
Acetyl-CoA carboxylase (ACC) is a biotin-dependent enzyme that catalyzes the irreversible carboxylation of acetyl-CoA to produce malonyl-CoA through its two catalytic activities, biotin carboxylase (BC) and carboxyltransferase (CT). ACC is a multi-subunit enzyme in most prokaryotes and in the chloroplasts of most plants and algae, whereas it is a large, multi-domain enzyme in the cytoplasm of most eukaryotes. The most important function of ACC is to provide the malonyl-CoA substrate for the biosynthesis of fatty acids. The activity of ACC can be controlled at the transcriptional level as well as by small molecule modulators and covalent modification. The human genome contains the genes for two different ACCs—ACACA and ACACB.

Fedotozine is an opioid drug which acts as a peripherally specific selective κ1-opioid receptor agonist with preference for the κ1A subtype. It was under investigation for the treatment of gastrointestinal conditions like irritable bowel syndrome and functional dyspepsia and made it to phase III clinical trials, but ultimately development was discontinued and it was never marketed.

Elafibranor is an experimental medication that is being studied and developed by Genfit for the treatment of cardiometabolic diseases including diabetes, insulin resistance, dyslipidemia, and non-alcoholic fatty liver disease (NAFLD).

Seladelpar is a PPARδ receptor agonist that is being investigated for drug use by Metabolex. According to a press release they are examining its potential use for the treatment of dyslipidemia, metabolic syndrome, type 2 diabetes, and non-alcoholic steatohepatitis (NASH). The compound was licensed from Janssen Pharmaceutica NV. The drug completed a phase II trial for primary biliary cholangitis. "Seladelpar demonstrated robust, dose-dependent, clinically significant, and durable improvements in biochemical markers of cholestasis and inflammation in patients with PBC at risk of disease progression. Seladelpar appeared safe and well tolerated and was not associated with any increase in pruritus." A phase III trial in patients with PBC also found reduced pruritus and improved liver biochemistry, despite being terminated early.

A deuterated drug is a small molecule medicinal product in which one or more of the hydrogen atoms contained in the drug molecule have been replaced by its heavier stable isotope deuterium. Because of the kinetic isotope effect, deuterium-containing drugs may have significantly lower rates of metabolism, and hence a longer half-life.

Radalbuvir is an experimental antiviral drug for the treatment of hepatitis C virus (HCV) infection developed by Gilead Sciences. Radalbuvir acts as an NS5B inhibitor. It is currently in clinical trials. It targets NS5B polymerase.
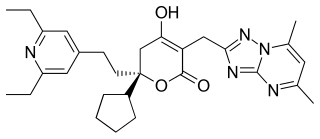
Filibuvir was a non-nucleoside orally available NS5B inhibitor developed by Pfizer for the treatment of hepatitis C. It binds to the non-catalytic Thumb II allosteric pocket of NS5B viral polymerase and causes a decrease in viral RNA synthesis. It is a potent and selective inhibitor, with a mean IC50 of 0.019 μM against genotype 1 polymerases. Several filibuvir-resistant mutations have been identified, M423 being the most common that occurred after filibuvir monotherapy. It was intended to be taken twice-daily.

Mericitabine (RG-7128) is an antiviral drug, a deoxycytidine analog. It was developed as a treatment for hepatitis C, acting as a NS5B RNA polymerase inhibitor, but while it showed a good safety profile in clinical trials, it was not sufficiently effective to be used as a stand-alone agent. However mericitabine has been shown to boost the efficacy of other antiviral drugs when used alongside them, and as most modern treatment regimens for hepatitis C use a combination therapy of several antiviral drugs, clinical trials have continued to see if it can form a part of a clinically useful drug treatment program.

Emricasan is a potential drug invented in 1998 by Idun Pharmaceuticals. The drug was acquired by Pfizer in 2005 and then sold to Conatus Pharmaceuticals in 2010. Conatus in turn licensed emricasan to Novartis in 2017 for exclusive development and commercialization.

