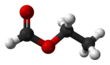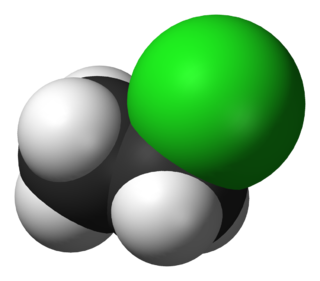
Chloroethane, commonly known as ethyl chloride, is a chemical compound with chemical formula CH3CH2Cl, once widely used in producing tetraethyllead, a gasoline additive. It is a colorless, flammable gas or refrigerated liquid with a faintly sweet odor.

Ethyl acetate is the organic compound with the formula CH3CO2CH2CH3, simplified to C4H8O2. This colorless liquid has a characteristic sweet smell and is used in glues, nail polish removers, and in the decaffeination process of tea and coffee. Ethyl acetate is the ester of ethanol and acetic acid; it is manufactured on a large scale for use as a solvent.

Acetic anhydride, or ethanoic anhydride, is the chemical compound with the formula (CH3CO)2O. Commonly abbreviated Ac2O, it is the simplest isolable anhydride of a carboxylic acid and is widely used as a reagent in organic synthesis. It is a colorless liquid that smells strongly of acetic acid, which is formed by its reaction with moisture in the air.

Ethylbenzene is an organic compound with the formula C6H5CH2CH3. It is a highly flammable, colorless liquid with an odor similar to that of gasoline. This monocyclic aromatic hydrocarbon is important in the petrochemical industry as a reaction intermediate in the production of styrene, the precursor to polystyrene, a common plastic material. In 2012, more than 99% of ethylbenzene produced was consumed in the production of styrene.

Hydrogen selenide is an inorganic compound with the formula H2Se. This hydrogen chalcogenide is the simplest and most commonly encountered hydride of selenium. H2Se is a colorless, flammable gas under standard conditions. It is the most toxic selenium compound with an exposure limit of 0.05 ppm over an 8-hour period. Even at extremely low concentrations, this compound has a very irritating smell resembling that of decayed horseradish or "leaking gas", but smells of rotten eggs at higher concentrations.
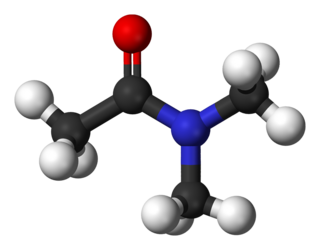
Dimethylacetamide (DMAc or DMA) is the organic compound with the formula CH3C(O)N(CH3)2. This colorless, water-miscible, high-boiling liquid is commonly used as a polar solvent in organic synthesis. DMA is miscible with most other solvents, although it is poorly soluble in aliphatic hydrocarbons.

The chemical compound isobutyl acetate, also known as 2-methylpropyl ethanoate or β-methylpropyl acetate, is a common solvent. It is produced from the esterification of isobutanol with acetic acid. It is used as a solvent for lacquer and nitrocellulose. Like many esters it has a fruity or floral smell at low concentrations and occurs naturally in raspberries, pears and other plants. At higher concentrations the odor can be unpleasant and may cause symptoms of central nervous system depression such as nausea, dizziness and headache.
2-Chloroethanol (also called ethylene chlorohydrin or glycol chlorohydrin) is an organic chemical compound with the chemical formula HOCH2CH2Cl and the simplest beta-halohydrin (chlorohydrin). This colorless liquid has a pleasant ether-like odor. It is miscible with water. The molecule is bifunctional, consisting of both an alkyl chloride and an alcohol functional group.
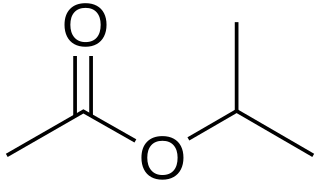
Isopropyl acetate is an ester, an organic compound which is the product of esterification of acetic acid and isopropanol. It is a clear, colorless liquid with a characteristic fruity odor.

