Related Research Articles

A lithium-ion or Li-ion battery is a type of rechargeable battery that uses the reversible intercalation of Li+ ions into electronically conducting solids to store energy. In comparison with other commercial rechargeable batteries, Li-ion batteries are characterized by higher specific energy, higher energy density, higher energy efficiency, a longer cycle life, and a longer calendar life. Also noteworthy is a dramatic improvement in lithium-ion battery properties after their market introduction in 1991: within the next 30 years, their volumetric energy density increased threefold while their cost dropped tenfold.

A rechargeable battery, storage battery, or secondary cell, is a type of electrical battery which can be charged, discharged into a load, and recharged many times, as opposed to a disposable or primary battery, which is supplied fully charged and discarded after use. It is composed of one or more electrochemical cells. The term "accumulator" is used as it accumulates and stores energy through a reversible electrochemical reaction. Rechargeable batteries are produced in many different shapes and sizes, ranging from button cells to megawatt systems connected to stabilize an electrical distribution network. Several different combinations of electrode materials and electrolytes are used, including lead–acid, zinc–air, nickel–cadmium (NiCd), nickel–metal hydride (NiMH), lithium-ion (Li-ion), lithium iron phosphate (LiFePO4), and lithium-ion polymer.
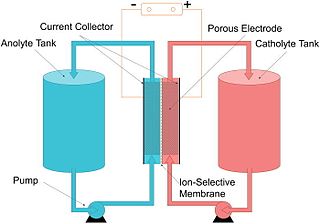
A flow battery, or redox flow battery, is a type of electrochemical cell where chemical energy is provided by two chemical components dissolved in liquids that are pumped through the system on separate sides of a membrane. Ion transfer inside the cell occurs through the membrane while both liquids circulate in their own respective space. Cell voltage is chemically determined by the Nernst equation and ranges, in practical applications, from 1.0 to 2.43 volts. The energy capacity is a function of the electrolyte volume and the power is a function of the surface area of the electrodes.

Batteries provided the primary source of electricity before the development of electric generators and electrical grids around the end of the 19th century. Successive improvements in battery technology facilitated major electrical advances, from early scientific studies to the rise of telegraphs and telephones, eventually leading to portable computers, mobile phones, electric cars, and many other electrical devices.

Lithium iron phosphate or lithium ferro-phosphate (LFP) is an inorganic compound with the formula LiFePO
4. It is a gray, red-grey, brown or black solid that is insoluble in water. The material has attracted attention as a component of lithium iron phosphate batteries, a type of Li-ion battery. This battery chemistry is targeted for use in power tools, electric vehicles, solar energy installations and more recently large grid-scale energy storage.

Lithium cobalt oxide, sometimes called lithium cobaltate or lithium cobaltite, is a chemical compound with formula LiCoO
2. The cobalt atoms are formally in the +3 oxidation state, hence the IUPAC name lithium cobalt(III) oxide.

A lithium-ion capacitor is a hybrid type of capacitor classified as a type of supercapacitor. It is called a hybrid because the anode is the same as those used in lithium-ion batteries and the cathode is the same as those used in supercapacitors. Activated carbon is typically used as the cathode. The anode of the LIC consists of carbon material which is often pre-doped with lithium ions. This pre-doping process lowers the potential of the anode and allows a relatively high output voltage compared to other supercapacitors.

The lithium–sulfur battery is a type of rechargeable battery. It is notable for its high specific energy. The low atomic weight of lithium and moderate atomic weight of sulfur means that Li–S batteries are relatively light. They were used on the longest and highest-altitude unmanned solar-powered aeroplane flight by Zephyr 6 in August 2008.

An electric battery is a source of electric power consisting of one or more electrochemical cells with external connections for powering electrical devices. When a battery is supplying power, its positive terminal is the cathode and its negative terminal is the anode. The terminal marked negative is the source of electrons that will flow through an external electric circuit to the positive terminal. When a battery is connected to an external electric load, a redox reaction converts high-energy reactants to lower-energy products, and the free-energy difference is delivered to the external circuit as electrical energy. Historically the term "battery" specifically referred to a device composed of multiple cells; however, the usage has evolved to include devices composed of a single cell.
The lithium–air battery (Li–air) is a metal–air electrochemical cell or battery chemistry that uses oxidation of lithium at the anode and reduction of oxygen at the cathode to induce a current flow.
A metal–air electrochemical cell is an electrochemical cell that uses an anode made from pure metal and an external cathode of ambient air, typically with an aqueous or aprotic electrolyte.

