Antioxidants are compounds that inhibit oxidation, a chemical reaction that can produce free radicals. Autoxidation leads to degradation of organic compounds, including living matter. Antioxidants are frequently added to industrial products, such as polymers, fuels, and lubricants, to extend their useable lifetimes. Food are also treated with antioxidants to forestall spoilage, in particular the rancidification of oils and fats. In cells, antioxidants such as glutathione, mycothiol or bacillithiol, and enzyme systems like superoxide dismutase, can prevent damage from oxidative stress.
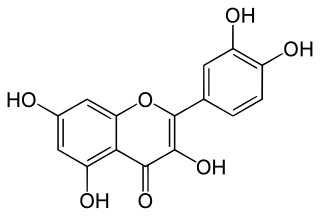
Quercetin is a plant flavonol from the flavonoid group of polyphenols. It is found in many fruits, vegetables, leaves, seeds, and grains; capers, red onions, and kale are common foods containing appreciable amounts of it. It has a bitter flavor and is used as an ingredient in dietary supplements, beverages, and foods.

Angiomotin (AMOT) is a protein that in humans is encoded by the AMOT gene. It belongs to the motin family of angiostatin binding proteins, which includes angiomotin, angiomotin-like 1 (AMOTL1) and angiomotin-like 2 (AMOTL2) characterized by coiled-coil domains at N-terminus and consensus PDZ-binding domain at the C-terminus. Angiomotin is expressed predominantly in endothelial cells of capillaries as well as angiogenic tissues such as placenta and solid tumor.
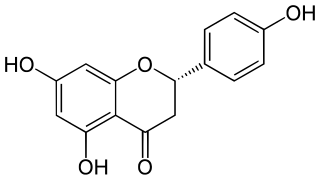
Naringenin is a flavorless, colorless flavanone, a type of flavonoid. It is the predominant flavanone in grapefruit, and is found in a variety of fruits and herbs.

Safranal is an organic compound isolated from saffron, the spice consisting of the stigmas of crocus flowers. It is the constituent primarily responsible for the aroma of saffron.
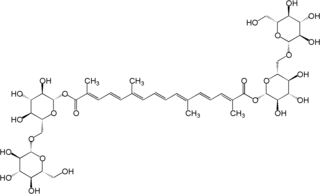
Crocin is a carotenoid chemical compound that is found in the flowers of crocus and gardenia. Its oxygen content also chemically makes it a xanthene. Crocin is the chemical primarily responsible for the color of saffron.
Cytochalasins are fungal metabolites that have the ability to bind to actin filaments and block polymerization and the elongation of actin. As a result of the inhibition of actin polymerization, cytochalasins can change cellular morphology, inhibit cellular processes such as cell division, and even cause cells to undergo apoptosis. Cytochalasins have the ability to permeate cell membranes, prevent cellular translocation and cause cells to enucleate. Cytochalasins can also have an effect on other aspects of biological processes unrelated to actin polymerization. For example, cytochalasin A and cytochalasin B can also inhibit the transport of monosaccharides across the cell membrane, cytochalasin H has been found to regulate plant growth, cytochalasin D inhibits protein synthesis and cytochalasin E prevents angiogenesis.

Myricetin is a member of the flavonoid class of polyphenolic compounds, with antioxidant properties. Common dietary sources include vegetables, fruits, nuts, berries, tea, and red wine. Myricetin is structurally similar to fisetin, luteolin, and quercetin and is reported to have many of the same functions as these other members of the flavonol class of flavonoids. Reported average intake of myricetin per day varies depending on diet, but has been shown in the Netherlands to average 23 mg/day.

Genistein (C15H10O5) is a naturally occurring compound that structurally belongs to a class of compounds known as isoflavones. It is described as an angiogenesis inhibitor and a phytoestrogen.

Cofilin 1 , also known as CFL1, is a human gene, part of the ADF/cofilin family.

