An oxyanion, or oxoanion, is an ion with the generic formula A
xOz−
y. Oxyanions are formed by a large majority of the chemical elements. The formulae of simple oxyanions are determined by the octet rule. The corresponding oxyacid of an oxyanion is the compound H
zA
xO
y. The structures of condensed oxyanions can be rationalized in terms of AOn polyhedral units with sharing of corners or edges between polyhedra. The oxyanions adenosine monophosphate (AMP), adenosine diphosphate (ADP) and adenosine triphosphate (ATP) are important in biology.
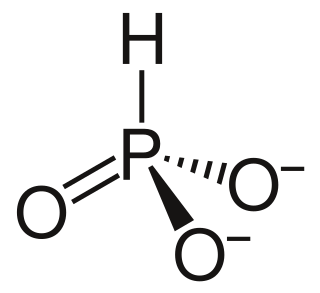
A phosphite anion or phosphite in inorganic chemistry usually refers to [HPO3]2− but includes [H2PO3]− ([HPO2(OH)]−). These anions are the conjugate bases of phosphorous acid (H3PO3). The corresponding salts, e.g. sodium phosphite (Na2HPO3) are reducing in character.

Lead(II) sulfide is an inorganic compound with the formula PbS. Galena is the principal ore and the most important compound of lead. It is a semiconducting material with niche uses.

Lead(II) oxide, also called lead monoxide, is the inorganic compound with the molecular formula PbO. PbO occurs in two polymorphs: litharge having a tetragonal crystal structure, and massicot having an orthorhombic crystal structure. Modern applications for PbO are mostly in lead-based industrial glass and industrial ceramics, including computer components. It is an amphoteric oxide.

Lead(II) chloride (PbCl2) is an inorganic compound which is a white solid under ambient conditions. It is poorly soluble in water. Lead(II) chloride is one of the most important lead-based reagents. It also occurs naturally in the form of the mineral cotunnite.

Phosphorus trichloride is a inorganic compound with the chemical formula PCl3. A colorless liquid when pure, it is an important industrial chemical, being used for the manufacture of phosphites and other organophosphorus compounds. It is toxic and reacts violently with water to release hydrogen chloride.
Phosphorous acid is the compound described by the formula H3PO3. This acid is diprotic (readily ionizes two protons), not triprotic as might be suggested by this formula. Phosphorous acid is an intermediate in the preparation of other phosphorus compounds. Organic derivatives of phosphorous acid, compounds with the formula RPO3H2, are called phosphonic acids.
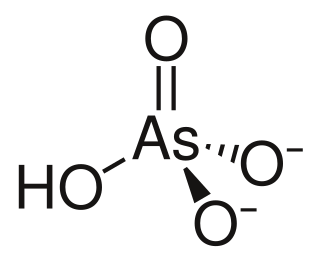
Lead hydrogen arsenate, also called lead arsenate, acid lead arsenate or LA, chemical formula PbHAsO4, is an inorganic insecticide used primarily against the potato beetle. Lead arsenate was the most extensively used arsenical insecticide. Two principal formulations of lead arsenate were marketed: basic lead arsenate (Pb5OH(AsO4)3, CASN: 1327-31-7) and acid lead arsenate (PbHAsO4.
In chemistry, a plumbate often refers to compounds that can be viewed as derivatives of the hypothetical [PbO3]2- anion. The term also refers to any anion of lead or any salt thereof. So the term is vague and somewhat archaic.

In industrial chemistry, a stabilizer or stabiliser is a chemical that is used to prevent degradation.

Phosphinates or hypophosphites are a class of phosphorus compounds conceptually based on the structure of hypophosphorous acid. IUPAC prefers the term phosphinate in all cases, however in practice hypophosphite is usually used to describe inorganic species, while phosphinate typically refers to organophosphorus species.
Dicalcium phosphate is the calcium phosphate with the formula CaHPO4 and its dihydrate. The "di" prefix in the common name arises because the formation of the HPO42– anion involves the removal of two protons from phosphoric acid, H3PO4. It is also known as dibasic calcium phosphate or calcium monohydrogen phosphate. Dicalcium phosphate is used as a food additive, it is found in some toothpastes as a polishing agent and is a biomaterial.
Lead(II) carbonate is the chemical compound PbCO3. It is a white solid with several practical uses, despite its toxicity. It occurs naturally as the mineral cerussite.
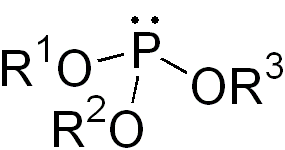
In chemistry a phosphite ester or organophosphite usually refers to an organophosphorous compound with the formula P(OR)3. They can be considered as esters of an unobserved tautomer phosphorous acid, H3PO3, with the simplest example being trimethylphosphite, P(OCH3)3. Some phosphites can be considered esters of the dominant tautomer of phosphorous acid (HP(O)(OH)2). The simplest representative is dimethylphosphite with the formula HP(O)(OCH3)2. Both classes of phosphites are usually colorless liquids.

Dipotassium phosphate (K2HPO4) (also dipotassium hydrogen orthophosphate; potassium phosphate dibasic) is the inorganic compound with the formula K2HPO4.(H2O)x (x = 0, 3, 6). Together with monopotassium phosphate (KH2PO4.(H2O)x), it is often used as a fertilizer, food additive, and buffering agent. It is a white or colorless solid that is soluble in water.
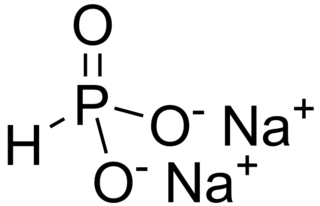
Disodium hydrogen phosphite is the name for inorganic compounds with the formula Na2HPO3.(H2O)x. The commonly encountered salt is the pentahydrate. A derivative of phosphorous acid (HP(O)(OH)2), it contains the anion HPO32−. Its common name suggests that it contains an acidic hydrogen atom, as in sodium hydrogen carbonate. However, this name is misleading as the hydrogen atom is not acidic, being bonded to phosphorus rather than oxygen. The salt has reducing properties. It is white or colorless solid, and is little studied.
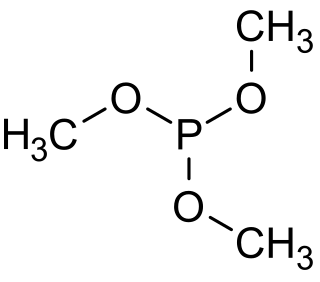
Trimethyl phosphite is an organophosphorus compound with the formula P(OCH3)3, often abbreviated P(OMe)3. It is a colorless liquid with a highly pungent odor. It is used as a ligand in organometallic chemistry and as a reagent in organic synthesis. The molecule features a pyramidal phosphorus(III) center bound to three methoxy groups.

Lead(II) bromide is the inorganic compound with the formula PbBr2. It is a white powder. It is produced in the burning of typical leaded gasolines.
Monofluorophosphate is an anion with the formula PO3F2−, which is a phosphate group with one oxygen atom substituted with a fluoride atom. The charge of the ion is −2. The ion resembles sulfate in size, shape and charge, and can thus form compounds with the same structure as sulfates. These include Tutton's salts and langbeinites. The most well-known compound of monofluorophosphate is sodium monofluorophosphate, commonly used in toothpaste.

The oxalate phosphates are chemical compounds containing oxalate and phosphate anions. They are also called oxalatophosphates or phosphate oxalates. Some oxalate-phosphate minerals found in bat guano deposits are known. Oxalate phosphates can form metal organic framework compounds.

















