
A carboxylic acid is an organic acid that contains a carboxyl group (C(=O)OH) attached to an R-group. The general formula of a carboxylic acid is R–COOH or R-CO2H, with R referring to the alkyl, alkenyl, aryl, or other group. Carboxylic acids occur widely. Important examples include the amino acids and fatty acids. Deprotonation of a carboxylic acid gives a carboxylate anion.

Adipic acid or hexanedioic acid is the organic compound with the formula (CH2)4(COOH)2. From an industrial perspective, it is the most important dicarboxylic acid: about 2.5 billion kilograms of this white crystalline powder are produced annually, mainly as a precursor for the production of nylon. Adipic acid otherwise rarely occurs in nature, but it is known as manufactured E number food additive E355.

Dimethyl terephthalate (DMT) is an organic compound with the formula C6H4(COOCH3)2. It is the diester formed from terephthalic acid and methanol. It is a white solid that melts to give a distillable colourless liquid.

Dimethyl sulfite is a sulfite ester with the chemical formula (CH3O)2SO.

Adipates are the salts and esters of adipic acid. The anionic (HO2C(CH2)4CO2−) and dianionic (−O2C(CH2)4CO2−) forms of adipic acid are also referred to as adipate.
Dioctyl adipate (DOA) is an organic compound with the formula (CH2CH2CO2C8H17)2. It is a colorless oily liquid. As well as related diesters derived from 2-ethylhexanol, decanol, isodecanol, etc., it is used as a plasticizer.

Potassium adipate is a compound with formula K2C6H8O4. It is the potassium salt of adipic acid.
Sodium adipate is a compound with formula Na2C6H8O4. It is the sodium salt of adipic acid.

Dimethyl acetylenedicarboxylate (DMAD) is an organic compound with the formula CH3O2CC2CO2CH3. It is a di-ester in which the ester groups are conjugated with a C-C triple bond. As such, the molecule is highly electrophilic, and is widely employed as a dienophile in cycloaddition reactions, such as the Diels-Alder reaction. It is also a potent Michael acceptor. This compound exists as a colorless liquid at room temperature. This compound was used in the preparation of nedocromil.

Dimethyl carbonate (DMC) is an organic compound with the formula OC(OCH3)2. It is a colourless, flammable liquid. It is classified as a carbonate ester. This compound has found use as a methylating agent and more recently as a solvent that is exempt from the restrictions placed on most volatile organic compounds (VOCs) in the US. Dimethyl carbonate is often considered to be a green reagent.
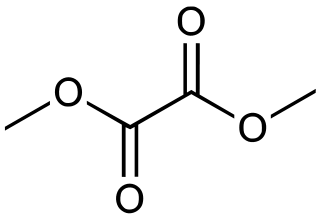
Dimethyl oxalate is the organic compound with the formula (CO2CH3)2. It is the dimethyl ester of oxalic acid. Dimethyl oxalate is a colorless or white solid that is soluble in water.

Dimethyl malonate is a diester derivative of malonic acid. It is a common reagent for organic synthesis used, for example, as a precursor for barbituric acid. It is also used in the malonic ester synthesis. It can be synthesized from dimethoxymethane and carbon monoxide.
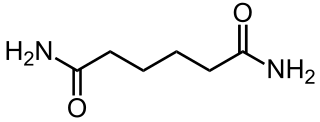
Adipamide is the organic compound with the formula (CH2CH2C(O)NH2)2. It is a white solid. The dominant commercial interest in adipamides is related to their presence in nylons.
PBAT is a biodegradable random copolymer, specifically a copolyester of adipic acid, 1,4-butanediol and terephthalic acid. PBAT is produced by many different manufacturers and may be known by the brand names ecoflex, Wango,Ecoworld, Eastar Bio, and Origo-Bi. It is also called poly(butylene adipate-co-terephthalate) and sometimes polybutyrate-adipate-terephthalate or even just "polybutyrate". It is generally marketed as a fully biodegradable alternative to low-density polyethylene, having many similar properties including flexibility and resilience, allowing it to be used for many similar uses such as plastic bags and wraps. The structure of the PBAT polymer is shown to the right. It is depicted as a block co-polymer here due to the common synthetic method of first synthesizing two copolymer blocks and then combining them. However, it is important to note that the actual structure of the polymer is a random co-polymer of the blocks shown.

Ammonium adipate is a compound with formula (NH4)2(C4H8(COO)2). It is the ammonium salt of adipic acid. It is used as a food additive and has the E number E359.
Dibasic ester or DBE is an ester of a dicarboxylic acid. Depending on the application, the alcohol may be methanol or higher molecular weight monoalcohols.
Bis(2-ethylhexyl) adipate or DEHA is an organic compound with the formula (CH2CH2CO2C8H17)2. It is the diester of 2-ethylhexanol and adipic acid. It is a colorless oily liquid.
1,6-Hexanediol is an organic compound with the formula (CH2CH2CH2OH)2. It is a colorless water-soluble solid.

Poly(ethylene adipate) or PEA is an aliphatic polyester. It is most commonly synthesized from a polycondensation reaction between ethylene glycol and adipic acid. PEA has been studied as it is biodegradable through a variety of mechanisms and also fairly inexpensive compared to other polymers. Its lower molecular weight compared to many polymers aids in its biodegradability.

2-Methylglutaronitrile is the organic compound with the formula NCCH2CH2CH(CH3)CN. This dinitrile is obtained in the large-scale synthesis of adiponitrile. It is a colorless liquid with an unpleasant odor. It is the starting compound for the vitamin nicotinamide and for the diester dimethyl-2-methylglutarate and the ester amide methyl 5-(dimethylamino)-2-methyl-5-oxopentanoate, which are promoted as green solvents. 2-Methylglutaronitrile is chiral but is mainly encountered as the racemate.