A polyphenol antioxidant is a hypothetical type of antioxidant containing a polyphenolic substructure and studied in vitro. Numbering over 4,000 distinct species mostly from plants, polyphenols may have antioxidant activity in vitro, but are unlikely to be antioxidants in vivo. Hypothetically, they may affect cell-to-cell signaling, receptor sensitivity, inflammatory enzyme activity or gene regulation, although high-quality clinical research has not confirmed any of these possible effects in humans as of 2020.
Actibind is an actin-binding fungal T(2)-RNase protein that is produced by the black mold Aspergillus niger, a microorganism used in biotechnology and food technology. In plants, actibind binds actin, a major component of the cytoskeleton, interfering with the plants' pollen tubes and halting cell growth. Research published in the journal Cancer on 15 May 2006 reports evidence that actibind has antiangiogenic and anticarcinogenic characteristics. In human colon cancer, breast cancer and melanoma, increasing the level of actibind was found to reduce the ability of these cells to form tumorogenic colonies. In animal models, increased actibind inhibited the growth of colon cancer-derived tumors, metastases and blood vessel formation. During the completion of the Human Genome Project, the gene encoding for RNaseT2, the human actibind-like protein, was found on chromosome 6.

Triterpenes are a class of terpenes composed of six isoprene units with the molecular formula C30H48; they may also be thought of as consisting of three terpene units. Animals, plants and fungi all produce triterpenes, including squalene, the precursor to all steroids.

ROCK1 is a protein serine/threonine kinase also known as rho-associated, coiled-coil-containing protein kinase 1. Other common names are ROKβ and P160ROCK. ROCK1 is a major downstream effecter of the small GTPase RhoA and is a regulator of the actomyosin cytoskeleton which promotes contractile force generation. ROCK1 plays a role in cancer and in particular cell motility, metastasis, and angiogenesis.

Invadopodia are actin-rich protrusions of the plasma membrane that are associated with degradation of the extracellular matrix in cancer invasiveness and metastasis. Very similar to podosomes, invadopodia are found in invasive cancer cells and are important for their ability to invade through the extracellular matrix, especially in cancer cell extravasation. Invadopodia are generally visualized by the holes they create in ECM -coated plates, in combination with immunohistochemistry for the invadopodia localizing proteins such as cortactin, actin, Tks5 etc. Invadopodia can also be used as a marker to quantify the invasiveness of cancer cell lines in vitro using a hyaluronic acid hydrogel assay.

Deacetylasperulosidic acid is an iridoid compound found in a few medicinal plants, such as Morinda citrifolia. Some in vitro and in vivo bioactivities of deacetylasperulosidic acid include anti-inflammatory, analgesic, anti-cancer, antioxidant, anti-arthritic, anti-mutagenic, anti-clastogenic, and hepatoprotection.
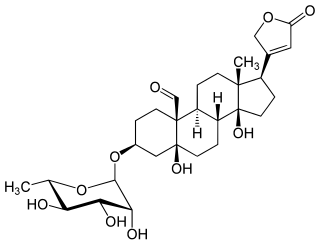
Convallatoxin is a glycoside extracted from Convallaria majalis.

Withaferin A is a steroidal lactone, derived from Acnistus arborescens, Withania somnifera and other members of family Solanaceae. It has been traditionally used in ayurvedic medicine. It is the first member of the withanolide class of ergostane type product to be discovered. This natural product has wide range of pharmacological activities including cardioprotective, anti-inflammatory, immuno-modulatory, anti-angiogenesis, anti-metastasis and anti-carcinogenic properties.
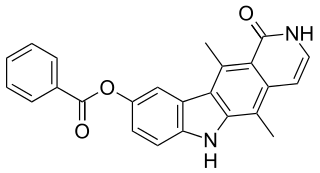
Pyr1 (LIMINIB) is an organic compound composed of carbon, hydrogen, oxygen and nitrogen that inhibits the enzyme LIM kinase.

2-Hydroxyestradiol (2-OHE2), also known as estra-1,3,5(10)-triene-2,3,17β-triol, is an endogenous steroid, catechol estrogen, and metabolite of estradiol, as well as a positional isomer of estriol.




