Sodium-ion batteries (NIBs, SIBs, or Na-ion batteries) are several types of rechargeable batteries, which use sodium ions (Na+) as its charge carriers. In some cases, its working principle and cell construction are similar to those of lithium-ion battery (LIB) types, but it replaces lithium with sodium as the intercalating ion. Sodium belongs to the same group in the periodic table as lithium and thus has similar chemical properties. Although, in some cases (such as aqueous Na-ion batteries) they are quite different from Li-ion batteries.
Aluminium-ion batteries are a class of rechargeable battery in which aluminium ions serve as charge carriers. Aluminium can exchange three electrons per ion. This means that insertion of one Al3+ is equivalent to three Li+ ions. Thus, since the ionic radii of Al3+ (0.54 Å) and Li+ (0.76 Å) are similar, significantly higher numbers of electrons and Al3+ ions can be accepted by cathodes with little damage. Al has 50 times (23.5 megawatt-hours m-3) the energy density of Li and is even higher than coal.
Research in lithium-ion batteries has produced many proposed refinements of lithium-ion batteries. Areas of research interest have focused on improving energy density, safety, rate capability, cycle durability, flexibility, and cost.
A magnesium sulfur battery is a rechargeable battery that uses magnesium ion as its charge carrier, magnesium metal as anode and sulfur as cathode. To increase the electronic conductivity of cathode, sulfur is usually mixed with carbon to form a cathode composite. Magnesium sulfur battery is an emerging energy storage technology and now is still in the stage of research. It is of great interest since in theory the Mg/S chemistry can provide 1722 Wh/kg energy density with a voltage at ~1.7 V.

Kuzhikalail M. Abraham is an American scientist, a recognized expert on lithium-ion and lithium-ion polymer batteries and is the inventor of the ultrahigh energy density lithium–air battery. Abraham is the principal of E-KEM Sciences in Needham, Massachusetts and a professor at the Northeastern University Center for Renewable Energy Technologies, Northeastern University, in Boston, Massachusetts.
Magnesium batteries are batteries that utilize magnesium cations as charge carriers and possibly in the anode in electrochemical cells. Both non-rechargeable primary cell and rechargeable secondary cell chemistries have been investigated. Magnesium primary cell batteries have been commercialised and have found use as reserve and general use batteries.
An aqueous lithium-ion battery is a lithium-ion battery (Li-ion) that uses a concentrated saline solution as an electrolyte to facilitate the transfer of lithium ions between electrodes and induce an electrical current. In contrast to non-aqueous lithium-ion batteries, aqueous Li-ion batteries are nonflammable and do not pose any significant risks of explosion, because of the water-based nature of their electrolyte. They also lack the poisonous chemicals and environmental risks associated with their non-aqueous counterparts.
Linda Faye Nazar is a Senior Canada Research Chair in Solid State Materials and Distinguished Research Professor of Chemistry at the University of Waterloo. She develops materials for electrochemical energy storage and conversion. Nazar demonstrated that interwoven composites could be used to improve the energy density of lithium–sulphur batteries. She was awarded the 2019 Chemical Institute of Canada Medal.

This is a history of the lithium-ion battery.
References
- 1 2 3 4 Liang & Yao 2022, p. 110.
- 1 2 Liang & Yao 2022, p. 111.
- ↑ Pistola 2013, pp. 33–34.
- 1 2 3 Chao et al. 2020, p. 1.
- 1 2 Tahir, Agarwal & Csóka 2020, p. 379.
- 1 2 Liang & Yao 2022, p. 112.
- ↑ Suo, Liumin; Borodin, Oleg; Gao, Tao; Olguin, Marco; Ho, Janet; Fan, Xiulin; Luo, Chao; Wang, Chunsheng; Xu, Kang (20 November 2015). ""Water-in-salt" electrolyte enables high-voltage aqueous lithium-ion chemistries". Science. 350 (6263): 938–943. doi:10.1126/science.aab1595.
- ↑ Pistola 2013, p. 33.
- ↑ Tahir, Agarwal & Csóka 2020.