In chemistry, an ester is a compound derived from an oxoacid in which at least one hydroxyl group is replaced by an alkoxy group, as in the substitution reaction of a carboxylic acid and an alcohol. Glycerides are fatty acid esters of glycerol; they are important in biology, being one of the main classes of lipids and comprising the bulk of animal fats and vegetable oils.

In chemistry, an acyl group is a moiety derived by the removal of one or more hydroxyl groups from an oxoacid, including inorganic acids. It contains a double-bonded oxygen atom and an alkyl group. In organic chemistry, the acyl group is usually derived from a carboxylic acid, in which case it has the formula RCO−, where R represents an alkyl group that is linked to the carbon atom of the group by a single bond. Although the term is almost always applied to organic compounds, acyl groups can in principle be derived from other types of acids such as sulfonic acids and phosphonic acids. In the most common arrangement, acyl groups are attached to a larger molecular fragment, in which case the carbon and oxygen atoms are linked by a double bond.

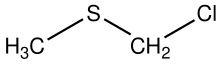
In organic chemistry, an organic sulfide or thioether is an organosulfur functional group with the connectivity R−S−R' as shown on right. Like many other sulfur-containing compounds, volatile sulfides have foul odors. A sulfide is similar to an ether except that it contains a sulfur atom in place of the oxygen. The grouping of oxygen and sulfur in the periodic table suggests that the chemical properties of ethers and sulfides are somewhat similar, though the extent to which this is true in practice varies depending on the application.
In organic chemistry, an acyl chloride is an organic compound with the functional group −C(=O)Cl. Their formula is usually written R−COCl, where R is a side chain. They are reactive derivatives of carboxylic acids. A specific example of an acyl chloride is acetyl chloride, CH3COCl. Acyl chlorides are the most important subset of acyl halides.
In chemistry, halogenation is a chemical reaction that entails the introduction of one or more halogens into a compound. Halide-containing compounds are pervasive, making this type of transformation important, e.g. in the production of polymers, drugs. This kind of conversion is in fact so common that a comprehensive overview is challenging. This article mainly deals with halogenation using elemental halogens (F2, Cl2, Br2, I2). Halides are also commonly introduced using salts of the halides and halogen acids. Many specialized reagents exist for and introducing halogens into diverse substrates, e.g. thionyl chloride.

An organic acid anhydride is an acid anhydride that is an organic compound. An acid anhydride is a compound that has two acyl groups bonded to the same oxygen atom. A common type of organic acid anhydride is a carboxylic anhydride, where the parent acid is a carboxylic acid, the formula of the anhydride being (RC(O))2O. Symmetrical acid anhydrides of this type are named by replacing the word acid in the name of the parent carboxylic acid by the word anhydride. Thus, (CH3CO)2O is called acetic anhydride. Mixed (or unsymmetrical) acid anhydrides, such as acetic formic anhydride (see below), are known, whereby reaction occurs between two different carboxylic acids. Nomenclature of unsymmetrical acid anhydrides list the names of both of the reacted carboxylic acids before the word "anhydride" (for example, the dehydration reaction between benzoic acid and propanoic acid would yield "benzoic propanoic anhydride").

In organic chemistry, a sulfoxide, also called a sulphoxide, is an organosulfur compound containing a sulfinyl functional group attached to two carbon atoms. It is a polar functional group. Sulfoxides are oxidized derivatives of sulfides. Examples of important sulfoxides are alliin, a precursor to the compound that gives freshly crushed garlic its aroma, and dimethyl sulfoxide (DMSO), a common solvent.

Trimethylsilyldiazomethane is the organosilicon compound with the formula (CH3)3SiCHN2. It is classified as a diazo compound. Trimethylsilyldiazomethane is a commercially available reagent used in organic chemistry as a methylating agent and as a source of CH2 group. Its behavior is akin to the less convenient reagent diazomethane.
Chloroalkyl ethers are a class of organic compounds with the general structure R-O-(CH2)n-Cl, characterized as an ether connected to a chloromethyl group via an alkane chain.
The Blanc chloromethylation is the chemical reaction of aromatic rings with formaldehyde and hydrogen chloride to form chloromethyl arenes. The reaction is catalyzed by Lewis acids such as zinc chloride. The reaction was discovered by Gustave Louis Blanc (1872-1927) in 1923

