Arsenic is a chemical element; it has symbol As and atomic number 33. Arsenic occurs in many minerals, usually in combination with sulfur and metals, but also as a pure elemental crystal. Arsenic is a notoriously toxic metalloid. It has various allotropes, but only the grey form, which has a metallic appearance, is important to industry.

Arsenic poisoning is a medical condition that occurs due to elevated levels of arsenic in the body. If arsenic poisoning occurs over a brief period of time, symptoms may include vomiting, abdominal pain, encephalopathy, and watery diarrhea that contains blood. Long-term exposure can result in thickening of the skin, darker skin, abdominal pain, diarrhea, heart disease, numbness, and cancer.
A period 4 element is one of the chemical elements in the fourth row of the periodic table of the chemical elements. The periodic table is laid out in rows to illustrate recurring (periodic) trends in the chemical behaviour of the elements as their atomic number increases: a new row is begun when chemical behaviour begins to repeat, meaning that elements with similar behaviour fall into the same vertical columns. The fourth period contains 18 elements beginning with potassium and ending with krypton – one element for each of the eighteen groups. It sees the first appearance of d-block in the table.

Arsenic trioxide is an inorganic compound with the formula As
2O
3. As an industrial chemical, its major uses include the manufacture of wood preservatives, pesticides, and glass. It is sold under the brand name Trisenox among others when used as a medication to treat a type of cancer known as acute promyelocytic leukemia. For this use it is given by injection into a vein.
Lead hydrogen arsenate, also called lead arsenate, acid lead arsenate or LA, chemical formula PbHAsO4, is an inorganic insecticide used primarily against the potato beetle. Lead arsenate was the most extensively used arsenical insecticide. Two principal formulations of lead arsenate were marketed: basic lead arsenate (Pb5OH(AsO4)3, CASN: 1327-31-7) and acid lead arsenate (PbHAsO4).

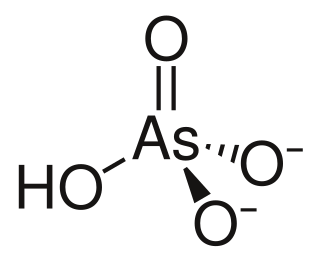
Arsenic acid or arsoric acid is the chemical compound with the formula H3AsO4. More descriptively written as AsO(OH)3, this colorless acid is the arsenic analogue of phosphoric acid. Arsenate and phosphate salts behave very similarly. Arsenic acid as such has not been isolated, but is only found in solution, where it is largely ionized. Its hemihydrate form (2H3AsO4·H2O) does form stable crystals. Crystalline samples dehydrate with condensation at 100 °C.
In chemistry, an arsenite is a chemical compound containing an arsenic oxyanion where arsenic has oxidation state +3. Note that in fields that commonly deal with groundwater chemistry, arsenite is used generically to identify soluble AsIII anions. IUPAC have recommended that arsenite compounds are to be named as arsenate(III), for example ortho-arsenite is called trioxidoarsenate(III). Ortho-arsenite contrasts to the corresponding anions of the lighter members of group 15, phosphite which has the structure HPO2−3 and nitrite, NO−2 which is bent.

Arsenic pentoxide is the inorganic compound with the formula As2O5. This glassy, white, deliquescent solid is relatively unstable, consistent with the rarity of the As(V) oxidation state. More common, and far more important commercially, is arsenic(III) oxide (As2O3). All inorganic arsenic compounds are highly toxic and thus find only limited commercial applications.

Copper arsenate (Cu3(AsO4)2·4H2O, or Cu5H2(AsO4)4·2H2O), also called copper orthoarsenate, tricopper arsenate, cupric arsenate, or tricopper orthoarsenate, is a blue or bluish-green powder insoluble in water and alcohol and soluble in aqueous ammonium and dilute acids. Its CAS number is 7778-41-8 or 10103-61-4.

Scheele's Green, also called Schloss Green, is chemically a cupric hydrogen arsenite, CuHAsO
3. It is chemically related to Paris Green. Scheele's Green was invented in 1775 by Carl Wilhelm Scheele. By the end of the 19th century, it had virtually replaced the older green pigments based on copper carbonate. It is a yellowish-green pigment commonly used during the early to mid-19th century in paints as well as being directly incorporated into a variety of products as a colorant. It began to fall out of favor after the 1860s because of its toxicity and the instability of its color in the presence of sulfides and various chemical pollutants. The acutely toxic nature of Scheele's green as well as other arsenic-containing green pigments such as Paris Green may have contributed to the sharp decline in the popularity of the color green in late Victorian society. By the dawn of the 20th century, Scheele's green had completely fallen out of use as a pigment but was still in use as an insecticide into the 1930s. At least two modern reproductions of Scheele's green hue with modern non-toxic pigments have been made, with similar but non-identical color coordinates: one with hex#3c7a18 and another with hex#478800. The latter is the more typically reported color coordinate for Scheele's green.
Chromated copper arsenate (CCA) is a wood preservative containing compounds of chromium, copper, and arsenic, in various proportions. It is used to impregnate timber and other wood products, especially those intended for outdoor use, in order to protect them from attack by microbes and insects. Like other copper-based wood preservatives, it imparts a greenish tint to treated timber.
The uranyl ion is an oxycation of uranium in the oxidation state +6, with the chemical formula UO2+
2. It has a linear structure with short U–O bonds, indicative of the presence of multiple bonds between uranium and oxygen. Four or more ligands may be bound to the uranyl ion in an equatorial plane around the uranium atom. The uranyl ion forms many complexes, particularly with ligands that have oxygen donor atoms. Complexes of the uranyl ion are important in the extraction of uranium from its ores and in nuclear fuel reprocessing.

