatteries are fabricated batteries employing technology at the nanoscale, particles that measure less than 100 nanometers or 10−7 meters. These batteries may be nano in size or may use nanotechnology in a macro scale battery. Nanoscale batteries can be combined to function as a macrobattery such as within a nanopore battery.
A nanowire battery uses nanowires to increase the surface area of one or both of its electrodes, which improves the capacity of the battery. Some designs, variations of the lithium-ion battery have been announced, although none are commercially available. All of the concepts replace the traditional graphite anode and could improve battery performance. Each type of nanowire battery has specific advantages and disadvantages, but a challenge common to all of them is their fragility.
Rechargeable lithium metal batteries are secondary lithium metal batteries. They have metallic lithium as a negative electrode, sometimes referred to as the battery anode. The high specific capacity of lithium metal, very low redox potential and low density make it the ideal anode material for high energy density battery technologies. Rechargeable lithium metal batteries can have a long run time due to the high charge density of lithium. Several companies and many academic research groups are currently researching and developing rechargeable lithium metal batteries as they are considered a leading pathway for development beyond lithium-ion batteries. Some rechargeable lithium metal batteries employ a liquid electrolyte and some employ a solid-state electrolyte.

The lithium–sulfur battery is a type of rechargeable battery. It is notable for its high specific energy. The low atomic weight of lithium and moderate atomic weight of sulfur means that Li–S batteries are relatively light. They were used on the longest and highest-altitude unmanned solar-powered aeroplane flight by Zephyr 6 in August 2008.
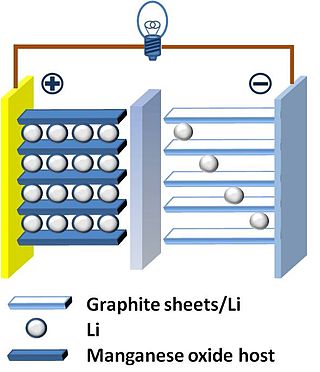
The thin film lithium-ion battery is a form of solid-state battery. Its development is motivated by the prospect of combining the advantages of solid-state batteries with the advantages of thin-film manufacturing processes.

A solid-state battery uses solid electrodes and a solid electrolyte, instead of the liquid or polymer gel electrolytes found in lithium-ion or lithium polymer batteries.
The lithium–air battery (Li–air) is a metal–air electrochemical cell or battery chemistry that uses oxidation of lithium at the anode and reduction of oxygen at the cathode to induce a current flow.
A potassium-ion battery or K-ion battery is a type of battery and analogue to lithium-ion batteries, using potassium ions for charge transfer instead of lithium ions. It was invented by the Iranian/American chemist Ali Eftekhari in 2004.

Sodium-ion batteries (NIBs, SIBs, or Na-ion batteries) are several types of rechargeable batteries, which use sodium ions (Na+) as its charge carriers. In some cases, its working principle and cell construction are similar to those of lithium-ion battery (LIB) types, but it replaces lithium with sodium as the intercalating ion. Sodium belongs to the same group in the periodic table as lithium and thus has similar chemical properties. Although, in some cases (such as aqueous Na-ion batteries) they are quite different from Li-ion batteries.

Sir Peter George Bruce, is a British chemist, and Wolfson Professor of Materials in the Department of Materials at the University of Oxford. Between 2018 and 2023, he served as Physical Secretary and Vice President of the Royal Society. Bruce is a founder and Chief Scientist of the Faraday Institution.
Aluminium-ion batteries are a class of rechargeable battery in which aluminium ions serve as charge carriers. Aluminium can exchange three electrons per ion. This means that insertion of one Al3+ is equivalent to three Li+ ions. Thus, since the ionic radii of Al3+ (0.54 Å) and Li+ (0.76 Å) are similar, significantly higher numbers of electrons and Al3+ ions can be accepted by cathodes with little damage. Al has 50 times (23.5 megawatt-hours m-3) the energy density of Li and is even higher than coal.
Research in lithium-ion batteries has produced many proposed refinements of lithium-ion batteries. Areas of research interest have focused on improving energy density, safety, rate capability, cycle durability, flexibility, and cost.
A dual carbon battery is a type of battery that uses graphite as both its cathode and anode material. Compared to lithium-ion batteries, dual-ion batteries (DIBs) require less energy and emit less CO2 during production, have a reduced reliance on critical materials such as Ni or Co, and are more easily recyclable.
Magnesium batteries are batteries that utilize magnesium cations as charge carriers and possibly in the anode in electrochemical cells. Both non-rechargeable primary cell and rechargeable secondary cell chemistries have been investigated. Magnesium primary cell batteries have been commercialised and have found use as reserve and general use batteries.
The glass battery is a type of solid-state battery. It uses a glass electrolyte and lithium or sodium metal electrodes. The battery was invented by John B. Goodenough, inventor of the lithium cobalt oxide and lithium iron phosphate electrode materials used in the lithium-ion battery (Li-ion), and Maria H. Braga, an associate professor at the University of Porto and a senior research fellow at Cockrell School of Engineering at The University of Texas.
Calcium (ion) batteries are energy storage and delivery technologies (i.e., electro–chemical energy storage) that employ calcium ions (cations), Ca2+, as the active charge carrier. Calcium (ion) batteries remain an active area of research, with studies and work persisting in the discovery and development of electrodes and electrolytes that enable stable, long-term battery operation.
A solid-state silicon battery or silicon-anode all-solid-state battery is a type of rechargeable lithium-ion battery consisting of a solid electrolyte, solid cathode, and silicon-based solid anode.

This is a history of the lithium-ion battery.
References
- 1 2 3 Zuleta, Juan Carlos (2020-12-20). "QuantumScape: A Technical Note On All-Solid-State 'Anode-Free' Lithium Metal Batteries (NYSE:QS) | Seeking Alpha". seekingalpha.com. Retrieved 2023-12-27.
- 1 2 Zhao, Pei; Pan, Jun; Zhang, Dongqi; Tang, Yufeng; Tai, Zhixin; Liu, Yajie; Gao, Hong; Huang, Fuqiang (2023-07-17). "Designs of Anode-Free Lithium-Ion Batteries". Batteries. 9 (7): 381. doi: 10.3390/batteries9070381 . ISSN 2313-0105.
- 1 2 3 Liu, Yuzhao; Meng, Xiangyu; Wang, Zhiyu; Qiu, Jieshan (2022-07-29). "Development of quasi-solid-state anode-free high-energy lithium sulfide-based batteries". Nature Communications. 13 (1): 4415. Bibcode:2022NatCo..13.4415L. doi:10.1038/s41467-022-32031-7. ISSN 2041-1723. PMC 9338099 . PMID 35906196.
- ↑ Oberhaus, Daniel (December 8, 2020). "Did QuantumScape Just Solve a 40-Year-Old Battery Problem?". Wired. ISSN 1059-1028 . Retrieved 2023-12-27.
- ↑ Manchester, Bette (30 November 2020). "QuantumScape successfully goes public". electrive.com.
- ↑ Kane, Mark (September 18, 2022). "ONE Reveals High Energy Dense Anode-Free Battery Cell: 1,007 Wh/L". InsideEVs. Retrieved 2023-12-28.
- ↑ Pacific Northwest National Laboratory. "Anode-Free Rechargeable Battery". www.pnnl.gov. Retrieved 2023-12-28.
- ↑ Lewis, Michelle (2024-03-06). "This anodeless, compressionless solid-state battery could be the next big thing". Electrek. Retrieved 2024-03-08.