Daprodustat, sold under the brand name Duvroq among others, is a medication that is used for the treatment of anemia due to chronic kidney disease. It is a hypoxia-inducible factor prolyl hydroxylase inhibitor. It is taken by mouth.
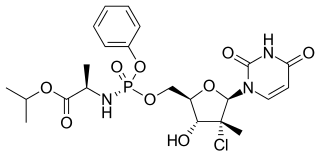
Uprifosbuvir (MK-3682) is an antiviral drug developed for the treatment of hepatitis C. It is a nucleotide analogue which acts as an NS5B RNA polymerase inhibitor. As of 2019 it was in Phase III human clinical trials.
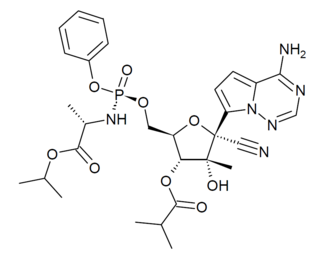
GS-6620 is an antiviral drug which is a nucleotide analogue. It was developed for the treatment of Hepatitis C but while it showed potent antiviral effects in early testing, it could not be successfully formulated into an oral dosage form due to low and variable absorption in the intestines which made blood levels unpredictable. It has however continued to be researched as a potential treatment for other viral diseases such as Ebola virus disease.
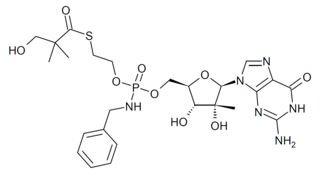
IDX-184 is an antiviral drug which was developed as a treatment for hepatitis C, acting as a NS5B RNA polymerase inhibitor. While it showed reasonable effectiveness in early clinical trials it did not progress past Phase IIb. However research using this drug has continued as it shows potentially useful activity against other emerging viral diseases such as Zika virus, and coronaviruses including MERS, and SARS-CoV-2.

CPI-0610 is a drug which acts as a BET inhibitor, mainly acting at the BRD2 and BRD4 subtypes. It has potential applications in the treatment of various forms of cancer.

Nidufexor (LMB-763) is a drug which acts as a partial agonist of the farnesoid X receptor (FXR). It has reached Phase II clinical trials for the treatment of diabetic nephropathy and nonalcoholic steatohepatitis.
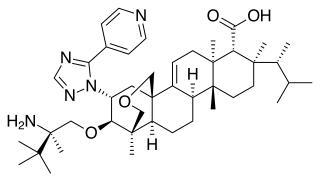
Ibrexafungerp, sold under the brand name Brexafemme, is an antifungal medication used to treat vulvovaginal candidiasis (VVC). It is taken orally. It is also currently undergoing clinical trials for other indications via an intravenous (IV) formulation. An estimated 75% of women will have at least one episode of VVC and 40 to 45% will have two or more episodes in their lifetime.

Sofpironium bromide is a drug used to treat hyperhidrosis. It was approved in Japan in 2020 as a 5% gel for the treament of primary axillary hyperhidrosis (PAH).
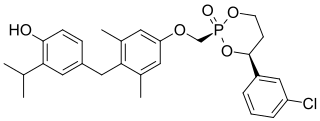
VK2809 is a thyromimetic prodrug whose active form is selective for the THR-β isoform. It is being developed by Viking Therapeutics in a phase II trial for the treatment of nonalcoholic steatohepatitis and is also being investigated for glycogen storage disease type Ia.
Thyromimetic drugs are synthetic agonists of the thyroid hormone receptor's isoforms TR α1, TRα2, TRβ1, or TRβ2, mimicking some or all of the effects of endogenously produced thyroid hormones that regulate metabolism. Some thyromimetic drugs are selective for various of these receptors over others, enabling more targeted effects and reducing toxicity. Thyromimetics selective for TRβ—including eprotirome, sobetirome, resmetirom, and the prodrug VK2809—have been investigated for the treatment of non-alcoholic fatty liver disease, dyslipidemia, and other metabolic and neurodegenerative diseases.
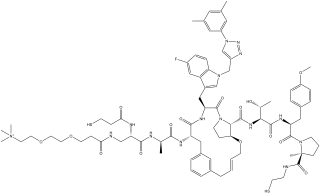
MK-0616 is a macrocyclic peptide investigational drug who is being evaluated for the treatment of hypercholesterolaemia. It is a PCSK9 inhibitor.



