Molybdenum blue is a term applied to:

Sodium arsenate is the inorganic compound with the formula Na3AsO4. Related salts are also called sodium arsenate, including Na2HAsO4 (disodium hydrogen arsenate) and NaH2AsO4 (sodium dihydrogen arsenate). The trisodium salt is a white or colourless solid that is highly toxic. It is usually handled as the dodecahydrate Na3AsO4.12H2O.
Arsenic biochemistry refers to biochemical processes that can use arsenic or its compounds, such as arsenate. Arsenic is a moderately abundant element in Earth's crust, and although many arsenic compounds are often considered highly toxic to most life, a wide variety of organoarsenic compounds are produced biologically and various organic and inorganic arsenic compounds are metabolized by numerous organisms. This pattern is general for other related elements, including selenium, which can exhibit both beneficial and deleterious effects. Arsenic biochemistry has become topical since many toxic arsenic compounds are found in some aquifers, potentially affecting many millions of people via biochemical processes.
Ammonium orthomolybdate is the inorganic compound with the chemical formula (NH4)2MoO4. It is a white solid that is prepared by treating molybdenum trioxide with aqueous ammonia. Upon heating these solutions, ammonia is lost, to give ammonium heptamolybdate ((NH4)6Mo7O24·4H2O).
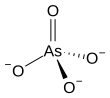
Arsenate-reducing bacteria are bacteria which reduce arsenates. Arsenate-reducing bacteria are ubiquitous in arsenic-contaminated groundwater (aqueous environment). Arsenates are salts or esters of arsenic acid (H3AsO4), consisting of the ion AsO43−. They are moderate oxidizers that can be reduced to arsenites and to arsine. Arsenate can serve as a respiratory electron acceptor for oxidation of organic substrates and H2S or H2. Arsenates occur naturally in minerals such as adamite, alarsite, legrandite, and erythrite, and as hydrated or anhydrous arsenates. Arsenates are similar to phosphates since arsenic (As) and phosphorus (P) occur in group 15 (or VA) of the periodic table. Unlike phosphates, arsenates are not readily lost from minerals due to weathering. They are the predominant form of inorganic arsenic in aqueous aerobic environments. On the other hand, arsenite is more common in anaerobic environments, more mobile, and more toxic than arsenate. Arsenite is 25–60 times more toxic and more mobile than arsenate under most environmental conditions. Arsenate can lead to poisoning, since it can replace inorganic phosphate in the glyceraldehyde-3-phosphate --> 1,3-biphosphoglycerate step of glycolysis, producing 1-arseno-3-phosphoglycerate instead. Although glycolysis continues, 1 ATP molecule is lost. Thus, arsenate is toxic due to its ability to uncouple glycolysis. Arsenate can also inhibit pyruvate conversion into acetyl-CoA, thereby blocking the TCA cycle, resulting in additional loss of ATP.
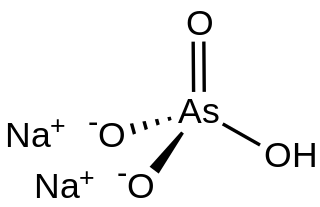
Disodium hydrogen arsenate is the inorganic compound with the formula Na2HAsO4.7H2O. The compound consists of a salt and seven molecules of water of crystallization although for simplicity the formula usually omits the water component. The other sodium arsenates are NaH2AsO4 and Na3AsO4, the latter being called sodium arsenate. Disodium hydrogen arsenate is highly toxic. The salt is the conjugate base of arsenic acid. It is a white, water-soluble solid.
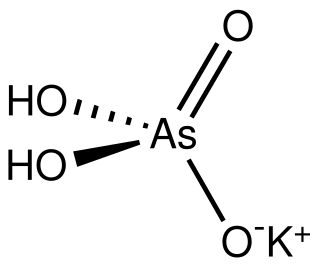
Monopotassium arsenate is the inorganic compound with the formula KH2AsO4. A white solid, this salt is used to prepared other arsenic-containing compounds, mainly pesticides. It is prepared by calcining arsenic oxide and potassium nitrate, followed by extraction with water.

Compounds of arsenic resemble in some respects those of phosphorus which occupies the same group (column) of the periodic table. The most common oxidation states for arsenic are: −3 in the arsenides, which are alloy-like intermetallic compounds, +3 in the arsenites, and +5 in the arsenates and most organoarsenic compounds. Arsenic also bonds readily to itself as seen in the square As3−
4 ions in the mineral skutterudite. In the +3 oxidation state, arsenic is typically pyramidal owing to the influence of the lone pair of electrons.