Related Research Articles
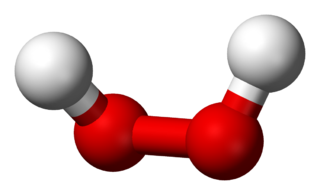
Hydrogen peroxide is a chemical compound with the formula H2O2. In its pure form, it is a very pale blue liquid that is slightly more viscous than water. It is used as an oxidizer, bleaching agent, and antiseptic, usually as a dilute solution in water for consumer use, and in higher concentrations for industrial use. Concentrated hydrogen peroxide, or "high-test peroxide", decomposes explosively when heated and has been used both as a monopropellant and an oxidizer in rocketry.
In toxicology, the median lethal dose, LD50 (abbreviation for "lethal dose, 50%"), LC50 (lethal concentration, 50%) or LCt50 is a toxic unit that measures the lethal dose of a toxin, radiation, or pathogens. The value of LD50 for a substance is the dose required to kill half the members of a tested population after a specified test duration. LD50 figures are frequently used as a general indicator of a substance's acute toxicity. A lower LD50 is indicative of increased toxicity.
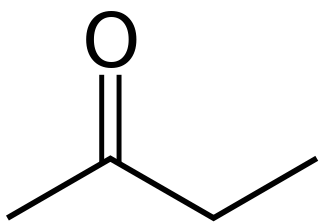
Butanone, also known as methyl ethyl ketone (MEK), is an organic compound with the formula CH3C(O)CH2CH3. This colourless liquid ketone has a sharp, sweet odor reminiscent of acetone. It is produced industrially on a large scale, but occurs in nature only in trace amounts. It is partially soluble in water, and is commonly used as an industrial solvent. It is an isomer of another solvent, tetrahydrofuran.
Cyclohexene is a hydrocarbon with the formula C6H10. This cycloalkene is a colorless liquid with a sharp smell. It is an intermediate in various industrial processes. Cyclohexene is not very stable upon long term storage with exposure to light and air because it forms peroxides.

Butan-2-ol, or sec-butanol, is an organic compound with formula CH3CH(OH)CH2CH3. Its structural isomers are 1-butanol, isobutanol, and tert-butanol. 2-Butanol is chiral and thus can be obtained as either of two stereoisomers designated as (R)-(−)-butan-2-ol and (S)-(+)-butan-2-ol. It is normally encountered as a 1:1 mixture of the two stereoisomers — a racemic mixture.
This page provides supplementary chemical data on carbon monoxide.

Cumene (isopropylbenzene) is an organic compound that contains a benzene ring with an isopropyl substituent. It is a constituent of crude oil and refined fuels. It is a flammable colorless liquid that has a boiling point of 152 °C. Nearly all the cumene that is produced as a pure compound on an industrial scale is converted to cumene hydroperoxide, which is an intermediate in the synthesis of other industrially important chemicals, primarily phenol and acetone.
This page provides supplementary chemical data on phosphorus trichloride.
This page provides supplementary chemical data on Menthol.
International Chemical Safety Cards (ICSC) are data sheets intended to provide essential safety and health information on chemicals in a clear and concise way. The primary aim of the Cards is to promote the safe use of chemicals in the workplace and the main target users are therefore workers and those responsible for occupational safety and health. The ICSC project is a joint venture between the World Health Organization (WHO) and the International Labour Organization (ILO) with the cooperation of the European Commission (EC). This project began during the 1980s with the objective of developing a product to disseminate the appropriate hazard information on chemicals at the workplace in an understandable and precise way.

The Globally Harmonized System of Classification and Labelling of Chemicals (GHS) is an internationally agreed-upon standard managed by the United Nations that was set up to replace the assortment of hazardous material classification and labelling schemes previously used around the world. Core elements of the GHS include standardized hazard testing criteria, universal warning pictograms, and harmonized safety data sheets which provide users of dangerous goods with a host of information. The system acts as a complement to the UN Numbered system of regulated hazardous material transport. Implementation is managed through the UN Secretariat. Although adoption has taken time, as of 2017, the system has been enacted to significant extents in most major countries of the world. This includes the European Union, which has implemented the United Nations' GHS into EU law as the CLP Regulation, and United States Occupational Safety and Health Administration standards.

The Hazardous Materials Identification System (HMIS) is a numerical hazard rating that incorporates the use of labels with color developed by the American Coatings Association as a compliance aid for the OSHA Hazard Communication (HazCom) Standard.

Barium peroxide is an inorganic compound with the formula BaO2. This white solid (gray when impure) is one of the most common inorganic peroxides, and it was the first peroxide compound discovered. Being an oxidizer and giving a vivid green colour upon ignition (as do all barium compounds), it finds some use in fireworks; historically, it was also used as a precursor for hydrogen peroxide.
This page provides supplementary chemical data on barium nitrate.

Vinyl fluoride is an organic halide with the chemical formula C2H3F. It is a colorless gas with a faint ether-like odor. It is used as the monomeric precursor to the fluoropolymer polyvinylfluoride.
This page provides supplementary chemical data on Hexafluoroethane.
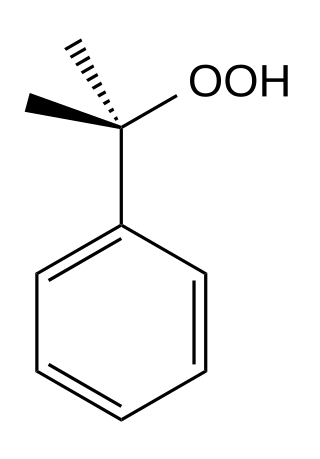
Cumene hydroperoxide is the organic compound with the formula C6H5C(CH3)2OOH. An oily liquid, it is classified as an organic hydroperoxide. Products of decomposition of cumene hydroperoxide are methylstyrene, acetophenone, and 2-Phenyl-2-propanol.
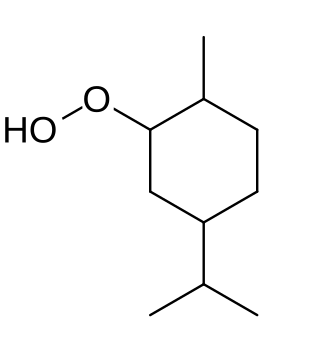
Paramenthane hydroperoxide (PMHP) is an organic peroxide with a distinctive odor. It is used on an industrial scale as a polymerization initiator for emulsion polymerizations. It is usually sold in a light yellow liquid solutions of about 50% strength.
Sodium bifluoride is the inorganic compound with the formula Na[HF2]. It is a salt of sodium cation and bifluoride anion. It is a white, water-soluble solid that decomposes upon heating. Sodium bifluoride is non-flammable, hygroscopic, and has a pungent smell. Sodium bifluoride has a number of applications in industry.
References
- ↑ Shreve, O. D.; Heether, M. R.; Knight, H. B.; Swern, Daniel (February 1951). "Infrared Absorption Spectra of Some Hydroperoxides, Peroxides, and Related Compounds". Analytical Chemistry. 23 (2): 282–285. doi:10.1021/ac60050a015.