2-Ethoxyethanol, also known by the trademark Ethyl cellosolve, is a solvent used widely in commercial and industrial applications. It is a clear, colorless, nearly odorless liquid that is miscible with water, ethanol, diethyl ether, acetone, and ethyl acetate.

An occupational hazard is a hazard experienced in the workplace. This encompasses many types of hazards, including chemical hazards, biological hazards (biohazards), psychosocial hazards, and physical hazards. In the United States, the National Institute for Occupational Safety and Health (NIOSH) conduct workplace investigations and research addressing workplace health and safety hazards resulting in guidelines. The Occupational Safety and Health Administration (OSHA) establishes enforceable standards to prevent workplace injuries and illnesses. In the EU, a similar role is taken by EU-OSHA.

Glycidol is an organic compound that contains both epoxide and alcohol functional groups. Being bifunctional, it has a variety of industrial uses. The compound is a slightly viscous liquid that is slightly unstable and is not often encountered in pure form.

Dioxathion, systematically known as p-dioxane-2,3-diyl ethyl phosphorodithioate, is an organophosphate pesticide. It is used as an insecticide on livestock and as an acaricide on citrus fruits, deciduous fruits and nuts.
A recommended exposure limit (REL) is an occupational exposure limit that has been recommended by the United States National Institute for Occupational Safety and Health. The REL is a level that NIOSH believes would be protective of worker safety and health over a working lifetime if used in combination with engineering and work practice controls, exposure and medical monitoring, posting and labeling of hazards, worker training and personal protective equipment. To formulate these recommendations, NIOSH evaluates all known and available medical, biological, engineering, chemical, trade, and other information. Although not legally enforceable limits, RELS are transmitted to the Occupational Safety and Health Administration (OSHA) or the Mine Safety and Health Administration (MSHA) of the U.S. Department of Labor for use in promulgating legal standards.
The US National Institute for Occupational Safety and Health funds the Adult Blood Lead Epidemiology and Surveillance (ABLES) program, a state-based surveillance program of laboratory-reported adult blood lead levels. In 2009, the ABLES program updated its case definition for an Elevated Blood Lead Level to a blood lead concentration equal or greater than 10 micrograms per deciliter (10 μg/dL). This chart shows CDC/NIOSH/ABLES Elevated blood lead level case definition in perspective.
Workplace health surveillance or occupational health surveillance (U.S.) is the ongoing systematic collection, analysis, and dissemination of exposure and health data on groups of workers. The Joint ILO/WHO Committee on Occupational Health at its 12th Session in 1995 defined an occupational health surveillance system as "a system which includes a functional capacity for data collection, analysis and dissemination linked to occupational health programmes".

Manure management refers to capture, storage, treatment, and utilization of animal manures in an environmentally sustainable manner. It can be retained in various holding facilities. Animal manure can occur in a liquid, slurry, or solid form. It is utilized by distribution on fields in amounts that enrich soils without causing water pollution or unacceptably high levels of nutrient enrichment. Manure management is a component of nutrient management.

Diethylethanolamine (DEAE) is the organic compound with the molecular formula (C2H5)2NCH2CH2OH. A colorless liquid, is used as a precursor in the production of a variety of chemical commodities such as the local anesthetic procaine.

Parathion methyl, or methyl parathion, is an organophosphate insecticide, possessing an organothiophosphate group. It is structurally very similar to parathion-ethyl. It is not allowed for sale and import in nearly all countries around the world, while a few allow it under subject to specified conditions only.
Occupational hazards of fire debris cleanup are the hazards to health and safety of the personnel tasked with clearing the area of debris and combustion products after a conflagration. Once extinguished, fire debris cleanup poses several safety and health risks for workers. Employers responsible for fire debris cleanup and other work in areas damaged or destroyed by fire are generally obliged by occupational safety and health legislation of the relevant national or regional authority to identify and evaluate hazards, correct any unsafe or unhealthy conditions and provide any necessary training and instruction and personal protective equipment to employees to enable them to carry out the task without undue exposure to hazards. Many of the approaches to control risk in occupational settings can be applied to preventing injuries and disease. This type of work can be completed by general construction firms who may not be fully trained specifically for fire safety and on fire hazards.