1,3,5-Trithiane is the chemical compound with the formula (CH2S)3. This heterocycle is the cyclic trimer of the otherwise unstable species thioformaldehyde. It consists of a six-membered ring with alternating methylene bridges and thioether groups. It is prepared by treatment of formaldehyde with hydrogen sulfide.
Acetyl iodide is an organoiodine compound with the formula CH3COI. It is a colourless liquid. It is formally derived from acetic acid. Although far rarer in the laboratory than the related acetyl bromide and acetyl chloride, acetyl iodide is produced, transiently at least, on a far larger scale than any other acid halide. Specifically, it is generated by the carbonylation of methyl iodide in the Cativa and Monsanto processes, which are the main industrial processes that generate acetic acid. It is also an intermediate in the production of acetic anhydride from methyl acetate.

Chloromethyl methyl ether (CMME) is a compound with formula CH3OCH2Cl. A colorless liquid, it is a chloroalkyl ether. It is used as an alkylating agent. In organic synthesis, it is used for introducing the methoxymethyl ether (MOM) protecting group, and is thus often called MOM-Cl or MOM chloride. It also finds application as a chloromethylating agent in some variants of the Blanc chloromethylation.
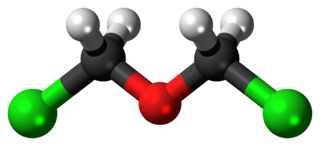
Bis(chloromethyl) ether is an organic compound with the chemical formula (CH2Cl)2O. It is a colourless liquid with an unpleasant suffocating odour and it is one of the chloroalkyl ethers. Bis(chloromethyl) ether was once produced on a large scale, but was found to be highly carcinogenic and thus such production has ceased.
In organic chemistry, a methoxymethyl ether is a functional group with the formula ROCH2OCH3, abbreviated MOM. Methoxymethyl ethers are often employed in organic synthesis to protect alcohols. They are usually derived from 2-methoxymethyl chloride. Closely related to MOM ethers are methoxyethoxymethoxy (MEM) protecting groups, introduced using 2-methoxyethoxymethyl chloride. The MEM protecting groups are more easily installed and more easily removed.
Allyl methyl sulfide is an organosulfur compound with the chemical formula CH2=CHCH2SCH3. The molecule features two functional groups, an allyl (CH2=CHCH2) and a sulfide. It is a colourless liquid with a strong odor characteristic of alkyl sulfides. It is a metabolite of garlic, and "garlic breath" is attributed to its presence.
Dimethyl disulfide (DMDS) is an organic chemical compound with the molecular formula CH3SSCH3 which is the simplest disulfide. It is a flammable liquid with an unpleasant, garlic-like odor.
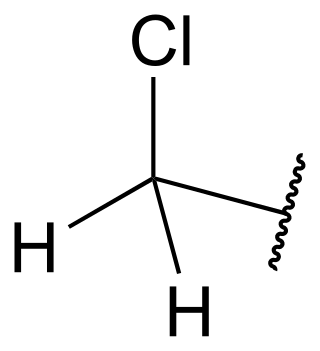
In organic chemistry, the chloromethyl group is a functional group that has the chemical formula −CH2−Cl. The naming of this group is derived from the methyl group, by replacing one hydrogen atom by a chlorine atom. Compounds with this group are a subclass of the organochlorines.

(Trimethylsilyl)methyl chloride is the organosilicon compound with the formula (CH3)3SiCH2Cl. A colorless, volatile liquid, it is an alkylating agent that is employed in organic synthesis, especially as a precursor to (trimethylsilyl)methyllithium. In the presence of triphenylphosphine, it olefinates benzophenones:
2-Methoxyethoxymethyl chloride is an organic compound with formula CH3OCH2CH2OCH2Cl. A colorless liquid, it is classified as a chloroalkyl ether. It is used as an alkylating agent. In organic synthesis, it is used for introducing the methoxyethoxy ether (MEM) protecting group. MEM protecting groups are generally preferred to methoxymethyl (MOM) protecting groups, both in terms of formation and removal.